Introduction
Pilocytic astrocytoma, previously referred to as cystic cerebellar astrocytoma or juvenile pilocytic astrocytoma, was first described in 1931 by Harvey Cushing based on a case series of cerebellar astrocytomas.[1] Pilocytic astrocytomas are low-grade, usually benign, slow-growing, well-circumscribed brain tumors that tend to occur in the pediatric population and also young adults. This condition arises from astrocytes—the star-shaped cells that support nerve cells in the brain. According to the World Health Organization (WHO) classification of central nervous system (CNS) tumors, pilocytic astrocytomas are grade I gliomas with a generally good prognosis.[2]
While pilocytic astrocytomas most often occur in the cerebellum, they can also be found along the optic pathways, hypothalamus, and brainstem. These tumors may also occur in the cerebral hemispheres, though this is more common in young adults. The presentation and treatments for pilocytic astrocytomas vary based on their location; however, this article will focus on cerebellar pilocytic astrocytomas. According to the new 2021 WHO classification of CNS tumors, pilocytic astrocytomas are grouped with other circumscribed astrocytic gliomas, such as pleomorphic xanthoastrocytoma, subependymal giant cell astrocytoma, and choroid gliomas.[2]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
A strong association exists between neurofibromatosis type 1 (NF1) and pilocytic astrocytomas; up to 20% of patients with NF1 develop a pilocytic astrocytoma, most commonly along the optic pathway.[3][4] However, most pilocytic astrocytomas are believed to be caused by sporadic rather than inherited mutations.
BRAF gene alterations and mitogen-activated protein kinase (MAPK) signaling pathway alterations have been found in the majority of pilocytic astrocytomas.[5][6][7] Roughly 60% of pilocytic astrocytomas found within the cerebellum harbor the KIAA1549-BRAF gene fusion.[8][9] BRAF is an intracellular serine/threonine kinase involved in activating the MAPK pathway.[10] This is a proto-oncogene, mutations of which have been found to cause human cancers.[11][12] Other mutations frequently seen in pilocytic astrocytomas include BRAFV600E point mutations found in 5% to 10% of all cases, although more prevalent in supratentorial tumors.[2]
Epidemiology
Brain tumors are the most prevalent form of solid cancer in childhood.[13] Pilocytic astrocytoma is the most common childhood brain tumor, with an incidence of 0.8 per 100,000 individuals.[14][15] Pilocytic astrocytoma most often presents in the second decade of life, with 75% occurring before the age of 20, and accounts for 15% of all brain tumors in children.[16] This condition comprises 27% to 40% of all pediatric posterior fossa tumors.[17][18]
Pilocytic astrocytoma can also occur in adults, although typically in young adults.[19] This condition comprises 5% of all primary brain tumors in adults and is often located in the cerebellum. However, a case series found them most commonly in the temporal and parietal lobes.[20]
Pathophysiology
Pilocytic astrocytoma can be located anywhere within the neuroaxis and tends to occur close to the midline. In adults, the tumor can be more lateral within the cerebellum.[18] More common locations of pilocytic astrocytoma include:
Histopathology
Pilocytic astrocytoma is a WHO grade I tumor, with a small case series of anaplastic pilocytic astrocytoma displaying uncertain behavior.[22] The tumor's distinct histological features, including its characteristic cell morphology and patterns, provide key insights into its behavior and classification.
Microscopic Features
Pilocytic astrocytoma gets its name from the microscopic appearance of cells with long, thin bipolar processes that resemble hairs, hence the term "pilocytic."[5] Rosenthal fibers, which are elongated eosinophilic bundles, are often found on hematoxylin and eosin (H&E) staining (see Image. Microscopic Features of Pilocytic Astrocytoma). Another pathological characteristic is the presence of eosinophilic granular bodies. Pilocytic astrocytomas typically exhibit low-to-moderate cellularity, and multinucleated giant cells with peripheral nuclei can be observed. Tumors present for an extended period may have hemosiderin-laden macrophages and calcifications.[23] Areas of necrosis may also be seen, albeit rarely. Although pilocytic astrocytomas are radiologically described as well-circumscribed, nearly two-thirds infiltrate the surrounding brain parenchyma.[24][25]
Microscopically, differentiating between pilocytic astrocytoma and low-grade diffuse astrocytoma can be challenging.[26] For this reason, the pathology team should have access to demographic and radiographic information to help guide the diagnosis. Small biopsy samples further compound this diagnostic issue. Pilocytic astrocytomas stain positive for glial fibrillary acidic protein (GFAP), S100, and OLIG2 while being negative for neurofilament, chromogranin, and CD34.[27]
Molecular Features
The most common genetic abnormalities found in 70% to 75% of patients with pilocytic astrocytomas are BRAF alterations.[6] BRAF alterations are more common in pediatric pilocytic astrocytoma than in adult cases.[28] Pilocytic astrocytomas exhibit alterations in the MAPK signaling pathway in more than 80% of cases.[5][7][29] The KIAA1549-BRAF fusion is the most commonly identified mutation in individuals with pilocytic astrocytoma.[10][28] Importantly, as with other pediatric low-grade gliomas, these tumors are negative for IDH (isocitrate dehydrogenase) and TP53 mutations.[26][17]
History and Physical
In patients with pilocytic astrocytoma, findings from the history and physical examinations often reflect the tumor's location and growth pattern. Symptoms typically develop gradually. Patients with pilocytic astrocytomas can present symptoms secondary to a posterior fossa mass effect. This may include obstructive hydrocephalus with resultant headache, nausea, vomiting, and papilledema. Seizures are rare in cases of posterior fossa lesions.[30] If hydrocephalus occurs before the fusion of the cranial sutures, typically before 18 months of age, an increase in head circumference and splaying of cranial sutures will likely occur.[30]
Posterior fossa lesions can also cause cranial nerve palsies. Diplopia may occur due to abducens nerve palsy resulting from nerve stretching. Patients may also have blurred vision due to papilledema. Lesions of the cerebellar hemisphere result in peripheral ataxia, dysmetria, intention tremor, nystagmus, and dysarthria.
