Introduction
Somatostatinoma is a rare neuroendocrine tumor (NET) with an incidence of 1 in 40 million individuals[1] and accounts for less than 5% of pancreatic NETs (pNETs).[2] The tumor was first described in 1977 in a 46-year-old woman in the head of the pancreas.[3] The tumor originates from the delta cells of the pancreas and predominantly contains somatostatin with trace quantities of other pancreatic hormones such as insulin, glucagon, gastrin, and vasoactive intestinal polypeptide. It remains localized to the pancreas in 56 to 70% of the cases, out of which 36% occur in the head, 14% in the pancreatic body, and 32% in the tail.[4] Other common sites include duodenum (19%), ampulla of Vater (3%), and small bowel (3%).[5] Reports exist of rare instances of extra-gastrointestinal primaries in the lungs, kidneys, and thyroid.[6]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Somatostatinoma can be sporadic or may occur in association with neurocutaneous syndromes such as multiple endocrine neoplasia type 1 (the commonest, 40 to 50% of cases), neurofibromatosis type 1 (NF1), and Von Hippel-Lindau syndrome.[4][6] Moreover, a variety of chromosomal aberrations, including loss of heterozygosity at chromosome 11q, 6q, and allelic loss at chromosome 3p, are implicated in the pathogenesis of this disease.[4]
Epidemiology
Somatostatinoma commonly presents in late adulthood between 40 and 60 years of age with no gender predilection.[7][5]
Pathophysiology
Somatostatinoma can be functional or non-functional. Non-functional tumors are common in the duodenum and are identified incidentally or from mechanical obstruction of the biliary tree.[8][9][1] Functional or secretory tumors are common in the pancreas and are diagnosed with the clinical effects of somatostatin.[9] Somatostatin exerts an inhibitory effect on pituitary hormones such as growth hormone and thyroid-stimulating hormone.[5] In the neuro gastrointestinal system, somatostatin suppresses the release of glucagon, pancreatic polypeptide, cholecystokinin, gastrin, secretin, cholecystokinin, vasoactive intestinal polypeptide, gastric inhibitory polypeptide, motilin, glucagon, and neurotensin.[5] It has a direct inhibitory effect on gastric acid secretion, splanchnic perfusion, gastric emptying, gallbladder contractility, bile flow, and bowel motility.[5][10]
Histopathology
Pancreatic tumors are generally larger and more malignant than their duodenal counterparts.[11] Under light microscopy, most tumors show well-differentiated islet cell or carcinoid tumor morphology, while some show a mixed pattern of differentiated and anaplastic cells.[7] Psammoma bodies are concentric lamellated calcified structures commonly visualized in duodenal tumors more than the pancreatic tumors.[12] Neurosecretory granules are visible in the cytoplasm surrounding the Golgi complex and endoplasmic reticulum under electron microscopy[4], and immunohistochemistry is typically positive for somatostatin and non-specific neuroendocrine markers such as synaptophysin, neuron-specific enolase, chromogranin, and serotonin.[1][7] In one-fourth of the cases, the tumor is positive for other pancreatic hormones such as insulin, calcitonin, gastrin, glucagon, and vasoactive intestinal polypeptide.[6] Immunohistochemistry for Ki-67 (MIB-1) is mandatory to grade the tumor based on WHO classification.[13] WHO has classified gastro-entero-pancreatic tumors into grade 1 – Ki-67 less than or equal to 2%, grade 2 – Ki-67 3% to 20%, grade 3 – Ki-67 over 20%, mixed adenocarcinoma, and neuroendocrine carcinoma.[14]
History and Physical
Non-functional tumors are frequently asymptomatic. Later in the course of the disease, they may present with abdominal pain, vomiting, jaundice, and steatorrhea due to the mechanical effects of the tumor on the biliary and pancreatic drainage.[11] Functional tumors present with the inhibitory effects of somatostatin on the neuroendocrine system. Cholelithiasis is present in almost 70%, and diabetes mellitus in 60% of symptomatic cases.[4] Rarely, they manifest as a triad of diabetes mellitus, cholelithiasis, and steatorrhea, referred to as inhibitory syndrome due to the suppression of insulin, cholecystokinin, and pancreatic exocrine enzymes, respectively.[5][6] The physical examination might reveal icterus in the patients presenting with obstructive jaundice, abdominal tenderness in acute cholecystitis, and neurocutaneous markers such as cafe au lait spots, neurofibromas, and axillary freckling in patients with concomitant NF1.
