Introduction
Optic nerve sheath decompression, more commonly known as optic nerve sheath fenestration (ONSF), is a surgical procedure performed to decompress the optic nerves and relieve vision-threatening papilledema in the setting of elevated intracranial pressure (ICP). Performance of this procedure is generally in cases of pseudotumor cerebri syndrome (PTC), where patients are rapidly losing vision from significantly elevated ICP, or in cases of progressive vision loss in patients who do not respond to medical therapy or are non-adherent with therapy. The exact mechanism by which fenestration relieves papilledema is not entirely understood at this time; however, both decompression of the perioptic subarachnoid space with filtration of cerebrospinal fluid (CSF) out of the subarachnoid space through the dural opening as well as scarring of the surgical site that prevents further accumulation of CSF are felt to be contributing factors.[1] Optic nerve sheath decompression does not treat the source of the elevated ICP itself, but the procedure does diminish the associated visual sequelae.
Anatomy and Physiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Anatomy and Physiology
The optic nerve is distinctive among cranial nerves in that it is a central nervous system (CNS) white matter tract that shares a similar developmental origin with the CNS. As such, the optic nerve covering is the same outer connective tissue membranes as the brain: the pia, arachnoid, and dura mater. The pia lies on the surface of the brain and follows along the brain’s gyri and sulci. The arachnoid mater lies just above the pia mater, and CSF flows in the subarachnoid space (SAS) between the pia and the arachnoid. The dura mater is a strong connective tissue covering that overlies the other arachnoid and pia mater, encasing the brain and spinal cord. The SAS of the optic nerve is contiguous with that of the brain via the suprasellar cistern, allowing the circulation of CSF between the intracranial SAS and the optic nerves in a 3-compartment system. As a result, increases in ICP can be transmitted to the optic nerves and can cause optic disc edema known, in this clinical setting, as papilledema. Optic nerve sheath fenestration is a surgical procedure performed to relieve perioptic pressure in cases of elevated ICP causing papilledema.
The subarachnoid space of the human optic nerve is not homogenous but possesses a complex system of arachnoid trabeculae and septa that subdivide the subarachnoid space between the arachnoid and pia mater. The trabeculae, septa, and pillars vary in their density as well as their arrangement depending upon their specific location within the different portions of the optic nerve. They appear to be more densely and tightly packed in the immediate retrobulbar segment of the optic nerve than the more distal mid-orbital and canalicular segments. This variability may play a role in the CSF dynamics between the subarachnoid space of the optic nerve and the suprasellar cistern that connects it to the intracranial space and may contribute to the understanding of the pathophysiology of papilledema and the sometimes variable response seen to ONSF.[2]
Increased ICP can result from a multitude of CNS disease states, including intracranial hypertension, venous sinus thrombosis, CNS infections, inflammatory diseases, subarachnoid hemorrhage, and intracranial malignancies or other lesions causing obstructive hydrocephalus. Vision loss in the setting of papilledema is thought to occur as a result of the disruption of axonal transport and intraneuronal ischemic damage to the optic nerve.[3][4] An increase in CSF pressure increases tissue pressure on the optic nerve axons and disrupts the gradient between intraocular and retrolaminar pressure. This results in defective axonal transport, followed by intra-axonal swelling, which is seen as papilledema. This swelling can secondarily compress small arterioles to the nerve, consequently causing intraneuronal ischemia. Clinically, this manifests as vision loss, initially as blind spot enlargement, followed by loss of peripheral and then central vision and field loss. Ultimately, optic nerve atrophy ensues in the setting of chronically elevated ICP and papilledema, often with profound vision loss.
Indications
Indications for ONSF
As stated earlier, the indication for optic nerve sheath fenestration is in the setting of vision loss due to elevated ICP causing papilledema. Increased ICP can result from a multitude of CNS disease states. The most common of these include PTC syndrome, cerebral venous sinus thrombosis, and intracranial tumors causing ICP elevation from decreased CSF outflow. Other less common and historical indications are discussed as well.
Pseudotumor Cerebri Syndrome (Idiopathic Intracranial Hypertension)
ONSF is most frequent in the setting of PTC, also known as benign intracranial hypertension. It is a syndrome of elevated ICP in the absence of an underlying CNS pathology. Idiopathic intracranial hypertension (IIH) is a subset of PTC syndrome in which no secondary cause (e.g., transverse sinus stenosis, venous sinus thrombosis, corticosteroid use, hormonal abnormality, etc.) is present. IIH most commonly presents in women of childbearing age who are obese or have recently gained weight. Patients commonly present with symptoms of elevated ICP, including headaches and pulsatile tinnitus, as well as visual symptoms of transient visual obscurations and diplopia from unilateral or bilateral cranial nerve (CN) VI palsies.[5] Severe visual loss from progressive field constriction is typically a late finding associated with significantly elevated ICP and can lead to blindness in 5% of patients.[4] Non-invasive treatment measures include weight loss, acetazolamide, and furosemide.
