Neuroanatomy, Middle Cerebral Artery
Neuroanatomy, Middle Cerebral Artery
Introduction
The middle cerebral artery (MCA) is a critical blood vessel with significant anatomical, clinical, and educational importance. As a major branch of the internal carotid artery (ICA), the MCA plays an integral role in the circle of Willis, the anastomotic system connecting the anterior and posterior cerebral circulations. Oxygenated blood flows through the MCA to the lateral frontal, parietal, and temporal lobes, as well as key subcortical regions, including the basal ganglia and internal capsule.
The MCA is the most commonly affected vessel in cerebrovascular pathology, particularly in ischemic strokes, a leading cause of mortality and disability worldwide. MCA occlusion often leads to extensive neurological deficits due to the artery’s broad vascular territory. Advances in neuroimaging and endovascular therapy have further highlighted the clinical importance of this vessel, enabling targeted interventions for acute stroke management.
This activity explores the MCA’s anatomy, functional role, and clinical implications. Emphasis shall be given to this artery's relevance in cerebrovascular diseases and the necessity of its study for continuing medical education.
Structure and Function
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Structure and Function
The aortic arch gives rise to the brachiocephalic artery, from which the right common carotid artery (CCA) subsequently emerges. The left CCA branches directly from the aortic arch just downstream of the brachiocephalic trunk. The left and right CCAs run parallel to each other and divide near the angle of the mandible into the external carotid artery and ICA. The ECA supplies the face and neck, branching off immediately, while the ICA remains unbranched until the origin of the ophthalmic artery bilaterally. Each ICA then bifurcates into the anterior cerebral artery (ACA) and MCA.
The MCA is the largest branch of the ICA and plays a vital role in cerebral perfusion. This blood vessel is anatomically divided into 4 main segments: M1 (sphenoidal), M2 (insular), M3 (opercular), and M4 (cortical). The M1 segment courses laterally from the ICA within the Sylvian fissure, giving rise to perforating lenticulostriate arteries that supply the basal ganglia and internal capsule. The term "sphenoidal segment" refers to the M1 segment’s parallel course along the sphenoid ridge. The M2 segment traverses the insular surface, where significant branching occurs. The M3 and M4 segments extend over the opercular and cortical surfaces, respectively.
The lenticulostriate vessels form a contiguous group, originating from the A2 and A1 segments of the ACA and the M1 and M2 segments of the MCA (see Image. Course of the Middle Cerebral Artery). These vessels are subdivided into medial and lateral subgroups. The most medial lenticulostriate artery, known as the recurrent artery of Heubner, frequently arises from the proximal A2 segment. A dominant recurrent artery of Heubner may supply the medial lenticulostriate territory of the proximal M1 segment, though in some cases, this region is perfused by other vessels from the MCA’s medial lenticulostriate group.[1]
The MCA's branching patterns exhibit significant variability. Most commonly, the main trunk bifurcates into superior and inferior trunks, though trifurcations and multiple trunks also occur. These trunks give rise to stem arteries, which supply cortical branches. Each hemisphere typically has 8 stem arteries (range: 6-11), varying in size and the number of areas they perfuse. Each generally supplies 1 to 5 cortical branches, often serving 1 cortical area or 1 to 2 adjacent areas. Single-area stem arteries frequently supply the temporooccipital, angular, and central regions, while those supplying multiple areas usually perfuse regions beneath the Sylvian fissure.[2]
The stem arteries perfuse cortical areas organized by lobe. The frontal lobe includes the orbitofrontal, prefrontal, precentral, and central regions. The parietal lobe includes the anterior parietal, posterior parietal, and angular regions. The temporal and occipital lobes include the temporopolar, anterior temporal, middle temporal, posterior temporal, and temporooccipital regions. These areas are also called the "cortical divisions of the MCA vascular territory" (see Image. Middle Cerebral Artery Cortical Divisions). The MCA's cortical branches supply most of the lateral cerebral hemisphere, including the frontal, parietal, and temporal lobes.