In contrast, lesions of the vermis can cause a broad-based gait, truncal ataxia, and titubation. Lesions involving the hypothalamus may result in hormonal imbalances or growth issues. Physical examination often reveals signs of increased intracranial pressure and cerebellar dysfunction. Focal neurological deficits depend on the tumor's specific location and its impact on surrounding structures.
Evaluation
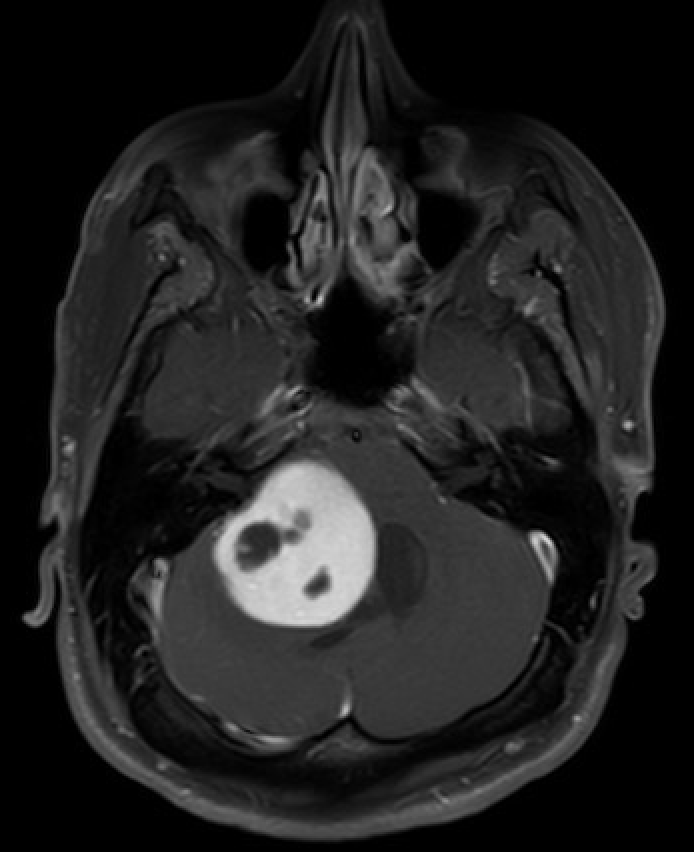
The imaging modality of choice is brain magnetic resonance imaging (MRI) with and without contrast (see Image. MRI of the Brain Showing Pilocytic Astrocytoma). Radiation exposure is minimized, which is crucial for patients with NF1. However, if the patient is neurologically unstable, an urgent head computed tomography (CT) scan may be necessary.
In clinical practice, children with posterior fossa tumors often undergo whole neuroaxis imaging, as more malignant tumors can form drop metastases. Pilocytic astrocytomas typically do not form drop metastases, such as ependymomas and medulloblastomas, and thus may not require whole neuroaxis imaging. However, the variant pilomyxoid astrocytoma is more aggressive and can form drop metastases. While spinal seeding or leptomeningeal dissemination is extremely rare in pilocytic astrocytomas, whole neuroaxis imaging is recommended only in suspected or uncertain cases.[31][32][33][34]
Pilocytic astrocytomas can exhibit various radiological appearances. In 66% of cases, pilocytic astrocytoma presents with a significant cystic component and an avidly enhancing mural nodule; in 46% of cases, the cyst wall also shows enhancement.[35] Up to 17% of tumors are solid with minimal or no cystic component, and up to 20% may show calcification.[24] In addition, pilocytic astrocytomas are periventricular in 82% of cases.[24]
The cyst content in pilocytic astrocytomas is proteinaceous and often denser than cerebrospinal fluid (CSF).[36] On MRI, the cyst content typically shows a hyperintense signal on T2-weighted images, similar to CSF. The mural nodule is usually hyperintense on T2-weighted images and iso- or hypointense on T1-weighted images. As described above, the nodule’s avid enhancement helps differentiate it from other low-grade gliomas.
Treatment / Management
The mainstay of treatment is surgical excision to achieve gross total resection, as this can be considered curative for the disease. However, involvement of the brainstem or cranial nerves may limit complete resection. Resection of the mural nodule rather than the cyst wall is recommended.[35] However, if the cyst wall is thick, it may be considered part of the nodule and thus removed.[35](B3)
Conventional chemoradiation is not typically required upfront; clinical follow-up with serial imaging is preferred. Radiotherapy, especially in this brain region, can have significant adverse effects.[37] If recurrence occurs, additional surgical resection is generally considered if feasible. Radiotherapy may be appropriate if the tumor is unresectable or if malignant histology is present.[38]
Some studies have shown that stereotactic radiosurgery yields excellent results for residual and recurrent tumors, although there is criticism about the duration of patient follow-up. Concerns exist that stereotactic radiosurgery might promote anaplastic transformation.[39] As mentioned previously, radiation is typically avoided in patients with NF1 unless necessary, as these patients are predisposed to further tumor development.