Diagnostic criteria for IIH in adults and children were revised in 2013 by Friedman et al.[6] Definitive IIH will exhibit the following:
- Papilledema
- Normal neurologic exam, except for cranial neuropathies
- Neuroimaging will show one or more of the following:
- Typical female patient with obesity: MRI will show normal brain parenchyma and no meningeal enhancement without hydrocephalus, masses, or structural lesions
- Other types of patients: normal MRI and magnetic resonance venography
- If MRI is unavailable or unobtainable, CT imaging may is an option.
- Normal CSF composition
- Elevated opening lumbar puncture pressure of greater than 250 mm CSF in adults and greater than 280 mm CSF in children or 250 mm CSF in non-sedated or normal weight child
If papilledema is not present, IIH can be diagnosed if 2 to 5 of the above are present and if it is also unilateral or bilateral CN VI palsy. If both papilledema and CN VI palsy are absent, it is suggestive of IIH (but not positively diagnosed). If 2 to 5 of the above are present, and the patient has at least 3 of the following on neuroimaging: empty sella; flattening of the posterior globe; distension of the perioptic SAS with or without a tortuous optic nerve; and/or transverse venous sinus stenosis.[6]
Optic nerve sheath fenestration is an attempt to preserve visual function in patients with PTC who present with vision-threatening papilledema or who have not responded adequately to or cannot tolerate maximal medical therapy. It is an essential tool in the acute setting for patients presenting with “malignant” PTC (extremely high opening pressure, evidence of optic neuropathy, and poor prognosis if treated with medical therapy alone).[5] Serial lumbar punctures (LP) are not recommended to reduce ICP in this clinical setting, given the rapid reaccumulation of CSF and return of elevated ICP. CSF diversion procedures in the form of ventriculoperitoneal shunting (VPS) or lumboperitoneal shunting (LPS) can also be used to decrease ICP. While they can be effective at reducing papilledema and preventing vision loss, there is the attendant risk of brain and spinal surgery and the not uncommon complication of shunt failure. ONSF and CSF diversion procedures may both be necessary for patients with malignant PTC syndrome, as well as those with severe vision loss and ICP elevation in PTC refractory to conservative management.
Unilateral ONSF can result in papilledema resolution in both eyes; however, in most cases of severe papilledema and vision loss, bilateral ONSF is required. A review of unilateral ONSF shows that the unfenestrated eye typically shows less papilledema reduction than the operated eye.[7] Some patients may also experience an improvement in their headaches, but ONSF is not primarily for this indication. Repeat ONSF may be necessary on patients with recurrent visual disturbance or further visual deterioration after surgery.[8][5][9] A ventriculoperitoneal shunt is preferable in most cases in this setting; however, scarring of the optic nerve sheath from prior surgery makes repeat fenestration more technically challenging, and there is a potentially higher risk of complications.
Cerebral Vein Thrombosis or Stenosis
Obstruction of venous drainage from the cerebral venous system secondary to thrombosis or stenosis can result in increased ICP and papilledema from decreased CSF outflow. This condition can present very similarly to PTC and IIH but tends to have a more rapid onset and more severe visual loss from significant ICP elevation; it is not limited to the demographics of PTC syndrome and should be suspected in non-obese women or men with symptoms and signs of PTC syndrome. It presents in patients with prothrombotic states, including pregnancy, oral contraceptives, Factor V Leiden mutations, Factor XII deficiency, G20210A mutations, antithrombin III, and protein S deficiency. Therapies include anticoagulation, stenting, or thrombectomy of the cerebral sinus and CSF diversion procedures. ONSF can also be performed to protect visual function in patients with vision-threatening papilledema. The European Federation of Neurological Sciences (EFNS) 2010 guidelines appropriates ONSF in cases of serial lumbar punctures, acetazolamide, and/or VPS do not prevent the progression of visual loss in these conditions.[5][10]
Intracranial Masses
Intracranial masses/tumors create increased ICP primarily by restricting the flow of CSF through the ventricles or outflow of CSF and blood from the brain via the dural venous sinuses. Optic nerve sheath fenestration is a viable option for patients in whom ICP develops rapidly and causes profound vision loss and in whom complete resection of the mass is not possible with resulting persistent intracranial hypertension. Performing ONSF can dramatically increase the quality of life in these patients by preserving their vision. Of all indications for ONSF, this has the poorest visual prognosis, as the surgery is generally a therapeutic option in late-stage patients with intracranial illness.