Evaluation
Early diagnosis of somatostatinoma can be difficult as non-functional tumors often produce no symptoms, and functional tumors present with non-specific symptoms that can masquerade other clinical conditions. Hence most non-functional tumors are discovered incidentally or during the workup of vague gastrointestinal symptoms.[4] Functional tumors typically have elevated fasting serum somatostatin levels (greater than 14 mmol/l).[4] However, it is important to recognize that serum somatostatin levels may also increase in medullary thyroid cancer, lung cancer, pheochromocytoma, and paraganglioma.[15] 24-hour urine level of 5-hydroxy indole acetic acid (5-HIAA), the breakdown product of serotonin, has been used as an alternative in some centers, but the level can be affected by a variety of dietary items and drugs.[16]
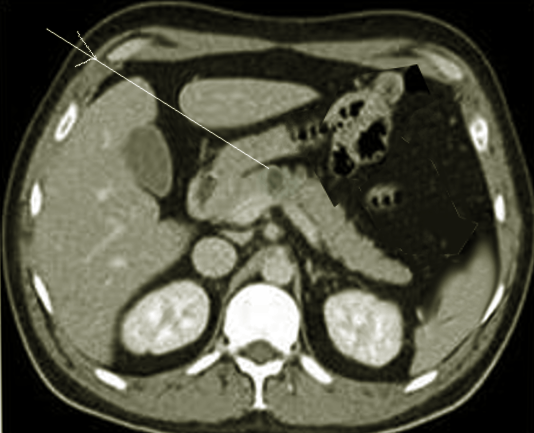
Since somatostatinoma usually presents late, tumors are large at diagnosis and are easily visualized using imaging modalities such as CT, MRI, or MRCP.[4] Multiphasic contrast-enhanced CT abdomen is the initial investigation of choice due to the non-invasiveness and the widespread availability.[17] MRI is increasingly in use where pNETs have a low signal density on T1-weighted images and high signal density on T2 weighted images.[18] MRI also has a higher sensitivity than CT to detect small liver metastases.[17] Procedures such as esophagogastroduodenoscopy (EGD), endoscopic retrograde cholangiopancreatography (ERCP), or endoscopic ultrasound (EUS) can be performed to allow luminal visualization and biopsy of the tumor to confirm the diagnosis. EUS offers high sensitivity to detect lesions as small as 0.5 cm and is best indicated for lesions in the head of the pancreas and lesions of the duodenal wall.[4][17]
Treatment / Management
Treatment depends upon the functionality, operability, and staging of the tumor. Surgery with the excision of the primary tumor and all the metastasized lymph nodes is the only curative treatment.[19] Tumors, where nearly 90% are excisable, are treated with curative resection only.[20] Tumors with local and locoregional (liver and lymph node) metastases receive treatment with tumor debulking surgeries and hepatic resection to relieve symptoms secondary to mechanical obstruction and heavy tumor load.[13][20] Depending on the location of the primary tumor, pylorus-preserving proximal pancreaticoduodenectomy, distal pancreatectomy with splenectomy, and Whipple’s procedure are some of the frequently performed procedures.[1][3][21] Grade 3 pNETs are often widely metastasized at diagnosis, thus are generally rendered inoperable.[13] (A1)
Isolated liver metastasis should have treatment with resection or selective hepatic transcatheter arterial embolization (TAE).[17] Prospective trials have shown that long-term survival significantly increases with early and aggressive surgical resection of hepatic metastases.[22] In cases of partially resectable multicentric disease or inoperable liver metastases, ablative procedures such as radiofrequency ablation (RFA), laser-induced thermotherapy, TAE, transcatheter arterial chemoembolization (TACE), and selective internal radiotherapy (SIRT) are employed in addition to systemic chemotherapy to downstage the tumor.[23]
Differential Diagnosis
Symptoms related to the obstruction of the biliary tree require differentiation from other extrahepatic causes of cholestasis such as choledocholithiasis, choledochal cyst, sclerosing cholangitis, cholangiocarcinoma, pancreatic pseudocysts, and pancreatic adenocarcinoma. Similarly, symptoms secondary to pancreatic insufficiencies such as steatorrhea and insulin-dependent diabetes mellitus should raise suspicion of somatostatinoma in addition to chronic pancreatitis and other pancreatic tumors.