Chemotherapy is generally not used as a first-line treatment for pilocytic astrocytoma; however, several trials have reported 5-year progression-free survival rates between 34% and 45%.[40][41] Given the propensity for these tumors to harbor mutations that affect the MAPK signaling pathway, BRAF- and MEK inhibitors have been studied, with variable response rates.[42][43]
Several approaches are available for managing hydrocephalus in neurologically stable patients. These approaches include:
- CSF diversion at the time of surgery, followed by immediate tumor resection.
- Surgical resection without CSF diversion. If hydrocephalus persists postoperatively, then CSF diversion is performed.
- CSF diversion before imaging studies if patients require anesthesia.
If the patient is neurologically unstable due to hydrocephalus or brainstem compression, urgent intervention is necessary. For patients presenting with hydrocephalus, some experts recommend initial CSF diversion before definitive surgery, using methods such as an external ventricular drain (EVD), endoscopic third ventriculostomy (ETV), or a ventricular-peritoneal shunt (VPS). In certain medical centers, an EVD or ETV may be performed concurrently with surgery, followed by immediate tumor resection. Surgical considerations with CSF diversion include the risk of upward transtentorial herniation and CSF infection from VPS or EVD contamination.
Differential Diagnosis
The differential diagnosis of pilocytic astrocytomas involves distinguishing them from other brain tumors and lesions with similar clinical and radiological features. Accurate diagnosis and appropriate treatment planning depend on identifying key differences. This process includes evaluating imaging characteristics, patient demographics, and histopathological findings to differentiate pilocytic astrocytomas from other low-grade gliomas, high-grade gliomas, and non-neoplastic conditions.
Most Common Pediatric Posterior Fossa Tumors
- Medulloblastoma: Typically located midline, at the roof of the fourth ventricle, or in the vermis of the brain, less than 10% demonstrate calcification.
- Diffuse pontine glioma: Often presents with multiple cranial nerve palsies.
- Ependymoma: Usually arises in the floor of the fourth ventricle; calcification is common.[30][44]
Additional Pediatric Posterior Fossa Lesions: Clinical and Radiological Differentials
- Hemangioblastoma.
- Atypical teratoid or rhabdoid tumor.
- Cerebellar abscess.
- Choroid plexus papilloma.
- Metastasis, which includes neuroblastoma, rhabdomyosarcoma, Wilms tumor.
Adult Posterior Fossa Tumors: Clinical and Radiological Differentials
Prognosis
Pilocytic astrocytoma is a slow-growing tumor that is often curative with gross total resection. Similar to many tumors, the extent of resection is the best prognostic factor. The 10-year survival rate is approximately 95% if the tumor is completely resected.[15][47] Recurrence is rare if a complete resection is achieved.
Tumors that do recur tend to do so within a few years.[48] Collins' law states that "the period of risk for tumor recurrence is the age of the child at diagnosis plus 9 months."[49] Using this concept, pilocytic astrocytoma can be considered cured if it does not recur within that time. However, patients should be monitored for late recurrence. For tumors with gross total resection, some authors recommend a maximum of 3 years of surveillance imaging due to the minimal risk of recurrence in pediatric pilocytic astrocytoma.[50] If there is incomplete resection, recurrence may occur with progression of symptoms. Several risk factors for recurrence have been identified, including a solid tumor, exophytic component, and tumor invasion into the brain parenchyma.[51]
Patients aged 1 or younger generally have the worst prognosis, potentially due to the variant pilomyxoid astrocytoma, which is more common in very young children.[15] In adults, pilocytic astrocytoma has a 5-year survival rate of 85%, with progression-free survival at 70% and a recurrence rate of approximately 20%.[52]
Complications
Patients with pilocytic astrocytoma may experience complications, especially if the tumor is not promptly and effectively treated. Hydrocephalus is common due to obstruction of CSF pathways, leading to increased intracranial pressure. Patients with posterior fossa tumors may develop hydrocephalus and require a VP shunt, potentially making them shunt-dependent for life.[53][54]
Neurological deficits can arise from tumor growth affecting critical brain regions, leading to motor, sensory, or cognitive impairments. Postsurgical complications may include infection, bleeding, or damage to surrounding brain tissue, which can exacerbate neurological issues. Additionally, the risk of tumor recurrence necessitates ongoing monitoring and potential further treatment.
In some cases, radiation or chemotherapy used in treatment can lead to long-term adverse effects, impacting the patient’s quality of life. While recurrence is usually treatable with further resection, some pilocytic astrocytomas can undergo malignant degeneration, although this is rare. Most cases of malignant degeneration seem to follow radiotherapy.[55][56]
Consultations
Patients with pilocytic astrocytoma require consultations with a range of specialists to ensure comprehensive care. Neurosurgical consultation is essential for evaluating the need for surgical resection and planning the procedure. Neuro-oncologists and radiation oncologists may be consulted for adjuvant therapies if complete surgical removal is not feasible. Endocrinologists are often involved if the tumor affects the hypothalamus or other hormone-regulating structures. Ophthalmologists are critical to monitor and manage vision-related issues caused by tumors affecting the optic pathways.