Other Indications for ONSF
Cryptococcal meningitis: up to 40% of patients develop ocular disease; papilledema is the most common manifestation of CNS infection and may result in a profound visual loss for patients who survive. Cryptococcal meningitis usually results in a very high organism burden in the CSF and marked inflammation; this leads to increased ICP from the possible aggregation of cryptococcal capsular polysaccharide and the resulting CSF outflow obstruction through arachnoid granulations.[11] By some physicians, consideration of early ONSF is essential to avoid visual impairment. It is a reasonable consideration to practice caution before performing invasive surgical procedures in an infected optic nerve. However, studies show that postoperative orbital infections had not occurred even when histopathological studies revealed numerous cryptococci.[12][13] This is true even if ONSF takes place before antifungal medication administration.[5][14] Studies have shown that patients with papilledema from cryptococcal meningitis should be considered for ONSF early in the course, even if the infection is at an active stage in the disease.[5][15]
Local Disease of the Optic Nerve
- Traumatic optic neuropathy (TON)
- This can cause elevated intraorbital pressure. It commonly manifests early as an optic nerve hematoma. The treatment of TON is controversial, and a large prospective study showed no difference in outcomes whether treatment for TON was with observation, corticosteroids, or optic nerve decompression.[5]
- Optic nerve tumors
- The most common optic nerve tumors are gliomas and meningiomas. Visual improvement with ONSF in patients with meningiomas has mixed results in reported cases. Other optic nerve tumors reported having visual improvement after ONSF include bilateral infiltration with T-cell non-Hodgkin lymphoma and metastatic breast cancer.[5]
- Optic nerve drusen
- This may uncommonly cause central and peripheral visual loss through an unknown mechanism. A few small studies showed some improvement in the management of visual loss via ONSF. To date, no clear recommendations exist on the treatment of optic nerve drusen with ONSF. Further studies are warranted.[5][16]
Contraindications
Optic nerve sheath fenestration should not be performed in patients on chronic anticoagulation due to the attendant risk of bleeding into the orbit. The procedure should typically be avoided in patients with CNS infections due to the potential for seeding of the orbit with the infectious organism. It is typically not performed in patients with mild or moderate vision loss related to ICP elevation, as they should receive a full trial of medical therapy prior to the consideration of surgical intervention. While headache relief can result in up to 50% of patients [17], it is not indicated for the management of a headache in patients with PTC syndrome and patients with ICP elevation without optic disc edema or vision loss.
Technique or Treatment
Surgical Approaches
De Wecker was the first to describe optic nerve sheath fenestration in 1872. There are several surgical approaches for accessing the orbit for fenestration of the optic nerves; however, the final goal is to create a window or a series of slits in the optic nerve sheath just behind the globe to release CSF under pressure causing compression of the nerve.
Ophthalmologists typically perform the surgery after training in orbital surgery and/or neuro-ophthalmology. Based on a 2015 survey of ophthalmologists who perform ONSF, the three most commonly utilized surgical approaches are the medial transconjunctival approach (59%), the superomedial lid crease incision (31%), and lateral orbitotomy (10%).[18] The choice of surgical procedure employed is surgeon-specific, depending on individual surgical training and comfort level with their chosen technique.
All procedures are performed under general anesthesia and magnification either with loupes or a surgical microscope. Surgical preparation and draping should take place in the usual fashion for ophthalmic plastic surgery. Avoidance of injury to the posterior ciliary arteries that course along the surface of the optic nerve sheath and supply the optic nerve and choroid is critical to prevent ischemic optic neuropathy or choroidal infarction.
Medial Transconjunctival Approach
The medial transconjunctival approach is the most widely used approach for exposure and fenestration of the optic nerve. It was first described in 1973 by Galbraith and Sullivan.[19] One significant benefit of this approach is that it offers quick access to the retro-orbital optic nerve without creating an incision in the skin. However, it requires disinsertion of the medial rectus muscle that can result in postoperative strabismus and diplopia.