Embryology
The development of the MCA reflects the integration of genetic programming and the metabolic demands of the embryonic brain. The MCA arises during early cerebrovascular morphogenesis, a process involving vasculogenesis and angiogenesis. Vasculogenesis establishes the primitive vascular plexus as angioblasts form endothelial tubes within the meninx primitiva, a connective tissue layer enveloping the neural tube. Angiogenesis, driven by hypoxia-induced factors such as vascular endothelial growth factor (VEGF) and stromal cell-derived factor 1 (SDF1), promotes vessel sprouting to meet the brain's increasing metabolic demands.[3]
The MCA originates from the ICAs, which form through the coalescence of the 3rd branchial arch artery with dorsal aortic segments. At the 32-day embryonic stage, primitive buds emerge from the ICA to create the early MCA and ACA. By 44 days, the MCA develops cortical branches that perfuse the expanding cerebral hemispheres. The development of this blood vessel is closely linked to the formation of the Sylvian fissure and insula, as its branches deepen alongside these structures.[4][5] By birth, the MCA reaches its mature configuration, emerging as a lateral branch of the ACA and supplying critical cortical and subcortical regions.[6]
The MCA’s embryology also highlights the origins of its variations, such as accessory or duplicated vessels. These anomalies often result from persistent embryonic anastomoses and influence collateral flow patterns in ischemic stroke. Additionally, the MCA’s branching pattern and structural anomalies reflect the interplay between metabolic demands and genetic determinants during development.
Physiologic Variants
The MCA exhibits significant anatomical variability, affecting both function and susceptibility to pathology. These physiological variants involve differences in branching patterns, vessel origins, and cortical supply territories.
Differences in Branching Patterns
The most common branching pattern is bifurcation, occurring in approximately 69.9% of cases, followed by trifurcation in 27% and tetrafurcation in 1%. The primary trunk variant, where the main trunk lacks major branches, is rare, seen in 1.9% of cases.[7]
M1 Course Variation
The 5 different subtypes of the course of the M1 segment for the performance of mechanical thrombectomy include the following:
- Oblique descent
- Convexity downward
- Straight
- Oblique course dorsad
- Straight dorsad
These anatomical differences influence procedural success rates and complications. Certain M1 configurations may be associated with a higher likelihood of complete recanalization, while others may predispose to residual thrombus retention.
Duplications and Accessory Arteries
Duplicated MCAs originate from the ICA and supply the anterior temporal lobe, while accessory MCAs typically arise from the ACA and perfuse frontal regions. The reported prevalence is 0.17% for duplications and 0.03% for accessory arteries.
Fenestrations
Fenestrations occur when a single arterial trunk splits into 2 separate channels before reuniting. Most MCA fenestrations appear in the M1 segment and are associated with early branches such as the temporopolar artery. Although rare (0.15% prevalence), fenestrations may contribute to aneurysm formation due to altered hemodynamics.
Twig-like Middle Cerebral Artery
Twig-like MCA is a rare anomaly characterized by an abnormal plexiform arterial network replacing the M1 segment. This variant may resemble moyamoya angiopathy but differs in its unilateral presentation and absence of posterior circulation involvement. Twig-like MCA is associated with higher rates of aneurysm formation and requires careful clinical and radiological follow-up (see Image. Important Anatomical Variations of the Middle Cerebral Artery).
M2 Dominance Patterns
The M2 segment typically divides into superior and inferior trunks, with variations in dominance. Codominance reflects a balanced supply pattern, while superior or inferior dominance affects cortical territories in the frontal, parietal, and temporal lobes. These patterns influence surgical and interventional strategies.
Clinical Implications
Anomalies in MCA branching and morphology may increase the risk of cerebrovascular events, including aneurysm formation and ischemia. Understanding these variations is essential for accurate imaging interpretation, surgical planning, and endovascular interventions. Further studies are needed to explore the clinical implications of these variants.[8]
Surgical Considerations
Microsurgical techniques remain essential for treating MCA aneurysms, particularly in younger patients and cases involving large aneurysms, wide-neck configurations, branch vessel involvement, or hematomas requiring evacuation. Endovascular techniques are often preferred for individuals who are frail, have aneurysms with calcification, or require intervention during the vasospasm period. Device and technique advancements continue to expand indications for endovascular treatment, including coiling for wide-neck aneurysms.