Additionally, consultations with physical therapists, occupational therapists, and rehabilitation specialists are crucial for addressing functional impairments and facilitating recovery. Psychologists or social workers should also be involved to provide emotional support and address the psychosocial aspects of the patient's condition. This multidisciplinary approach ensures that all aspects of the patient's health are addressed, optimizing outcomes and quality of life.
Deterrence and Patient Education
Modifiable risk factors for pilocytic astrocytoma have not been identified. Genetic conditions, such as NF1, predispose families to pilocytic astrocytoma, thereby making genetic testing and counseling potentially appropriate. However, most pilocytic astrocytomas are due to sporadic mutations.[3][4]
Educating patients and their families about the importance of early detection and prompt treatment is crucial for improving outcomes. Regular follow-up appointments are essential for monitoring recurrence and managing the long-term effects of the tumor or its treatment. Patients should be informed about symptoms of increased intracranial pressure and other neurological changes to prompt timely medical consultation when they arise. Providing information on healthy lifestyle choices, supportive therapies, and available resources can further assist patients and families in navigating the challenges associated with pilocytic astrocytoma, thereby enhancing their overall quality of life.
Pearls and Other Issues
Clinicians can benefit from practical insights that enhance patient care when managing pilocytic astrocytoma. These clinical pearls provide valuable guidance on diagnostic approaches, treatment strategies, and patient care. Key clinical pearls to consider include:
- Pilocytic astrocytoma is the most common pediatric brain tumor and is classified as WHO grade 1 glioma.
- Prompt recognition of symptoms such as headaches, ataxia, and visual disturbances can lead to early diagnosis and better outcomes.
- Presentation typically includes cerebellar signs and features of hydrocephalus.
- MRI is the preferred imaging modality for diagnosing pilocytic astrocytoma, providing detailed information on tumor location and characteristics.
- MRI findings are typically cystic with a mural nodule that enhances avidly, although the tumor can also be completely solid.
- Microscopy shows long, bipolar cellular processes that appear "hair-like."
- Gross total resection typically results in an excellent prognosis; radiotherapy and chemotherapy are not routinely used.
- The survival rate is 95% at 10 years with surgery alone if complete resection is achieved.
- Regular monitoring through follow-up imaging is crucial for early detection of potential tumor recurrence.
- A strong association with NF1 exists; 5% to 20% of patients with NF1 develop pilocytic astrocytoma, mostly along the optic pathways.
- Patients may require CSF diversion.
Enhancing Healthcare Team Outcomes
Pilocytic astrocytoma is the most common pediatric brain tumor and necessitates urgent assessment and intervention, while also being managed as a chronic condition. These patients will initially present to pediatricians, emergency room clinicians, or general practitioners. Clinicians must recognize the signs and symptoms of masses affecting the cerebellum or brainstem, including the clinical presentation of hydrocephalus.
Enhancing patient-centered care, outcomes, patient safety, and team performance in the treatment of patients with pilocytic astrocytoma requires a collaborative, interprofessional approach involving various healthcare professionals. Physicians, advanced practitioners, nurses, pharmacists, and other healthcare professionals must possess specialized skills in their respective fields. Physicians and advanced practitioners should be adept at diagnosing and formulating treatment plans, utilizing advanced imaging techniques, and performing surgical interventions when necessary. Nurses require skills for monitoring for complications and providing patient care and emotional support. Pharmacists must be knowledgeable about chemotherapy regimens and potential drug interactions.
Rehabilitation of patients with pilocytic astrocytoma is crucial to address functional impairments and facilitate recovery. Physical therapists focus on improving motor skills and balance, while occupational therapists help patients regain daily living activities and independence. Social workers and counselors address psychosocial aspects and provide support services.
A well-coordinated strategy involves comprehensive treatment planning, where each healthcare team member understands their role and the overall care plan. This includes preoperative assessment, surgical planning, postoperative care, and long-term follow-up. Utilizing clinical guidelines and evidence-based practices ensures the treatment is standardized and effective.
Clear and timely communication between healthcare providers about patient status, treatment progress, and any emerging issues is essential. Regular interdisciplinary meetings and case conferences help ensure everyone is aligned with the treatment plan and any changes. Coordinated care involves seamless transitions between different phases of treatment and care settings, including preoperative care, inpatient surgical care, and postoperative rehabilitation and follow-up. Additionally, care coordination ensures patients receive appropriate referrals to specialists, such as oncologists or endocrinologists, when necessary.