Technique
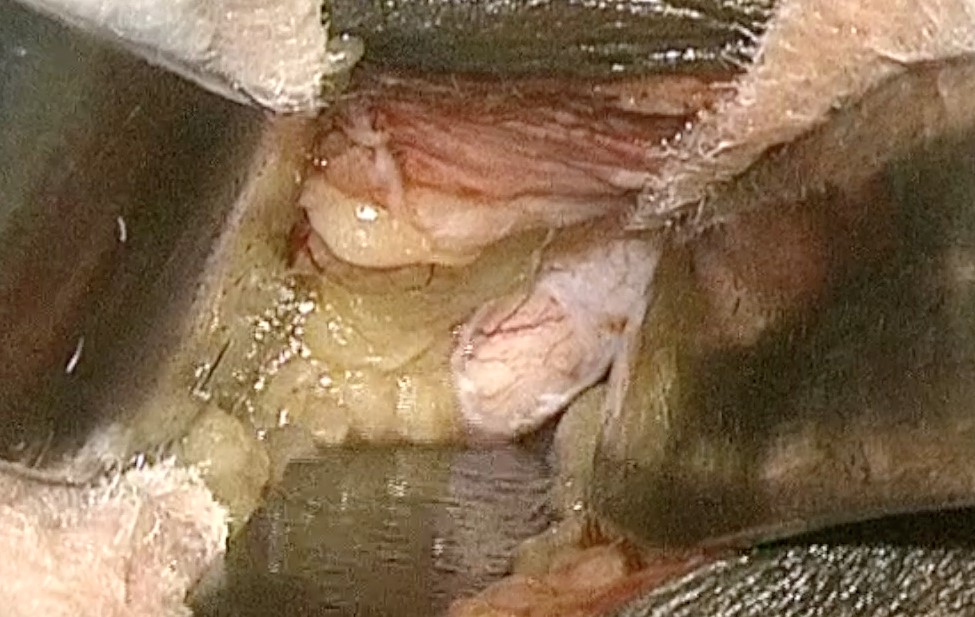
After the establishment of general anesthesia and patient preparation, a lid speculum is placed. A limbal peritomy is made nasally, and dissection is performed down to bare sclera, where the medial rectus muscle is identified. The medial rectus muscle is isolated with muscle hooks and secured with a 6-0 double-armed polyglactin at the insertion. The muscle is transected from the globe anterior to the suture. A traction suture may be passed through the muscle stump at the insertion so that the globe may be abducted for enhanced exposure. Continuous attention to pupil size and reaction to light is critical in the monitoring of optic nerve integrity throughout the rest of the procedure. Gentle blunt dissection is carried down along the medial aspect of the globe and into the intraconal space using the surgeon’s orbital retractor of choice. Once the optic nerve is visualized, the operating microscope is brought over the field. A fine, long forceps, such as a myringotomy or bayonet, is used to grasp the optic nerve sheath. Sharp scissors are used to incise the optic nerve sheath at least 1 mm away from the insertion of the nerve. Alternatively, a long fine-cutting instrument, such as a super sharp paracentesis knife, may be used to make the first cut. A gush of CSF indicates successful full-thickness penetration of the sheath. Additional cuts with the scissors are made until a window of tissue is excised and removed. All orbital instruments are removed, and the medial rectus muscle is reattached at its insertion. Hemostasis is confirmed, and the conjunctiva is closed with the surgeon’s suture of preference.
Farris and Lai described an alternative to this procedure in 2014. Their approach entails a transconjunctival approach to the medial intraconal space; however, instead of detaching the medial rectus muscle, this muscle and the superior rectus are engaged on bridle sutures used to rotate the globe inferiorly and temporally. The orbit is then entered with an orbital retractor in the space between these muscles to access and fenestrate the optic nerve as described above.
Superomedial Lid Crease Incision
Pelton and Patel described this surgical approach in 2001.[20] Historically, it has found wide utilization by orbital surgeons for access to the medial intraconal space for removal of intraorbital lesions. By creating the incision in the crease of the upper eyelid, the scar is well-hidden and is appealing for some patients for cosmetic considerations. It is favored by some orbital surgeons because it can be performed more quickly than the classical medial transconjunctival approach, affords a direct angle of approach to the optic nerve, and does not require disinsertion of any of the extraocular muscles. Additionally, an operative microscope is usually unnecessary due to the more direct approach to the nerve as compared to the medial orbitotomy approach. The dissection takes place medial to the levator aponeurosis and optic nerve, and as such, there is little danger of ptosis, ciliary ganglion injury, or strabismus. The constraining factor with this approach is the increased distance from the incision site to the optic nerve, and it is relatively more technically challenging to perform.
Technique
The lid crease is marked, and a local anesthetic with epinephrine is injected subcutaneously. After the establishment of general anesthesia and patient preparation, an incision is made through the medial one-half to two-thirds of the crease over the previously made marking. Sharp dissection is carried through the orbicularis and the septum either with cutting cautery or Wescott scissors. The nasal and central (preaponeurotic) fat pads are identified, and careful blunt dissection is performed between them into the orbit between the levator aponeurosis and superior oblique muscle. Dissection is carried out inferiorly and posteriorly toward the back of the globe in the loose areolar orbital fat until the optic nerve is strummed and then visualized. Continuous attention to pupil size and reaction to light is critical for the monitoring of optic nerve integrity throughout the rest of the procedure. Neurosurgical cottonoids may then be used to pack around the nerve. An operating microscope may be brought over the field at this point per surgeon preference, or loupes may be utilized for magnification. A fine long forceps, such as a myringotomy or bayonet, is used to grasp the optic nerve sheath. An incision is then made in the optic nerve sheath at least 1 mm away from the insertion of the nerve using the surgeon’s instrument of choice, taking care to avoid contact with the nerve itself. A gush of CSF indicates successful full-thickness penetration of the sheath. Additional cuts with the scissors are made until a window of tissue is excised and removed. Hemostasis is confirmed, and all instruments are removed from the orbit. The skin is closed with 6-0 plain gut sutures.
