Definition/Introduction
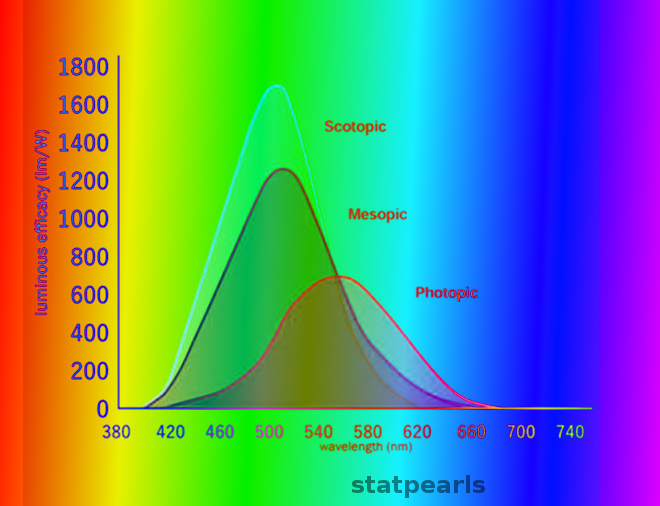
All visual perception begins with the conversion of light stimuli into neuronal signals by rod and cone photoreceptor neurons in the retina. Rods are capable of generating signals at very low (scotopic) light levels, while cones are responsible for vision at bright, or photopic, light levels. Both rod and cone photoreceptors maintain membrane depolarization and release excitatory vesicles of glutamate continuously in darkness; light encoding occurs by membrane hyperpolarization and a decrease in the rate of vesicle release. Rods are exquisitely sensitive in darkness, but their release rate decreases to the point of saturation and are unable to signal further increases in light levels even at fairly dim intensities. By contrast, cones are incapable of transmitting weak light stimuli in darkness but adapt over a very broad range of light intensities and can perceive elements of a visual scene even at very high intensities.[1][2] Because photopic vision describes vision in bright light to which rods are unable to contribute, it is synonymous with cone-mediated vision.
Issues of Concern
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Issues of Concern
Cone Light Adaptation
Light adaptation principally involves Ca, which acts as a second messenger to effect negative feedback upon light-driven membrane hyperpolarization. This negative feedback helps photoreceptors to avoid response saturation. The difference between the light levels at which rods and cones are capable of signaling arises from the fact that cones forego high sensitivity for much more effective light adaptation and therefore a broader signaling range [3].
Cone Pathways
Within the retina, cone signals transmit through bipolar cells (BCs) to retinal ganglion cells (RGCs), the neurons that transmit retinal signals to the brain via the optic nerve. Influences from horizontal and amacrine cells shape Cone-to-BC and BC-to-RGC signaling within the retina.[4][5] As a rule, each cone transmits information to over a dozen functional subtypes of cone BCs, and therefore cone signals are relayed to RGCs via many functionally distinct parallel pathways.[6][7][8] Anatomical and physiological interactions among retinal neurons allow cone BCs and RGCs to encode specific features of visual scenes; for example, RGCs may be excited specifically by light increments, decrements, or movement in a particular direction.[9] Signaling within the retina that produces such functional RGC diversity is thought to enhance visual perception. Photopic visual signals travel via RGCs to many targets in the brain and contribute to both image forming and non-image forming vision, the latter of which includes light-mediated regulation of pupillary aperture, circadian cycles, and head-eye movement coordination.[10][11] The majority of RGCs project to the lateral geniculate nucleus (LGN), where they synapse with neurons that project to the visual cortex. This pathway mediates visual perception, and this article will discuss a few of these key features below.
Clinical Significance
High Acuity Vision
Cones in many species are capable of mediating high-acuity vision. In primates and many hunting species, cones are densely packed in a small area of the retina known as the fovea. At the center of the fovea (the foveola), cones have a “direct line” to the LGN via a 1:1:1 cone to BC to the RGC relationship. By contrast, outside the fovea, photoreceptor synapse onto multiple BCs which in turn synapse onto multiple RGCs, producing a convergence of inputs that reduces visual acuity. Because the fovea lacks rods, high-acuity foveal vision is less sensitive at low light than vision originating outside the fovea, where rods are present.