Media
(Click Image to Enlarge)
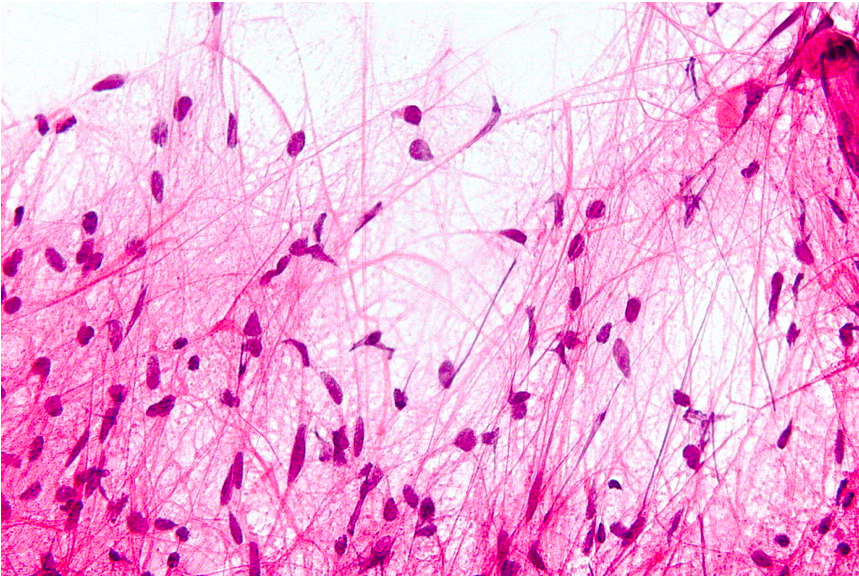
Microscopic Features of Pilocytic Astrocytoma. Hair-like cell processes can be observed with H&E staining.
Nephron, Public Domain, via Wikimedia Commons
References
Koeller KK, Rushing EJ. From the archives of the AFIP: pilocytic astrocytoma: radiologic-pathologic correlation. Radiographics : a review publication of the Radiological Society of North America, Inc. 2004 Nov-Dec:24(6):1693-708 [PubMed PMID: 15537977]
Louis DN, Perry A, Wesseling P, Brat DJ, Cree IA, Figarella-Branger D, Hawkins C, Ng HK, Pfister SM, Reifenberger G, Soffietti R, von Deimling A, Ellison DW. The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro-oncology. 2021 Aug 2:23(8):1231-1251. doi: 10.1093/neuonc/noab106. Epub [PubMed PMID: 34185076]
Listernick R, Charrow J, Greenwald M, Mets M. Natural history of optic pathway tumors in children with neurofibromatosis type 1: a longitudinal study. The Journal of pediatrics. 1994 Jul:125(1):63-6 [PubMed PMID: 8021787]
Sadighi Z, Slopis J. Pilocytic astrocytoma: a disease with evolving molecular heterogeneity. Journal of child neurology. 2013 May:28(5):625-32. doi: 10.1177/0883073813476141. Epub 2013 Feb 25 [PubMed PMID: 23439714]
Collins VP, Jones DT, Giannini C. Pilocytic astrocytoma: pathology, molecular mechanisms and markers. Acta neuropathologica. 2015 Jun:129(6):775-88. doi: 10.1007/s00401-015-1410-7. Epub 2015 Mar 20 [PubMed PMID: 25792358]
Jones DT, Kocialkowski S, Liu L, Pearson DM, Bäcklund LM, Ichimura K, Collins VP. Tandem duplication producing a novel oncogenic BRAF fusion gene defines the majority of pilocytic astrocytomas. Cancer research. 2008 Nov 1:68(21):8673-7. doi: 10.1158/0008-5472.CAN-08-2097. Epub [PubMed PMID: 18974108]
Level 3 (low-level) evidenceCollins KL, Pollack IF. Pediatric Low-Grade Gliomas. Cancers. 2020 May 4:12(5):. doi: 10.3390/cancers12051152. Epub 2020 May 4 [PubMed PMID: 32375301]
Salles D, Santino SF, Ribeiro DA, Malinverni ACM, Stávale JN. The involvement of the MAPK pathway in pilocytic astrocytomas. Pathology, research and practice. 2022 Apr:232():153821. doi: 10.1016/j.prp.2022.153821. Epub 2022 Feb 25 [PubMed PMID: 35231859]
Cler SJ, Skidmore A, Yahanda AT, Mackey K, Rubin JB, Cluster A, Perkins S, Gauvain K, King AA, Limbrick DD, McEvoy S, Park TS, Smyth MD, Mian AY, Chicoine MR, Dahiya S, Strahle JM. Genetic and histopathological associations with outcome in pediatric pilocytic astrocytoma. Journal of neurosurgery. Pediatrics. 2022 May 1:29(5):504-512. doi: 10.3171/2021.9.PEDS21405. Epub 2022 Feb 11 [PubMed PMID: 35148515]
Tjota MY, Pytel P. Educational Case: Pilocytic Astrocytoma With Atypical Features. Academic pathology. 2020 Jan-Dec:7():2374289520912498. doi: 10.1177/2374289520912498. Epub 2020 Mar 30 [PubMed PMID: 32284965]
Level 3 (low-level) evidenceSithanandam G, Kolch W, Duh FM, Rapp UR. Complete coding sequence of a human B-raf cDNA and detection of B-raf protein kinase with isozyme specific antibodies. Oncogene. 1990 Dec:5(12):1775-80 [PubMed PMID: 2284096]
Sithanandam G, Druck T, Cannizzaro LA, Leuzzi G, Huebner K, Rapp UR. B-raf and a B-raf pseudogene are located on 7q in man. Oncogene. 1992 Apr:7(4):795-9 [PubMed PMID: 1565476]
Allen JC. Childhood brain tumors: current status of clinical trials in newly diagnosed and recurrent disease. Pediatric clinics of North America. 1985 Jun:32(3):633-51 [PubMed PMID: 3889800]
Level 3 (low-level) evidenceOstrom QT, Gittleman H, Liao P, Rouse C, Chen Y, Dowling J, Wolinsky Y, Kruchko C, Barnholtz-Sloan J. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007-2011. Neuro-oncology. 2014 Oct:16 Suppl 4(Suppl 4):iv1-63. doi: 10.1093/neuonc/nou223. Epub [PubMed PMID: 25304271]
Tabash MA. Characteristics, survival and incidence rates and trends of pilocytic astrocytoma in children in the United States; SEER-based analysis. Journal of the neurological sciences. 2019 May 15:400():148-152. doi: 10.1016/j.jns.2019.03.028. Epub 2019 Mar 29 [PubMed PMID: 30953904]