Medical Oncology
Somatostatin analogs such as octreotide and lanreotide are indicated in functional tumors of any size.[13][24][25] They are also recommended as first-line therapy in nonfunctioning grade 1/2 NETs.[13] Prospective randomized phase III studies have shown that targeted therapy such as tyrosine kinase inhibitor sunitinib malate and mTOR inhibitor everolimus lengthen the time to tumor progression compared to placebo in advanced, low-grade, and intermediate-grade pNET, respectively.[26][27][26]
Chemotherapy is recommended in metastatic grade 2 and any grade 3 tumors. Streptozocin with 5–fluorouracil or doxorubicin is recommended in grade 2 NETs.[13] A multicenter prospective study has shown Streptozocin and doxorubicin to be more effective than Streptozocin and 5-fluorouracil in advanced pNETs.[28] Temozolomide and capecitabine were found to be effective in metastatic, well, or moderately differentiated pancreatic endocrine carcinoma in a retrospective uncontrolled study but the comparative effect with streptozocin combinations remains unknown as yet.[29] Similarly, dacarbazine has demonstrated clinical response in one-third of the patients with advanced pNET in a phase II uncontrolled trial.[30] Platinum-based agents combined with etoposide or irinotecan have proven efficacy in poorly differentiated grade 3 tumors and not well-differentiated tumors in a phase II trial.[13][31]
Staging
Around 75% of the tumors are metastatic at presentation, with liver involvement early in the course of the disease.[4] There are reports of distant metastasis involving the liver.[12] Staging occurs through functional imaging tests such as somatostatin receptor scintigraphy (octreotide scan) and positron emission tomography (PET).[13] Gallium-68 DOTATATE PET has a sensitivity of 94.5% and specificity of 97% for pNETs. They are particularly sensitive to detect bony metastasis compared to octreotide scans (95% vs. 15%, p < 0.001).[32] As per the American Joint Committee of Cancer (AJCC), pNETs classified into four stages: stage 1 – tumor limited to pancreas, stage 2 – tumor extends beyond pancreas not involving superior mesenteric artery or regional lymph node metastasis, stage 3 – involvement of the celiac axis or superior mesenteric artery, stage 4 – distant metastasis.[33] Endocrine tumors of the foregut classify into the following: stage 1 – size less than or equal to 1 cm with invasion of lamina propria or submucosa; stage 2 – tumors size > 1 cm or invades muscularis propria, pancreas, or retroperitoneum; stage 3 – metastasis of regional lymph node, peritoneum or adjacent structures; stage 4 – distant metastasis.[34]
Prognosis
The 5-year survival rate of patients with pancreatic or periampullary somatostatinoma is 60 to 100% with localized disease and 15 to 60% with metastatic disease.[1] Prognosis depends on the size of the tumor, the degree of cytological differentiation, the amount of residual tumor post-resection, and the extent of metastasis.[35][36] In patients who underwent resection, the 5-year survival rates for stages I, II, III, and IV were 89.9%, 82.6%, 75.8%, and 56.9%, respectively.[37] In a retrospective study on patients with liver metastases, the presence of bone metastasis significantly decreased the median survival (15.4–62.1 vs. 18.2–166.3 months, p < 0.0001).[38]
Complications
Systemic complications include diabetes mellitus, cholelithiasis, iron deficiency anemia, macrocytic anemia, deficiency manifestations of fat-soluble vitamins A, D, E, and K such as night blindness, osteopenia, and bleeding, respectively. Diabetes mellitus is usually mild, and ketoacidosis is rare. It is treated with diet and/or oral hypoglycemic agents or with small doses of insulin.[7] Even if patients do not present with cholelithiasis, prophylactic cholecystectomy is often performed during the initial surgical exploration due to the high prevalence of cholelithiasis among these patients, especially if somatostatin analogs are indicated for treatment.[24]
Locally, the tumor can lead to obstructive hyperbilirubinemia and gastrointestinal bleeding.