Preoperative Planning and Imaging
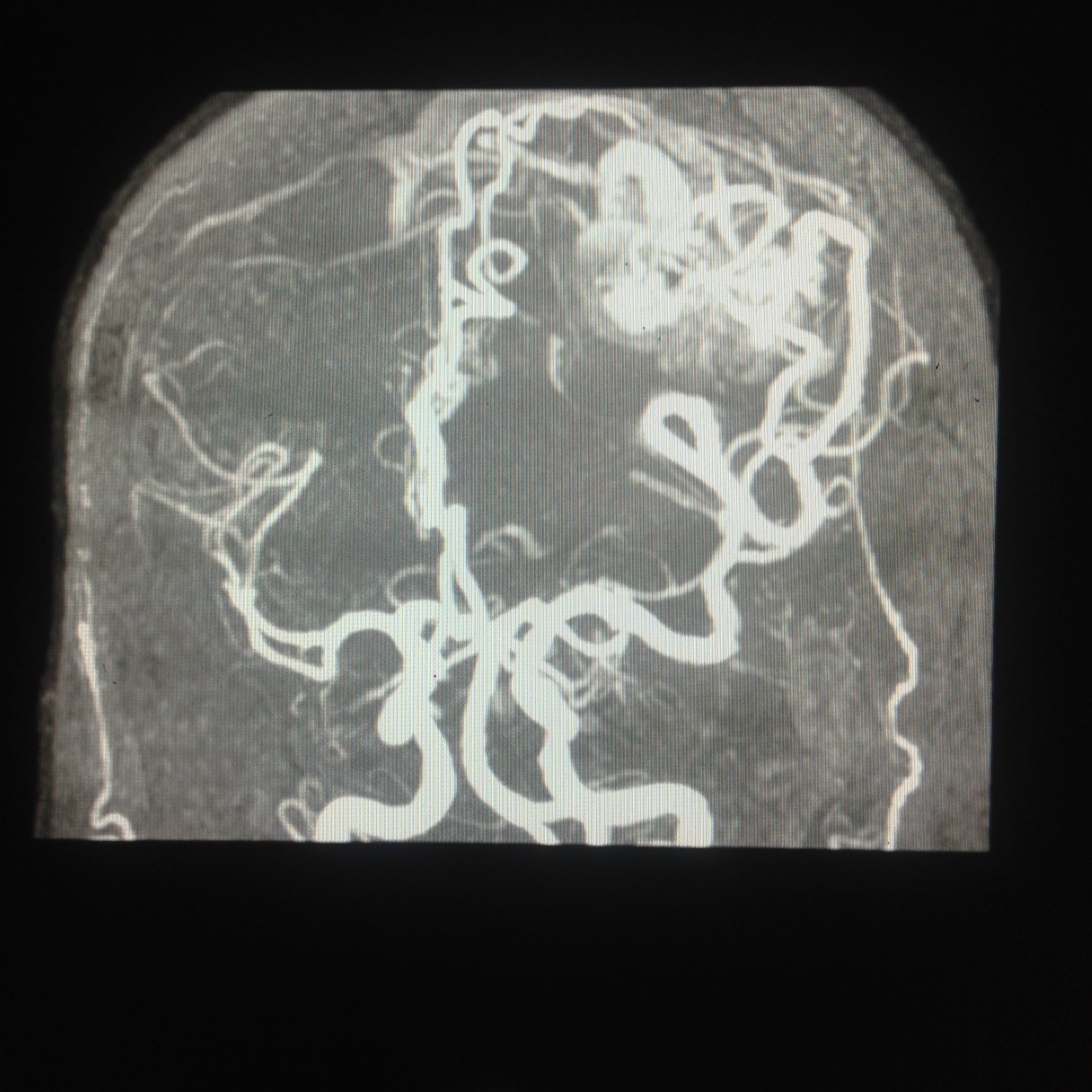
Advanced imaging modalities such as computed tomography angiography, magnetic resonance angiography, and digital subtraction angiography are critical for preoperative planning (see Image. Middle Cerebral Artery on Magnetic Resonance Angiography.) These imaging techniques provide detailed visualization of MCA anatomy and pathology, aiding in surgical approach selection, clip placement, and identification of anatomical variants or high-risk features.