Wallner KE, Gonzales MF, Edwards MS, Wara WM, Sheline GE. Treatment results of juvenile pilocytic astrocytoma. Journal of neurosurgery. 1988 Aug:69(2):171-6 [PubMed PMID: 3392563]
AlRayahi J, Zapotocky M, Ramaswamy V, Hanagandi P, Branson H, Mubarak W, Raybaud C, Laughlin S. Pediatric Brain Tumor Genetics: What Radiologists Need to Know. Radiographics : a review publication of the Radiological Society of North America, Inc. 2018 Nov-Dec:38(7):2102-2122. doi: 10.1148/rg.2018180109. Epub [PubMed PMID: 30422762]
Burkhard C, Di Patre PL, Schüler D, Schüler G, Yaşargil MG, Yonekawa Y, Lütolf UM, Kleihues P, Ohgaki H. A population-based study of the incidence and survival rates in patients with pilocytic astrocytoma. Journal of neurosurgery. 2003 Jun:98(6):1170-4 [PubMed PMID: 12816259]
Level 2 (mid-level) evidenceRINGERTZ N, NORDENSTAM H. Cerebellar astrocytoma. Journal of neuropathology and experimental neurology. 1951 Oct:10(4):343-67 [PubMed PMID: 14874138]
Boschetti G, Santos AJ, Fermon KP, de Freitas Honório GL, Batistella G, Kusma SZ, Stávale JN, de Paiva Neto MA, Fleury Malheiros SM. Adult Pilocytic Astrocytomas: A Brazilian Series. World neurosurgery. 2020 Jan:133():e115-e120. doi: 10.1016/j.wneu.2019.08.146. Epub 2019 Sep 21 [PubMed PMID: 31550545]
Pollack IF, Hoffman HJ, Humphreys RP, Becker L. The long-term outcome after surgical treatment of dorsally exophytic brain-stem gliomas. Journal of neurosurgery. 1993 Jun:78(6):859-63 [PubMed PMID: 8487066]
Fiechter M, Hewer E, Knecht U, Wiest R, Beck J, Raabe A, Oertel MF. Adult anaplastic pilocytic astrocytoma - a diagnostic challenge? A case series and literature review. Clinical neurology and neurosurgery. 2016 Aug:147():98-104. doi: 10.1016/j.clineuro.2016.06.005. Epub 2016 Jun 6 [PubMed PMID: 27341279]
Level 2 (mid-level) evidenceWippold FJ 2nd, Perry A, Lennerz J. Neuropathology for the neuroradiologist: Rosenthal fibers. AJNR. American journal of neuroradiology. 2006 May:27(5):958-61 [PubMed PMID: 16687524]
Coakley KJ, Huston J 3rd, Scheithauer BW, Forbes G, Kelly PJ. Pilocytic astrocytomas: well-demarcated magnetic resonance appearance despite frequent infiltration histologically. Mayo Clinic proceedings. 1995 Aug:70(8):747-51 [PubMed PMID: 7630212]
Level 2 (mid-level) evidenceHayostek CJ, Shaw EG, Scheithauer B, O'Fallon JR, Weiland TL, Schomberg PJ, Kelly PJ, Hu TC. Astrocytomas of the cerebellum. A comparative clinicopathologic study of pilocytic and diffuse astrocytomas. Cancer. 1993 Aug 1:72(3):856-69 [PubMed PMID: 8334640]
Level 2 (mid-level) evidenceLouis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta neuropathologica. 2016 Jun:131(6):803-20. doi: 10.1007/s00401-016-1545-1. Epub 2016 May 9 [PubMed PMID: 27157931]
Pizzimenti C, Fiorentino V, Germanò A, Martini M, Ieni A, Tuccari G. Pilocytic astrocytoma: The paradigmatic entity in low‑grade gliomas (Review). Oncology letters. 2024 Apr:27(4):146. doi: 10.3892/ol.2024.14279. Epub 2024 Feb 8 [PubMed PMID: 38385109]
Level 3 (low-level) evidenceKurani H, Gurav M, Shetty O, Chinnaswamy G, Moiyadi A, Gupta T, Jalali R, Epari S. Pilocytic astrocytomas: BRAFV600E and BRAF fusion expression patterns in pediatric and adult age groups. Child's nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery. 2019 Sep:35(9):1525-1536. doi: 10.1007/s00381-019-04282-1. Epub 2019 Jul 18 [PubMed PMID: 31321520]
Reitman ZJ, Paolella BR, Bergthold G, Pelton K, Becker S, Jones R, Sinai CE, Malkin H, Huang Y, Grimmet L, Herbert ZT, Sun Y, Weatherbee JL, Alberta JA, Daley JF, Rozenblatt-Rosen O, Condurat AL, Qian K, Khadka P, Segal RA, Haas-Kogan D, Filbin MG, Suva ML, Regev A, Stiles CD, Kieran MW, Goumnerova L, Ligon KL, Shalek AK, Bandopadhayay P, Beroukhim R. Mitogenic and progenitor gene programmes in single pilocytic astrocytoma cells. Nature communications. 2019 Aug 19:10(1):3731. doi: 10.1038/s41467-019-11493-2. Epub 2019 Aug 19 [PubMed PMID: 31427603]