Color Vision
One of the most important features of photopic vision is color perception. In humans, a cone may contain one of three photopigment proteins: S, M, or L opsin. Each type of opsin absorbs light most efficiently at a specific wavelength (S = short, 415 nm; M = medium, 530 nm; and L = long, 560 nm). As a consequence, colored stimuli are absorbed and propagated slightly differently by the three cone classes. Cone signals are then integrated and compared by retinal neurons to deliver color information to the brain.[12] Because rods contain only a single type of opsin, color vision does not occur at low light levels. In contrast to photopic vision, scotopic vision is, therefore, inherently achromatic.
Movement
The intracellular pathways that mediate phototransduction are activated much more transiently in cones than rods. The brief duration of signal transduction provides the physiological basis for the reduced sensitivity and higher temporal fidelity of cones compared to rods.[3] The spatial summation of rod signals through anatomical convergence (discussed above) also degrades the ability of the rod system to signal motion. Indeed, perceptions of moving stimuli mediated by rod pathways are approximately 25% slower than equivalent stimuli signaled via cone pathways.[13][14] The natural transience of phototransduction in cones, therefore, allows for enhanced temporal resolution at photopic light levels.
Disorders of Photopic Vision
Disorders of photopic vision typically result from mutations in one of many proteins critical for cone or retinal pigment epithelium function.[15] The most common cone disorder is color blindness, a relatively benign disorder in which a single opsin gene mutation (usually M- or L-opsin, both present on the X chromosome).[15] More severe inherited cone disorders include achromatopsia, in which most or all cones are nonfunctional, cone-only dystrophy, mixed rod-cone dystrophies, and cone dysfunctions with a syndromic cause. Signs of cone dysfunction include color vision abnormalities, reduced visual acuity, photophobia, and nystagmus.[16] The therapeutic approach for patients with these disorders is an interprofessional and depends heavily on the degree of disability. Individuals with simple color blindness may need no assistance, while patients with retinal degeneration may require extensive medical and occupational therapy.[17] Developing visual restoration approaches include gene therapy, regenerative medicine, and visual prosthetics (varying evidence levels from I to V).[18][19]
Other Clinical Issues
Trichromatic color vision arises at the level of individual cone photoreceptors (distributed coding). However, these receptors are also interconnected through specific stimulatory and inhibitory pathways, as per the opponent process theory, forming three opposing color pairs- blue/yellow, red/green and black/white. This theory also accounts for the formation of complementary afterimages.
There can be a defect in assessing the correct brightness of specific colors, as seen in conditions like protanopia, deuteranopia, and tritanopia, due to the dysfunction of a single cone subtype.
The inability to distinguish any color without a total loss of cone function is termed monochromacy, and is due to the dysfunction of two cone subtypes.
A total loss of cone function is known as achromatopsia, which manifests as a combined defect in vision in normal light intensity as well as the absence of color discrimination.
Cone defects can be assessed clinically by the use of electroretinography (ERG). Clinicians can use Ishihara plates to perform color vision assessments.