Postoperative and Rehabilitation Care
Follow-up is recommended for a minimum of 10 years.[19] For fully resected, well-differentiated, or moderately differentiated stage I – III gastroenteropancreatic NETs, consensus recommendation from international multidisciplinary working group exists to guide surveillance using cross-sectional imaging and biomarker testing.[19] Follow-up with cross-sectional imaging (triple-phase CT of the abdomen) or MRI is recommended for pancreatic and midgut NET annually for the first three years, then every 1 to 2 years.[19] More frequent surveillance is recommended for the Ki-67 mitotic index greater than 10% or lymph node positivity.[19] Follow-up is not routinely recommended for pancreatic tumors with stage I or grade 1 tumors and midgut NETS, which are incidental TNM stage 1 or grade 1.[19] Biomarkers are not routinely recommended for surveillance.[19] European Neuroendocrine Tumor Society (ENETS) recommends functional imaging tests at baseline and every 2 years in grade 1, at 3 months, and yearly for grade 2 to 3 pNETs.[39]
Deterrence and Patient Education
Provider awareness and patient education are necessary for prompt diagnosis of the disease and early initiation of treatment. Reliable online resources such as the American Cancer Society can provide evidence-based information to providers and patients.
Pearls and Other Issues
The rarity of the tumor and the wide variation in the non-specific symptomatology requires a high index of suspicion for initial diagnosis. Early diagnosis is imperative as a significant majority of these tumors are metastatic at presentation.
Enhancing Healthcare Team Outcomes
Advanced cases of somatostatinoma with hepatic metastases often require an interprofessional approach inclusive of a gastroenterologist, surgeon, and oncologist to evaluate the prognosis and treatment choices between curative procedures and palliative therapy. Pharmacologists review treatment with somatostatin analogs or chemotherapy. Specialty-trained nurses assist with surgery, monitor patients preoperatively, and administer treatment.[40] [Level 5]
Media
References
Williamson JM, Thorn CC, Spalding D, Williamson RC. Pancreatic and peripancreatic somatostatinomas. Annals of the Royal College of Surgeons of England. 2011 Jul:93(5):356-60. doi: 10.1308/003588411X582681. Epub [PubMed PMID: 21943457]
Level 3 (low-level) evidencede Wilde RF, Edil BH, Hruban RH, Maitra A. Well-differentiated pancreatic neuroendocrine tumors: from genetics to therapy. Nature reviews. Gastroenterology & hepatology. 2012 Feb 7:9(4):199-208. doi: 10.1038/nrgastro.2012.9. Epub 2012 Feb 7 [PubMed PMID: 22310917]
Ganda OP, Weir GC, Soeldner JS, Legg MA, Chick WL, Patel YC, Ebeid AM, Gabbay KH, Reichlin S. "Somatostatinoma": a somatostatin-containing tumor of the endocrine pancreas. The New England journal of medicine. 1977 Apr 28:296(17):963-7 [PubMed PMID: 321960]
Level 3 (low-level) evidenceMansour JC, Chen H. Pancreatic endocrine tumors. The Journal of surgical research. 2004 Jul:120(1):139-61 [PubMed PMID: 15172200]
Mozell E, Stenzel P, Woltering EA, Rösch J, O'Dorisio TM. Functional endocrine tumors of the pancreas: clinical presentation, diagnosis, and treatment. Current problems in surgery. 1990 Jun:27(6):301-86 [PubMed PMID: 1973365]
Nesi G, Marcucci T, Rubio CA, Brandi ML, Tonelli F. Somatostatinoma: clinico-pathological features of three cases and literature reviewed. Journal of gastroenterology and hepatology. 2008 Apr:23(4):521-6 [PubMed PMID: 17645474]