Level 2 (mid-level) evidencePlum F, Posner JB. The diagnosis of stupor and coma. Contemporary neurology series. 1972:10():1-286 [PubMed PMID: 4664014]
Crabtree KL, Arnold PM. Spinal seeding of a pilocytic astrocytoma in an adult, initially diagnosed 18 years previously. Pediatric neurosurgery. 2010:46(1):66-70. doi: 10.1159/000315320. Epub 2010 Jun 1 [PubMed PMID: 20516744]
Level 3 (low-level) evidenceZorlu F, Selek U, Akyuz C, Ozturk A, Soylemezoglu F, Akalan N. Spinal seeding of a pilocytic astrocytoma following multiple subtotal resections. Pediatric neurosurgery. 2005 Sep-Oct:41(5):248-52 [PubMed PMID: 16195677]
Level 3 (low-level) evidenceSu YF, Chang CH, Chiou SS, Chai CY, Hwang SL, Loh JK. A Case of Leptomeningeal Dissemination of Pilocytic Astrocytoma in a Child. The Canadian journal of neurological sciences. Le journal canadien des sciences neurologiques. 2017 Sep:44(5):607-610. doi: 10.1017/cjn.2017.22. Epub 2017 Apr 27 [PubMed PMID: 28446265]
Level 3 (low-level) evidenceKanda M, Tanaka H, Shinoda S, Masuzawa T. Leptomeningeal dissemination of pilocytic astrocytoma via hematoma in a child. Case report. Neurosurgical focus. 2002 Jul 15:13(1):ECP2 [PubMed PMID: 15916413]
Level 3 (low-level) evidenceBeni-Adani L, Gomori M, Spektor S, Constantini S. Cyst wall enhancement in pilocytic astrocytoma: neoplastic or reactive phenomena. Pediatric neurosurgery. 2000 May:32(5):234-9 [PubMed PMID: 10965269]
Level 3 (low-level) evidenceZimmerman RA, Bilaniuk LT, Bruno L, Rosenstock J. Computed tomography of cerebellar astrocytoma. AJR. American journal of roentgenology. 1978 May:130(5):929-33 [PubMed PMID: 417594]
Austin EJ, Alvord EC Jr. Recurrences of cerebellar astrocytomas: a violation of Collins' law. Journal of neurosurgery. 1988 Jan:68(1):41-7 [PubMed PMID: 3335911]
Packer RJ, Lange B, Ater J, Nicholson HS, Allen J, Walker R, Prados M, Jakacki R, Reaman G, Needles MN. Carboplatin and vincristine for recurrent and newly diagnosed low-grade gliomas of childhood. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 1993 May:11(5):850-6 [PubMed PMID: 8487049]
Murphy ES, Parsai S, Kano H, Sheehan JP, Martinez-Alvarez R, Martinez-Moreno N, Kondziolka D, Simonova G, Liscak R, Mathieu D, Lee CC, Yang HC, Lee JY, McShane BJ, Fang F, Trifiletti DM, Sharma M, Barnett GH. Outcomes of stereotactic radiosurgery for pilocytic astrocytoma: an international multiinstitutional study. Journal of neurosurgery. 2019 Nov 29:134(1):162-170. doi: 10.3171/2019.9.JNS191335. Epub 2019 Nov 29 [PubMed PMID: 31783364]
Manoharan N, Liu KX, Mueller S, Haas-Kogan DA, Bandopadhayay P. Pediatric low-grade glioma: Targeted therapeutics and clinical trials in the molecular era. Neoplasia (New York, N.Y.). 2023 Feb:36():100857. doi: 10.1016/j.neo.2022.100857. Epub 2022 Dec 24 [PubMed PMID: 36566593]
Lassaletta A, Scheinemann K, Zelcer SM, Hukin J, Wilson BA, Jabado N, Carret AS, Lafay-Cousin L, Larouche V, Hawkins CE, Pond GR, Poskitt K, Keene D, Johnston DL, Eisenstat DD, Krishnatry R, Mistry M, Arnoldo A, Ramaswamy V, Huang A, Bartels U, Tabori U, Bouffet E. Phase II Weekly Vinblastine for Chemotherapy-Naïve Children With Progressive Low-Grade Glioma: A Canadian Pediatric Brain Tumor Consortium Study. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2016 Oct 10:34(29):3537-3543. doi: 10.1200/JCO.2016.68.1585. Epub [PubMed PMID: 27573663]
Fangusaro J, Onar-Thomas A, Young Poussaint T, Wu S, Ligon AH, Lindeman N, Banerjee A, Packer RJ, Kilburn LB, Goldman S, Pollack IF, Qaddoumi I, Jakacki RI, Fisher PG, Dhall G, Baxter P, Kreissman SG, Stewart CF, Jones DTW, Pfister SM, Vezina G, Stern JS, Panigrahy A, Patay Z, Tamrazi B, Jones JY, Haque SS, Enterline DS, Cha S, Fisher MJ, Doyle LA, Smith M, Dunkel IJ, Fouladi M. Selumetinib in paediatric patients with BRAF-aberrant or neurofibromatosis type 1-associated recurrent, refractory, or progressive low-grade glioma: a multicentre, phase 2 trial. The Lancet. Oncology. 2019 Jul:20(7):1011-1022. doi: 10.1016/S1470-2045(19)30277-3. Epub 2019 May 28 [PubMed PMID: 31151904]