Craniotomy and Surgical Exposure
A pterional craniotomy offers optimal exposure for MCA lesions. The patient is positioned supine with the head rotated 30° contralateral to the lesion, ensuring adequate access to the Sylvian fissure. A semicircular scalp incision is made from the zygoma to the midline, avoiding facial nerve branches. After raising the craniotomy flap, flattening the sphenoid ridge improves visualization of the proximal Sylvian fissure following dural opening.
Approaches to the Sylvian Fissure
The Sylvian fissure can be accessed through lateral or medial transsylvian approaches, with the choice depending on lesion location. The lateral transsylvian approach provides distal exposure, while the medial approach facilitates proximal control, particularly for M1 aneurysms. Sharp arachnoid dissection is essential to prevent damage to bridging veins or lenticulostriate arteries. Access through the superior temporal gyrus may be used for ruptured MCA aneurysms with large temporal lobe hematomas.[9]
Aneurysm Management
Wide-necked MCA aneurysms require advanced clipping techniques, including tandem and fenestrated clips, to ensure secure neck occlusion while preserving branch vessels. Temporary clipping of the M1 segment may reduce bleeding during dissection. Adenosine-induced cardiac arrest can provide a bloodless field for managing complex giant aneurysms during clipping.
Intraoperative Imaging and Hemodynamic Monitoring
Indocyanine green angiography and intraoperative digital subtraction angiography confirm clip placement, assess vessel patency, and identify residual aneurysm necks. Intraoperative Doppler sonography provides real-time blood flow evaluation.
Pitfalls and Bailout Options
Complications such as intraoperative rupture require rapid intervention. Temporary clipping and suction control help manage bleeding while securing the aneurysm. Suboptimal retraction or poor fissure dissection can increase the risk of vascular injury or venous infarction.
Postoperative Considerations
Immediate postoperative imaging evaluates clip positioning and assesses ischemic complications. Long-term follow-up imaging monitors aneurysm recurrence or residual flow in partially treated lesions.
Clinical Significance
The MCA is the largest branch of the ICA and is critical to cerebral circulation. This blood vessel supplies most of the lateral cerebral hemispheres, including regions essential for motor, sensory, and language functions. Deep perforators, the lenticulostriate arteries, arise from the MCA and perfuse the basal ganglia and internal capsule.
Ischemic Stroke of the Middle Cerebral Artery: Clinical Syndromes
An ischemic stroke occurs when reduced blood flow to a brain region leads to infarction. The primary mechanisms involve thrombotic or embolic infarcts.
Thrombotic strokes result from local clot formation at atherosclerotic plaques, leading to vessel occlusion. Embolic strokes occur when a detached mass, typically a dislodged thrombus, gas, or fat, travels through the bloodstream and becomes lodged in the MCA. Arterial occlusion disrupts oxygenated blood flow to the brain parenchyma, leading to ischemia, cerebral edema, and tissue necrosis.
The severity of ischemic stroke depends on the occlusion location. Proximal occlusions affect a larger brain volume and cause more extensive damage, while distal occlusions involve smaller territories and result in milder clinical presentations.
Thrombus formation is commonly associated with sites proximal to the MCA, such as plaques in the ICA and CCA, as well as atrial fibrillation, which increases the risk of embolism. Additionally, cardiac defects, such as atrial or ventricular septal defects, may lead to paradoxical embolism, which occurs when a thrombus bypasses the pulmonary circulation through an intracardiac shunt and enters systemic circulation, potentially causing MCA occlusion.
An ischemic stroke involving the MCA is one of the most common and clinically significant large-vessel strokes. The clinical presentation varies depending on the affected branches, whether the superior division, inferior division, or deep perforators.