Level 3 (low-level) evidenceVinik AI, Strodel WE, Eckhauser FE, Moattari AR, Lloyd R. Somatostatinomas, PPomas, neurotensinomas. Seminars in oncology. 1987 Sep:14(3):263-81 [PubMed PMID: 2820062]
O'Brien TD, Chejfec G, Prinz RA. Clinical features of duodenal somatostatinomas. Surgery. 1993 Dec:114(6):1144-7 [PubMed PMID: 8256221]
Soga J, Yakuwa Y. Somatostatinoma/inhibitory syndrome: a statistical evaluation of 173 reported cases as compared to other pancreatic endocrinomas. Journal of experimental & clinical cancer research : CR. 1999 Mar:18(1):13-22 [PubMed PMID: 10374671]
Level 2 (mid-level) evidenceBarnett P. Somatostatin and somatostatin receptor physiology. Endocrine. 2003 Apr:20(3):255-64 [PubMed PMID: 12721505]
Level 3 (low-level) evidenceTanaka S, Yamasaki S, Matsushita H, Ozawa Y, Kurosaki A, Takeuchi K, Hoshihara Y, Doi T, Watanabe G, Kawaminami K. Duodenal somatostatinoma: a case report and review of 31 cases with special reference to the relationship between tumor size and metastasis. Pathology international. 2000 Feb:50(2):146-52 [PubMed PMID: 10792774]
Level 3 (low-level) evidenceMao C, Shah A, Hanson DJ, Howard JM. Von Recklinghausen's disease associated with duodenal somatostatinoma: contrast of duodenal versus pancreatic somatostatinomas. Journal of surgical oncology. 1995 May:59(1):67-73 [PubMed PMID: 7745981]
Level 3 (low-level) evidenceÖberg K, Knigge U, Kwekkeboom D, Perren A, ESMO Guidelines Working Group. Neuroendocrine gastro-entero-pancreatic tumors: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of oncology : official journal of the European Society for Medical Oncology. 2012 Oct:23 Suppl 7():vii124-30 [PubMed PMID: 22997445]
Level 1 (high-level) evidenceKlöppel G. Classification and pathology of gastroenteropancreatic neuroendocrine neoplasms. Endocrine-related cancer. 2011 Oct:18 Suppl 1():S1-16. doi: 10.1530/ERC-11-0013. Epub 2011 Oct 17 [PubMed PMID: 22005112]
Ito T, Igarashi H, Jensen RT. Pancreatic neuroendocrine tumors: clinical features, diagnosis and medical treatment: advances. Best practice & research. Clinical gastroenterology. 2012 Dec:26(6):737-53. doi: 10.1016/j.bpg.2012.12.003. Epub [PubMed PMID: 23582916]
Level 3 (low-level) evidenceRamage JK, Ahmed A, Ardill J, Bax N, Breen DJ, Caplin ME, Corrie P, Davar J, Davies AH, Lewington V, Meyer T, Newell-Price J, Poston G, Reed N, Rockall A, Steward W, Thakker RV, Toubanakis C, Valle J, Verbeke C, Grossman AB, UK and Ireland Neuroendocrine Tumour Society. Guidelines for the management of gastroenteropancreatic neuroendocrine (including carcinoid) tumours (NETs). Gut. 2012 Jan:61(1):6-32. doi: 10.1136/gutjnl-2011-300831. Epub 2011 Nov 3 [PubMed PMID: 22052063]
O'Grady HL, Conlon KC. Pancreatic neuroendocrine tumours. European journal of surgical oncology : the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology. 2008 Mar:34(3):324-32 [PubMed PMID: 17967523]
Tjon A Tham RT, Jansen JB, Falke TH, Lamers CB. Imaging features of somatostatinoma: MR, CT, US, and angiography. Journal of computer assisted tomography. 1994 May-Jun:18(3):427-31 [PubMed PMID: 8188911]
Level 3 (low-level) evidenceSingh S, Moody L, Chan DL, Metz DC, Strosberg J, Asmis T, Bailey DL, Bergsland E, Brendtro K, Carroll R, Cleary S, Kim M, Kong G, Law C, Lawrence B, McEwan A, McGregor C, Michael M, Pasieka J, Pavlakis N, Pommier R, Soulen M, Wyld D, Segelov E, Commonwealth Neuroendocrine Tumour Collaboration (CommNETS) Follow-up Working Group. Follow-up Recommendations for Completely Resected Gastroenteropancreatic Neuroendocrine Tumors. JAMA oncology. 2018 Nov 1:4(11):1597-1604. doi: 10.1001/jamaoncol.2018.2428. Epub [PubMed PMID: 30054622]