Media
References
Arshavsky VY, Burns ME. Photoreceptor signaling: supporting vision across a wide range of light intensities. The Journal of biological chemistry. 2012 Jan 13:287(3):1620-6. doi: 10.1074/jbc.R111.305243. Epub 2011 Nov 10 [PubMed PMID: 22074925]
Level 3 (low-level) evidencePugh EN Jr, Nikonov S, Lamb TD. Molecular mechanisms of vertebrate photoreceptor light adaptation. Current opinion in neurobiology. 1999 Aug:9(4):410-8 [PubMed PMID: 10448166]
Level 3 (low-level) evidenceIngram NT, Sampath AP, Fain GL. Why are rods more sensitive than cones? The Journal of physiology. 2016 Oct 1:594(19):5415-26. doi: 10.1113/JP272556. Epub 2016 Jul 21 [PubMed PMID: 27218707]
Thoreson WB, Mangel SC. Lateral interactions in the outer retina. Progress in retinal and eye research. 2012 Sep:31(5):407-41. doi: 10.1016/j.preteyeres.2012.04.003. Epub 2012 May 3 [PubMed PMID: 22580106]
Level 3 (low-level) evidenceDemb JB, Singer JH. Functional Circuitry of the Retina. Annual review of vision science. 2015 Nov 24:1():263-289. doi: 10.1146/annurev-vision-082114-035334. Epub [PubMed PMID: 28532365]
Euler T, Haverkamp S, Schubert T, Baden T. Retinal bipolar cells: elementary building blocks of vision. Nature reviews. Neuroscience. 2014 Aug:15(8):507-19 [PubMed PMID: 25158357]
Level 3 (low-level) evidenceShekhar K, Lapan SW, Whitney IE, Tran NM, Macosko EZ, Kowalczyk M, Adiconis X, Levin JZ, Nemesh J, Goldman M, McCarroll SA, Cepko CL, Regev A, Sanes JR. Comprehensive Classification of Retinal Bipolar Neurons by Single-Cell Transcriptomics. Cell. 2016 Aug 25:166(5):1308-1323.e30. doi: 10.1016/j.cell.2016.07.054. Epub [PubMed PMID: 27565351]
Wässle H, Puller C, Müller F, Haverkamp S. Cone contacts, mosaics, and territories of bipolar cells in the mouse retina. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2009 Jan 7:29(1):106-17. doi: 10.1523/JNEUROSCI.4442-08.2009. Epub [PubMed PMID: 19129389]
Level 3 (low-level) evidenceMasland RH. The neuronal organization of the retina. Neuron. 2012 Oct 18:76(2):266-80. doi: 10.1016/j.neuron.2012.10.002. Epub 2012 Oct 17 [PubMed PMID: 23083731]
Level 3 (low-level) evidenceVan Essen DC. Behind the optic nerve: an inside view of the primate visual system. Transactions of the American Ophthalmological Society. 1995:93():123-33 [PubMed PMID: 8719674]
Level 3 (low-level) evidenceGiolli RA, Blanks RH, Lui F. The accessory optic system: basic organization with an update on connectivity, neurochemistry, and function. Progress in brain research. 2006:151():407-40 [PubMed PMID: 16221596]
Level 3 (low-level) evidenceKolb H, Fernandez E, Nelson R, Gouras P. Color Vision. Webvision: The Organization of the Retina and Visual System. 1995:(): [PubMed PMID: 21413395]
Gegenfurtner KR, Mayser H, Sharpe LT. Seeing movement in the dark. Nature. 1999 Apr 8:398(6727):475-6 [PubMed PMID: 10206642]
Level 3 (low-level) evidenceGegenfurtner KR, Mayser HM, Sharpe LT. Motion perception at scotopic light levels. Journal of the Optical Society of America. A, Optics, image science, and vision. 2000 Sep:17(9):1505-15 [PubMed PMID: 10975360]
Roosing S, Thiadens AA, Hoyng CB, Klaver CC, den Hollander AI, Cremers FP. Causes and consequences of inherited cone disorders. Progress in retinal and eye research. 2014 Sep:42():1-26. doi: 10.1016/j.preteyeres.2014.05.001. Epub 2014 May 22 [PubMed PMID: 24857951]
Level 3 (low-level) evidenceMoore AT. Cone and cone-rod dystrophies. Journal of medical genetics. 1992 May:29(5):289-90 [PubMed PMID: 1583653]
Markowitz SN. Principles of modern low vision rehabilitation. Canadian journal of ophthalmology. Journal canadien d'ophtalmologie. 2006 Jun:41(3):289-312 [PubMed PMID: 16767184]
Moore NA, Morral N, Ciulla TA, Bracha P. Gene therapy for inherited retinal and optic nerve degenerations. Expert opinion on biological therapy. 2018 Jan:18(1):37-49. doi: 10.1080/14712598.2018.1389886. Epub 2017 Oct 23 [PubMed PMID: 29057663]
Level 3 (low-level) evidenceDuncan JL,Pierce EA,Laster AM,Daiger SP,Birch DG,Ash JD,Iannaccone A,Flannery JG,Sahel JA,Zack DJ,Zarbin MA, Inherited Retinal Degenerations: Current Landscape and Knowledge Gaps. Translational vision science [PubMed PMID: 30034950]