The Alberta Stroke Programme Early CT Score (ASPECTS) is a quantitative tool used to predict poor recovery despite thrombolytic treatment. The MCA territory is scored out of 10, with 1 point deducted for each region showing early ischemic changes, such as swelling or hypoattenuation. A normal CT scan scores 10, while a score of 0 indicates widespread ischemia across the MCA territory.[10]
Superior division infarcts
Infarction of the superior division of the MCA affects the lateral frontal lobe, including the peri-Rolandic cortex. In the dominant hemisphere, this insult produces Broca aphasia, characterized by nonfluent speech with preserved comprehension. Motor deficits, including contralateral weakness of the face and upper limb, are common due to motor cortex involvement. Sensory deficits in the same regions may also occur but are typically milder than motor impairments. In the nondominant hemisphere, superior division infarcts may cause contralateral hemineglect, presenting as a lack of awareness of the left visual and somatosensory fields.
Inferior division infarcts
Inferior division strokes primarily affect the lateral temporal lobe and parts of the parietal lobe. In the dominant hemisphere, these insults lead to Wernicke aphasia, characterized by fluent but nonsensical speech with impaired comprehension. Contralateral homonymous hemianopia is frequently observed due to the involvement of the visual pathways. Motor deficits are generally absent in isolated inferior division strokes. In the nondominant hemisphere, hemineglect and apraxia are common clinical findings.
Deep territory infarcts
Deep territory infarcts involve the lenticulostriate arteries, which supply the internal capsule and basal ganglia. These strokes often cause pure motor hemiparesis due to disruption of the corticospinal and corticobulbar tracts within the posterior limb of the internal capsule. Infarcts affecting the thalamus or basal ganglia may result in ataxia or hemiballismus, respectively. Lipohyalinosis is thought to play a significant role in lenticulostriate infarcts, but further evidence is needed to support this association.[11]
Middle cerebral artery stem occlusion
Proximal occlusion of the MCA stem, affecting the superior, inferior, and deep territories, produces severe neurological deficits. Common features include contralateral hemiplegia, hemianesthesia, and homonymous hemianopia. Dominant hemisphere strokes cause global aphasia, while nondominant hemisphere strokes lead to severe hemineglect and gaze preference toward the side of the lesion. These extensive infarcts are associated with significant morbidity and often result in life-threatening cerebral edema.
Aneurysms of the Middle Cerebral Artery
MCA aneurysms are among the most common intracranial aneurysms. Risk factors include autosomal dominant polycystic kidney disease, Ehlers-Danlos syndrome, cigarette smoking, hypertension, and advanced age. These factors increase the likelihood of rupture and intracerebral hemorrhage. As discussed in the Surgical Considerations section, saccular aneurysms at high risk for rupture are managed with surgical clipping or endovascular embolization.
The MCA’s complex branching pattern, particularly at bifurcations and trifurcations, predisposes it to aneurysm formation. These aneurysms are typically saccular and develop from hemodynamic stress at vascular branching points.
Rupture typically presents with a sudden, severe headache—often described as the "worst headache of my life"—due to subarachnoid hemorrhage. Additional symptoms may include altered consciousness, nausea, and neck stiffness from meningeal irritation. Larger aneurysms may cause compressive symptoms, including contralateral motor deficits and cranial neuropathies, even without rupture.
Bifurcation and trifurcation aneurysms
MCA aneurysms commonly occur at the bifurcation of the M1 segment into superior and inferior trunks. Trifurcation aneurysms, though less common, present similarly and may involve more complex surgical considerations due to their proximity to multiple vascular branches.
Early branch aneurysms
These aneurysms arise from early branches of the MCA, such as the frontal and temporal branches. Although often smaller, early branch aneurysms carry a high risk of rupture due to their thin-walled morphology and elevated local hemodynamic stress.[12]
Lenticulostriate artery aneurysms
Although rare, aneurysms of the lenticulostriate arteries have significant clinical implications. Their deep location and small caliber make them prone to rupture, leading to intracranial rather than subarachnoid hemorrhage. These hemorrhages frequently involve the basal ganglia and internal capsule, resulting in profound motor deficits, including hemiplegia and sensorimotor syndromes. Prerupture diagnosis is particularly challenging due to their deep location and small size on imaging.[13]
Large and giant aneurysms
Large or giant MCA aneurysms, often exceeding 15 to 25 mm in diameter, are associated with significant mass effects. Compression of adjacent brain structures may lead to progressive neurological deficits, such as hemiparesis or visual field defects. The management of these lesions poses unique challenges due to their size, incorporation of branch vessels, and the presence of intraluminal thrombi or calcifications, requiring careful surgical or endovascular intervention.