Kunz PL, Reidy-Lagunes D, Anthony LB, Bertino EM, Brendtro K, Chan JA, Chen H, Jensen RT, Kim MK, Klimstra DS, Kulke MH, Liu EH, Metz DC, Phan AT, Sippel RS, Strosberg JR, Yao JC, North American Neuroendocrine Tumor Society. Consensus guidelines for the management and treatment of neuroendocrine tumors. Pancreas. 2013 May:42(4):557-77. doi: 10.1097/MPA.0b013e31828e34a4. Epub [PubMed PMID: 23591432]
Level 3 (low-level) evidenceZakaria A, Hammad N, Vakhariya C, Raphael M. Somatostatinoma Presented as Double-Duct Sign. Case reports in gastrointestinal medicine. 2019:2019():9506405. doi: 10.1155/2019/9506405. Epub 2019 May 9 [PubMed PMID: 31210994]
Level 3 (low-level) evidenceGlazer ES, Tseng JF, Al-Refaie W, Solorzano CC, Liu P, Willborn KA, Abdalla EK, Vauthey JN, Curley SA. Long-term survival after surgical management of neuroendocrine hepatic metastases. HPB : the official journal of the International Hepato Pancreato Biliary Association. 2010 Aug:12(6):427-33. doi: 10.1111/j.1477-2574.2010.00198.x. Epub [PubMed PMID: 20662794]
Berber E, Flesher N, Siperstein AE. Laparoscopic radiofrequency ablation of neuroendocrine liver metastases. World journal of surgery. 2002 Aug:26(8):985-90 [PubMed PMID: 12016479]
Anene C, Thompson JS, Saigh J, Badakhsh S, Ecklund RE. Somatostatinoma: atypical presentation of a rare pancreatic tumor. The American journal of gastroenterology. 1995 May:90(5):819-21 [PubMed PMID: 7733095]
Level 3 (low-level) evidenceDoherty GM. Rare endocrine tumours of the GI tract. Best practice & research. Clinical gastroenterology. 2005 Oct:19(5):807-17 [PubMed PMID: 16253902]
Raymond E, Dahan L, Raoul JL, Bang YJ, Borbath I, Lombard-Bohas C, Valle J, Metrakos P, Smith D, Vinik A, Chen JS, Hörsch D, Hammel P, Wiedenmann B, Van Cutsem E, Patyna S, Lu DR, Blanckmeister C, Chao R, Ruszniewski P. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. The New England journal of medicine. 2011 Feb 10:364(6):501-13. doi: 10.1056/NEJMoa1003825. Epub [PubMed PMID: 21306237]
Level 1 (high-level) evidenceYao JC, Shah MH, Ito T, Bohas CL, Wolin EM, Van Cutsem E, Hobday TJ, Okusaka T, Capdevila J, de Vries EG, Tomassetti P, Pavel ME, Hoosen S, Haas T, Lincy J, Lebwohl D, Öberg K, RAD001 in Advanced Neuroendocrine Tumors, Third Trial (RADIANT-3) Study Group. Everolimus for advanced pancreatic neuroendocrine tumors. The New England journal of medicine. 2011 Feb 10:364(6):514-23. doi: 10.1056/NEJMoa1009290. Epub [PubMed PMID: 21306238]
Level 1 (high-level) evidenceMoertel CG, Lefkopoulo M, Lipsitz S, Hahn RG, Klaassen D. Streptozocin-doxorubicin, streptozocin-fluorouracil or chlorozotocin in the treatment of advanced islet-cell carcinoma. The New England journal of medicine. 1992 Feb 20:326(8):519-23 [PubMed PMID: 1310159]
Strosberg JR, Fine RL, Choi J, Nasir A, Coppola D, Chen DT, Helm J, Kvols L. First-line chemotherapy with capecitabine and temozolomide in patients with metastatic pancreatic endocrine carcinomas. Cancer. 2011 Jan 15:117(2):268-75. doi: 10.1002/cncr.25425. Epub 2010 Sep 7 [PubMed PMID: 20824724]
Level 2 (mid-level) evidenceRamanathan RK, Cnaan A, Hahn RG, Carbone PP, Haller DG. Phase II trial of dacarbazine (DTIC) in advanced pancreatic islet cell carcinoma. Study of the Eastern Cooperative Oncology Group-E6282. Annals of oncology : official journal of the European Society for Medical Oncology. 2001 Aug:12(8):1139-43 [PubMed PMID: 11583197]
Kulke MH, Wu B, Ryan DP, Enzinger PC, Zhu AX, Clark JW, Earle CC, Michelini A, Fuchs CS. A phase II trial of irinotecan and cisplatin in patients with metastatic neuroendocrine tumors. Digestive diseases and sciences. 2006 Jun:51(6):1033-8 [PubMed PMID: 16865563]