Lateral Orbitotomy Approach
This approach was first described for use in ONSF in 1872 by De Wecker and then subsequently in 1988 by Tse et al. and Patel and Anderson, who performed the lateral approach without removal of the lateral orbital wall.[21] The benefits include an excellent perpendicular view of the optic nerve without the requirement of muscle disinsertion. However, it can potentially result in increased operating times and injury to the ciliary ganglion. Additionally, an external incision is required.
Technique
After the establishment of general anesthesia and patient preparation, a lateral canthotomy with cantholysis is performed. A traction suture is placed through the cut edge of the upper eyelid and the lid margin. This process is repeated for the lower eyelid. Finally, a third traction suture can be placed at the insertion of the lateral rectus muscle. The globe is adducted using the traction suture. The conjunctiva is incised with Wescott scissors as far laterally as possible, taking care not to injure the lacrimal gland or lateral rectus muscle belly. Blunt dissection with small malleables is carried out between the lacrimal gland and the lateral rectus muscle, directed posterior to the globe. It is often necessary to cut through any intermuscular septa that may be present before the intraconal compartment becomes accessible. Blunt dissection is continued through the intraconal fat until the optic nerve is identified. Neurosurgical cottonoids may then be used to pack around the nerve. As focus shifts to the optic nerve, an operating microscope may be brought over the field at this point per surgeon preference, or loupes may be utilized for magnification. A fine, long forceps, such as a myringotomy or bayonet, is used to grasp the optic nerve sheath. An incision is then made in the optic nerve sheath at least 1 mm away from the insertion of the nerve using the surgeon’s instrument of choice, taking care to avoid contact with the nerve itself. A gush of CSF indicates successful full-thickness penetration of the sheath. Additional cuts with the scissors are made until a window of tissue is excised and removed. Hemostasis is confirmed, and the canthotomy and skin are closed using the surgeon’s preferred technique.
A revised lateral approach is another technique that obviates the need to traverse the intraconal fat by following the curve of the globe with the posterior dissection. In this way, it is similar to the medial transconjunctival approach but takes advantage of the additional space afforded by the anatomy.[22]
Complications
Outcomes and Complications of ONSF for PTC/IIH
Optic nerve sheath fenestration is most commonly performed in the setting of IIH; therefore, most of the data published on postoperative complications and outcomes are in this clinical context. There are a variety of reported complications, most of which are transient and minor. There is no published comparison of outcomes and complications between different ONSF techniques.
In 2017, Kalyvas et al. looked at ONSF efficacy, complications, and the associated costs of surgical procedures for IIH. There were 525 ONSF procedures performed on 341 patients, with an average follow-up time of 42.3 months. The medial approach was performed in 342 eyes, the lateral in 53 eyes, combined in 3 eyes, and superomedial lid incision in 1 eye. They concluded that ONSF could reduce papilledema and improve vision. Disc swelling improved in 95% of patients, visual acuity improved in 67%, and visual fields showed improvement in 64%. However, ONSF was less efficacious in headache relief (41%). Approximately 11% required a second fenestration procedure even after initially improving.[23] In another study, 95% of patients who underwent ONSF had improved visual acuity and visual fields with no reported intraoperative complications. Mean follow-up was 18.7 months, and postoperative complications were ocular misalignment (6%) and corneal dellen (0.8%).[24][16]
Interestingly, a retrospective chart review published in 2011 by Alsuhaibani et al. showed bilateral improvement of papilledema and vision with unilateral ONSF.[7][24] A search of the literature on outcomes of ONSF for IIH overwhelmingly reveals a marked improvement in greater than 90% of patients in nearly every study, particularly in cases with acute papilledema.
In a 2008 prospective study in India that compared ONSF for papilledema from IIH vs. cerebral venous thrombosis, there was an improvement in 94% of patients in optic disc edema and vision with minimal and transient complications. These complications included tonic pupil (13.4%), diplopia (3.4%), and orbital cellulitis in 1 patient. Visual worsening was found in 2 eyes at 1 month postoperatively that failed to improve despite repeat fenestration.[25]
Fonseca et al. compared ONSF to CSF diversion procedures. They found that CSF shunting was superior to ONSF regarding visual improvement. However, ONSF patients in this study had worse preoperative papilledema and visual acuity. There were no complications reported, but 21% of ONSF patients eventually required CSF shunting.[26][24]
Additional studies on repeat fenestrations for papilledema also show significant improvement in visual function despite requiring multiple surgeries. There were two studies published in 1991 that looked at repeat ONSF procedures. In Kelman et al., 12 patients required repeated decompression with concurrent functioning lumboperitoneal shunts. All 12 patients showed improvement in visual function without surgical complications.[27] Spoor et al. studied 13 of 53 IIH patients with acute papilledema and visual loss who recently underwent ONSF. Eleven of 13 patients showed visual improvement even after secondary or tertiary decompressions.[28][24]
In general, visual outcomes tend to be worse the longer a nerve has been subjected to elevated perioptic ICP causing irreversible loss of axonal function. The earlier the intervention can be instituted to prevent progressive vision loss, the better.