Wen PY, Stein A, van den Bent M, De Greve J, Wick A, de Vos FYFL, von Bubnoff N, van Linde ME, Lai A, Prager GW, Campone M, Fasolo A, Lopez-Martin JA, Kim TM, Mason WP, Hofheinz RD, Blay JY, Cho DC, Gazzah A, Pouessel D, Yachnin J, Boran A, Burgess P, Ilankumaran P, Gasal E, Subbiah V. Dabrafenib plus trametinib in patients with BRAF(V600E)-mutant low-grade and high-grade glioma (ROAR): a multicentre, open-label, single-arm, phase 2, basket trial. The Lancet. Oncology. 2022 Jan:23(1):53-64. doi: 10.1016/S1470-2045(21)00578-7. Epub 2021 Nov 24 [PubMed PMID: 34838156]
Slater A, Moore NR, Huson SM. The natural history of cerebellar hemangioblastomas in von Hippel-Lindau disease. AJNR. American journal of neuroradiology. 2003 Sep:24(8):1570-4 [PubMed PMID: 13679272]
Fadul C, Misulis KE, Wiley RG. Cerebellar metastases: diagnostic and management considerations. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 1987 Jul:5(7):1107-15 [PubMed PMID: 3037034]
Level 3 (low-level) evidenceHo VB, Smirniotopoulos JG, Murphy FM, Rushing EJ. Radiologic-pathologic correlation: hemangioblastoma. AJNR. American journal of neuroradiology. 1992 Sep-Oct:13(5):1343-52 [PubMed PMID: 1414827]
Level 3 (low-level) evidencePark JH, Jung N, Kang SJ, Kim HS, Kim E, Lee HJ, Jung HR, Choe M, Shim YJ. Survival and Prognosis of Patients with Pilocytic Astrocytoma: A Single-Center Study. Brain tumor research and treatment. 2019 Oct:7(2):92-97. doi: 10.14791/btrt.2019.7.e36. Epub [PubMed PMID: 31686439]
Bucy PC, Thieman PW. Astrocytomas of the cerebellum. A study of a series of patients operated upon over 28 years ago. Archives of neurology. 1968 Jan:18(1):14-9 [PubMed PMID: 5634368]
Paulino AC. Collins' law revisited: can we reliably predict the time to recurrence in common pediatric tumors? Pediatric hematology and oncology. 2006 Jan-Feb:23(1):81-6 [PubMed PMID: 16326417]
McAuley E, Brophy H, Hayden J, Pettorini B, Parks C, Avula S, Mallucci C, Pizer B. The benefit of surveillance imaging for paediatric cerebellar pilocytic astrocytoma. Child's nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery. 2019 May:35(5):801-805. doi: 10.1007/s00381-019-04078-3. Epub 2019 Feb 15 [PubMed PMID: 30770993]
Villanueva KG, Rea ND, Krieger MD. Novel Surgical and Radiologic Risk Factors for Progression or Recurrence of Pediatric Pilocytic Astrocytoma. Pediatric neurosurgery. 2019:54(6):375-385. doi: 10.1159/000503110. Epub 2019 Oct 8 [PubMed PMID: 31593969]
Mair MJ, Wöhrer A, Furtner J, Simonovska A, Kiesel B, Oberndorfer S, Ungersböck K, Marosi C, Sahm F, Hainfellner JA, Rössler K, Preusser M, Widhalm G, Berghoff AS. Clinical characteristics and prognostic factors of adult patients with pilocytic astrocytoma. Journal of neuro-oncology. 2020 May:148(1):187-198. doi: 10.1007/s11060-020-03513-9. Epub 2020 Apr 27 [PubMed PMID: 32342331]
Albright L, Reigel DH. Management of hydrocephalus secondary to posterior fossa tumors. Journal of neurosurgery. 1977 Jan:46(1):52-5 [PubMed PMID: 830815]
Stein BM, Tenner MS, Fraser RA. Hydrocephalus following removal of cerebellar astrocytomas in children. Journal of neurosurgery. 1972 Jun:36(6):763-8 [PubMed PMID: 4537415]
Schwartz AM, Ghatak NR. Malignant transformation of benign cerebellar astrocytoma. Cancer. 1990 Jan 15:65(2):333-6 [PubMed PMID: 2403835]
Level 3 (low-level) evidenceBernell WR, Kepes JJ, Seitz EP. Late malignant recurrence of childhood cerebellar astrocytoma. Report of two cases. Journal of neurosurgery. 1972 Oct:37(4):470-4 [PubMed PMID: 5070873]
Level 3 (low-level) evidence