Sadowski SM, Neychev V, Millo C, Shih J, Nilubol N, Herscovitch P, Pacak K, Marx SJ, Kebebew E. Prospective Study of 68Ga-DOTATATE Positron Emission Tomography/Computed Tomography for Detecting Gastro-Entero-Pancreatic Neuroendocrine Tumors and Unknown Primary Sites. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2016 Feb 20:34(6):588-96. doi: 10.1200/JCO.2015.64.0987. Epub 2015 Dec 28 [PubMed PMID: 26712231]
Luo G, Javed A, Strosberg JR, Jin K, Zhang Y, Liu C, Xu J, Soares K, Weiss MJ, Zheng L, Wolfgang CL, Cives M, Wong J, Wang W, Sun J, Shao C, Wang W, Tan H, Li J, Ni Q, Shen L, Chen M, He J, Chen J, Yu X. Modified Staging Classification for Pancreatic Neuroendocrine Tumors on the Basis of the American Joint Committee on Cancer and European Neuroendocrine Tumor Society Systems. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2017 Jan 20:35(3):274-280. doi: 10.1200/JCO.2016.67.8193. Epub 2016 Sep 30 [PubMed PMID: 27646952]
Rindi G, Klöppel G, Alhman H, Caplin M, Couvelard A, de Herder WW, Erikssson B, Falchetti A, Falconi M, Komminoth P, Körner M, Lopes JM, McNicol AM, Nilsson O, Perren A, Scarpa A, Scoazec JY, Wiedenmann B, all other Frascati Consensus Conference participants, European Neuroendocrine Tumor Society (ENETS). TNM staging of foregut (neuro)endocrine tumors: a consensus proposal including a grading system. Virchows Archiv : an international journal of pathology. 2006 Oct:449(4):395-401 [PubMed PMID: 16967267]
Level 3 (low-level) evidenceMadeira I, Terris B, Voss M, Denys A, Sauvanet A, Flejou JF, Vilgrain V, Belghiti J, Bernades P, Ruszniewski P. Prognostic factors in patients with endocrine tumours of the duodenopancreatic area. Gut. 1998 Sep:43(3):422-7 [PubMed PMID: 9863490]
Level 2 (mid-level) evidenceHouse MG, Yeo CJ, Schulick RD. Periampullary pancreatic somatostatinoma. Annals of surgical oncology. 2002 Nov:9(9):869-74 [PubMed PMID: 12417508]
Level 3 (low-level) evidenceLi X, Gou S, Liu Z, Ye Z, Wang C. Assessment of the American Joint Commission on Cancer 8th Edition Staging System for Patients with Pancreatic Neuroendocrine Tumors: A Surveillance, Epidemiology, and End Results analysis. Cancer medicine. 2018 Mar:7(3):626-634. doi: 10.1002/cam4.1336. Epub 2018 Jan 29 [PubMed PMID: 29380547]
Skoura E, Michopoulou S, Mohmaduvesh M, Panagiotidis E, Al Harbi M, Toumpanakis C, Almukhailed O, Kayani I, Syed R, Navalkissoor S, Ell PJ, Caplin ME, Bomanji J. The Impact of 68Ga-DOTATATE PET/CT Imaging on Management of Patients with Neuroendocrine Tumors: Experience from a National Referral Center in the United Kingdom. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2016 Jan:57(1):34-40. doi: 10.2967/jnumed.115.166017. Epub 2015 Oct 15 [PubMed PMID: 26471695]
Partelli S, Bartsch DK, Capdevila J, Chen J, Knigge U, Niederle B, Nieveen van Dijkum EJM, Pape UF, Pascher A, Ramage J, Reed N, Ruszniewski P, Scoazec JY, Toumpanakis C, Kianmanesh R, Falconi M, Antibes Consensus Conference participants. ENETS Consensus Guidelines for Standard of Care in Neuroendocrine Tumours: Surgery for Small Intestinal and Pancreatic Neuroendocrine Tumours. Neuroendocrinology. 2017:105(3):255-265. doi: 10.1159/000464292. Epub 2017 Feb 25 [PubMed PMID: 28237989]
Level 3 (low-level) evidenceChamberlain RS, Canes D, Brown KT, Saltz L, Jarnagin W, Fong Y, Blumgart LH. Hepatic neuroendocrine metastases: does intervention alter outcomes? Journal of the American College of Surgeons. 2000 Apr:190(4):432-45 [PubMed PMID: 10757381]