Although not employed solely for the treatment of headaches, ONSF may reduce headaches in over half of patients with IIH undergoing the procedure.[9][17] The mechanism for the improvement of headaches after ONSF is unclear.
The most feared and serious complication of ONSF is complete visual loss, which occurs either as a complication of the procedure or therapeutic failure of an uncomplicated fenestration despite improvement of the appearance of the optic nerve. The risk of blindness from ONSF is rare and reportedly 1 to 2% based on a large series study.[29][30] Blindness occurs from damage to the posterior ciliary arteries causing arterial occlusion or ischemic neuropathy, and extraocular causes such as retrobulbar hemorrhage/hematoma formation. Optic nerve sheath cysts and pseudomeningoceles have also developed after ONSF and can present with postoperative vision changes, pain, and proptosis.[31] Ptosis, strabismus, or ciliary ganglion injury may also occur; however, the deficits related to these occurrences are often transient and minimal and are related to the surgical approach used for ONSF. The medial transconjunctival approach requires detachment of the medial rectus, which can result in exotropia and diplopia; however, the double vision is typically transient. Due to the close proximity of the posterior ciliary nerves and ciliary ganglion, the medial and lateral approaches may result in a tonic pupil.[29][32] Larger studies are required to determine the incidence of these complications.
Other potential complications of ONSF include the following:
- Conjunctiva and sclera: chemosis, globe perforation, conjunctival bleeding
- Cornea: corneal dellen, corneal ulceration
- Anterior chamber: acute angle-closure glaucoma, microhyphema
- Iris: tonic pupil
- Retina and choroid: branch retinal artery occlusion, central retinal artery occlusion, choroidal ischemia/infarction, chorioretinal scarring
- Optic nerve: optic nerve cysts/pseudomeningoceles, traumatic optic neuropathy
- Posterior ciliary nerve and ciliary ganglion: diplopia, ptosis, mydriasis
- Orbit: orbital apex syndrome, infection, hemorrhage/hematoma
- Extraocular muscles: strabismus, diplopia
Considering that visual loss is a very rare complication and others are relatively minor and transient, ONSF is an attractive option for the treatment of vision loss in PTC/IIH. This is especially true when considering the risks of alternative treatments, such as CSF diversion procedures and bariatric surgery.
Clinical Significance
Optical nerve sheath fenestration is a surgical procedure employed to prevent vision loss from papilledema in the setting of increased ICP. It is most commonly used in the prevention of vision loss from papilledema in IIH but has other indications reported in the literature. There are three major approaches to exposing the optic nerve for decompression, with the medial transconjunctival approach being the most common. Stability or improvement in visual function is seen in the majority of patients, as is a reduction of papilledema grade. The rate of major complications is relatively low, and many minor complications such as ptosis and diplopia are usually transient. Future studies are needed to evaluate other indications for ONSF and to compare outcomes between different surgical approaches. Comparison studies of ONSF and CSF diversion procedures for the treatment of visual compromise from papilledema will also be beneficial, and a major study is currently in the planning stages.
Enhancing Healthcare Team Outcomes
Prolonged optic nerve compression by significantly elevated ICP can cause irreversible ischemic injury to the optic nerve. While ONSF has been shown to improve visual outcomes in patients with papilledema, results tend to be better the earlier intervention takes place. Coordination between an interprofessional team of optometrists, primary care, emergency medicine, ophthalmologists, and nurses can improve outcomes. [Level 5]Because patients with malignant PTC or ICP elevation tend to present acutely and urgently, it is essential for ophthalmologists, primary care providers, and emergency care providers to be familiar with the signs and symptoms of ICP elevation and the potential for vision loss in this setting. Appropriate consultation with an ophthalmologist will demonstrate the requisite optic disc edema, vision loss, and peripheral field constriction associated with malignant PTC and prompt the appropriate workup to evaluate for an underlying cause of ICP elevation and the patient’s suitability for ONSF. Experienced orbital surgeons and neuro-ophthalmologists should perform the surgery in a controlled setting with experienced nursing support in which they are comfortable and familiar. It is sometimes necessary to get neurosurgery involved for consideration of a CSF diversion procedure in the cases where ONSF fails to prevent further vision loss or in severe cases, such as tumors, where shunting is indicated. [Level 3]
Media
(Click Image to Enlarge)
References
Davidson SI. A surgical approach to plerocephalic disc oedema. Transactions of the ophthalmological societies of the United Kingdom. 1970:89():669-90 [PubMed PMID: 5276692]
Killer HE, Laeng HR, Flammer J, Groscurth P. Architecture of arachnoid trabeculae, pillars, and septa in the subarachnoid space of the human optic nerve: anatomy and clinical considerations. The British journal of ophthalmology. 2003 Jun:87(6):777-81 [PubMed PMID: 12770980]
Rigi M, Almarzouqi SJ, Morgan ML, Lee AG. Papilledema: epidemiology, etiology, and clinical management. Eye and brain. 2015:7():47-57. doi: 10.2147/EB.S69174. Epub 2015 Aug 17 [PubMed PMID: 28539794]
Level 2 (mid-level) evidenceWall M. Idiopathic intracranial hypertension. Neurologic clinics. 2010 Aug:28(3):593-617. doi: 10.1016/j.ncl.2010.03.003. Epub [PubMed PMID: 20637991]
Chen H, Zhang Q, Tan S, Fu H, Farris BK, Yang Z. Update on the application of optic nerve sheath fenestration. Restorative neurology and neuroscience. 2017:35(3):275-286 [PubMed PMID: 28339414]
Friedman DI, Liu GT, Digre KB. Revised diagnostic criteria for the pseudotumor cerebri syndrome in adults and children. Neurology. 2013 Sep 24:81(13):1159-65. doi: 10.1212/WNL.0b013e3182a55f17. Epub 2013 Aug 21 [PubMed PMID: 23966248]
Alsuhaibani AH, Carter KD, Nerad JA, Lee AG. Effect of optic nerve sheath fenestration on papilledema of the operated and the contralateral nonoperated eyes in idiopathic intracranial hypertension. Ophthalmology. 2011 Feb:118(2):412-4. doi: 10.1016/j.ophtha.2010.06.025. Epub [PubMed PMID: 20801522]
Level 2 (mid-level) evidenceBanta JT, Farris BK. Pseudotumor cerebri and optic nerve sheath decompression. Ophthalmology. 2000 Oct:107(10):1907-12 [PubMed PMID: 11013197]
Level 2 (mid-level) evidenceSergott RC, Savino PJ, Bosley TM. Optic nerve sheath decompression: a clinical review and proposed pathophysiologic mechanism. Australian and New Zealand journal of ophthalmology. 1990 Nov:18(4):365-73 [PubMed PMID: 2076284]
Einhäupl K, Stam J, Bousser MG, De Bruijn SF, Ferro JM, Martinelli I, Masuhr F, European Federation of Neurological Societies. EFNS guideline on the treatment of cerebral venous and sinus thrombosis in adult patients. European journal of neurology. 2010 Oct:17(10):1229-35. doi: 10.1111/j.1468-1331.2010.03011.x. Epub [PubMed PMID: 20402748]
Graybill JR, Sobel J, Saag M, van Der Horst C, Powderly W, Cloud G, Riser L, Hamill R, Dismukes W. Diagnosis and management of increased intracranial pressure in patients with AIDS and cryptococcal meningitis. The NIAID Mycoses Study Group and AIDS Cooperative Treatment Groups. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2000 Jan:30(1):47-54 [PubMed PMID: 10619732]
Level 1 (high-level) evidenceMilman T, Mirani N, Turbin RE. Optic nerve sheath fenestration in cryptococcal meningitis. Clinical ophthalmology (Auckland, N.Z.). 2008 Sep:2(3):637-9 [PubMed PMID: 19668765]
Level 3 (low-level) evidenceGarrity JA, Herman DC, Imes R, Fries P, Hughes CF, Campbell RJ. Optic nerve sheath decompression for visual loss in patients with acquired immunodeficiency syndrome and cryptococcal meningitis with papilledema. American journal of ophthalmology. 1993 Oct 15:116(4):472-8 [PubMed PMID: 8213978]
Level 3 (low-level) evidenceTan CT. Intracranial hypertension causing visual failure in cryptococcus meningitis. Journal of neurology, neurosurgery, and psychiatry. 1988 Jul:51(7):944-6 [PubMed PMID: 3204403]
Cremer PD, Johnston IH, Halmagyi GM. Pseudotumour cerebri syndrome due to cryptococcal meningitis. Journal of neurology, neurosurgery, and psychiatry. 1997 Jan:62(1):96-8 [PubMed PMID: 9010409]
Level 3 (low-level) evidenceMoreau A, Lao KC, Farris BK. Optic nerve sheath decompression: a surgical technique with minimal operative complications. Journal of neuro-ophthalmology : the official journal of the North American Neuro-Ophthalmology Society. 2014 Mar:34(1):34-8. doi: 10.1097/WNO.0000000000000065. Epub [PubMed PMID: 24275984]
Level 2 (mid-level) evidenceBrazis PW. Clinical review: the surgical treatment of idiopathic pseudotumour cerebri (idiopathic intracranial hypertension). Cephalalgia : an international journal of headache. 2008 Dec:28(12):1361-73. doi: 10.1111/j.1468-2982.2008.01778.x. Epub [PubMed PMID: 19037972]
Sobel RK, Syed NA, Carter KD, Allen RC. Optic Nerve Sheath Fenestration: Current Preferences in Surgical Approach and Biopsy. Ophthalmic plastic and reconstructive surgery. 2015 Jul-Aug:31(4):310-2. doi: 10.1097/IOP.0000000000000326. Epub [PubMed PMID: 26168208]
Galbraith JE, Sullivan JH. Decompression of the perioptic meninges for relief of papilledema. American journal of ophthalmology. 1973 Nov:76(5):687-92 [PubMed PMID: 4748189]
Pelton RW, Patel BC. Superomedial lid crease approach to the medial intraconal space: a new technique for access to the optic nerve and central space. Ophthalmic plastic and reconstructive surgery. 2001 Jul:17(4):241-53 [PubMed PMID: 11476174]
Level 3 (low-level) evidenceTse DT, Nerad JA, Anderson RL, Corbett JJ. Optic nerve sheath fenestration in pseudotumor cerebri. A lateral orbitotomy approach. Archives of ophthalmology (Chicago, Ill. : 1960). 1988 Oct:106(10):1458-62 [PubMed PMID: 3052386]
Blessing NW, Tse DT. Optic nerve sheath fenestration: a revised lateral approach for nerve access. Orbit (Amsterdam, Netherlands). 2019 Apr:38(2):137-143. doi: 10.1080/01676830.2018.1452949. Epub 2018 Mar 22 [PubMed PMID: 29565728]
Kalyvas AV, Hughes M, Koutsarnakis C, Moris D, Liakos F, Sakas DE, Stranjalis G, Fouyas I. Efficacy, complications and cost of surgical interventions for idiopathic intracranial hypertension: a systematic review of the literature. Acta neurochirurgica. 2017 Jan:159(1):33-49. doi: 10.1007/s00701-016-3010-2. Epub 2016 Nov 9 [PubMed PMID: 27830325]
Level 1 (high-level) evidenceGilbert AL, Chwalisz B, Mallery R. Complications of Optic Nerve Sheath Fenestration as a Treatment for Idiopathic Intracranial Hypertension. Seminars in ophthalmology. 2018:33(1):36-41. doi: 10.1080/08820538.2017.1353810. Epub [PubMed PMID: 29420144]
Nithyanandam S, Manayath GJ, Battu RR. Optic nerve sheath decompression for visual loss in intracranial hypertension: report from a tertiary care center in South India. Indian journal of ophthalmology. 2008 Mar-Apr:56(2):115-20 [PubMed PMID: 18292621]
Fonseca PL, Rigamonti D, Miller NR, Subramanian PS. Visual outcomes of surgical intervention for pseudotumour cerebri: optic nerve sheath fenestration versus cerebrospinal fluid diversion. The British journal of ophthalmology. 2014 Oct:98(10):1360-3. doi: 10.1136/bjophthalmol-2014-304953. Epub 2014 May 12 [PubMed PMID: 24820047]
Level 2 (mid-level) evidenceKelman SE, Sergott RC, Cioffi GA, Savino PJ, Bosley TM, Elman MJ. Modified optic nerve decompression in patients with functioning lumboperitoneal shunts and progressive visual loss. Ophthalmology. 1991 Sep:98(9):1449-53 [PubMed PMID: 1945323]
Spoor TC, Ramocki JM, Madion MP, Wilkinson MJ. Treatment of pseudotumor cerebri by primary and secondary optic nerve sheath decompression. American journal of ophthalmology. 1991 Aug 15:112(2):177-85 [PubMed PMID: 1867302]
Mudumbai RC. Optic nerve sheath fenestration: indications, techniques, mechanisms and, results. International ophthalmology clinics. 2014 Winter:54(1):43-9. doi: 10.1097/IIO.0000000000000008. Epub [PubMed PMID: 24296370]
Sergott RC, Savino PJ, Bosley TM. Modified optic nerve sheath decompression provides long-term visual improvement for pseudotumor cerebri. Archives of ophthalmology (Chicago, Ill. : 1960). 1988 Oct:106(10):1384-90 [PubMed PMID: 3178549]
Naqvi SM, Thiagarajah C, Golnik K, Lee A, Kersten R, Nerad J. Optic nerve cyst-like formation presenting as a delayed complication of optic nerve sheath fenestration. Ophthalmic plastic and reconstructive surgery. 2014 May-Jun:30(3):e53-4. doi: 10.1097/IOP.0b013e31829bb3e8. Epub [PubMed PMID: 23880976]
Level 3 (low-level) evidenceArai H, Sato K, Katsuta T, Rhoton AL Jr. Lateral approach to intraorbital lesions: anatomic and surgical considerations. Neurosurgery. 1996 Dec:39(6):1157-62; discussion 1162-3 [PubMed PMID: 8938770]