Arteriovenous Malformations
Arteriovenous malformations are congenital arteriovenous connections that can occur anywhere in the brain. These lesions most commonly present in young adults between ages 20 and 40 and carry an annual hemorrhage risk of 1% to 4%. Treatment options include conventional surgery, radiosurgery, and endovascular embolization.[14]
Other Issues
Large hemispheric infarctions, particularly malignant MCA strokes, are associated with high morbidity and mortality rates. These outcomes result not only from primary ischemic tissue damage but also from secondary brain injury due to space-occupying cerebral edema.
Malignant MCA infarctions typically involve a complete MCA territory infarction, leading to significant brain swelling and increased intracranial pressure. Several factors predict progression to a malignant MCA stroke, including a hyperdense MCA sign, an admission ASPECTS score of 7 or lower, and mass effect with a midline shift greater than 4 mm accompanied by neurological deterioration within 48 hours of symptom onset.[15][16][17] The extent of ischemic tissue correlates with the severity of cytotoxic edema, which contributes to mass effect and intracranial hypertension.
Management should focus on reducing intracranial hypertension and preventing secondary brain injury. Hyperosmolar therapy provides temporary relief, but early decompressive hemicraniectomy significantly lowers mortality, especially in patients younger than 60. However, outcomes vary widely, requiring individualized decision-making.
Media
(Click Image to Enlarge)

Middle Cerebral Artery Cortical Divisions. This illustration shows the parts of the cerebral cortex supplied by the middle cerebral artery. Labeled regions include the precentral, central, anterior and posterior parietal, angular, temporooccipital, anterior, middle, and posterior temporal, temporopolar, orbitofrontal cortex, and prefrontal areas.
Contributed by Konstantinos Margetis MD, PhD
(Click Image to Enlarge)
(Click Image to Enlarge)

Course of the Middle Cerebral Artery. This illustration shows the course of the middle cerebral artery (MCA) after it diverges from the internal carotid artery. MCA parts include M1 to M4 and the lenticulostriate arteries. The anterior cerebral artery is also shown.
Contributed by Konstantinos Margetis MD, PhD
(Click Image to Enlarge)
References
Shapiro M, Raz E, Nossek E, Chancellor B, Ishida K, Nelson PK. Neuroanatomy of the middle cerebral artery: implications for thrombectomy. Journal of neurointerventional surgery. 2020 Aug:12(8):768-773. doi: 10.1136/neurintsurg-2019-015782. Epub 2020 Feb 27 [PubMed PMID: 32107286]
Gibo H, Carver CC, Rhoton AL Jr, Lenkey C, Mitchell RJ. Microsurgical anatomy of the middle cerebral artery. Journal of neurosurgery. 1981 Feb:54(2):151-69 [PubMed PMID: 7452329]
Rey S, Semenza GL. Hypoxia-inducible factor-1-dependent mechanisms of vascularization and vascular remodelling. Cardiovascular research. 2010 May 1:86(2):236-42. doi: 10.1093/cvr/cvq045. Epub 2010 Feb 17 [PubMed PMID: 20164116]
Raybaud C, Normal and abnormal embryology and development of the intracranial vascular system. Neurosurgery clinics of North America. 2010 Jul; [PubMed PMID: 20561492]
Level 3 (low-level) evidenceKlostranec JM, Krings T. Cerebral neurovascular embryology, anatomic variations, and congenital brain arteriovenous lesions. Journal of neurointerventional surgery. 2022 Sep:14(9):910-919. doi: 10.1136/neurintsurg-2021-018607. Epub 2022 Feb 15 [PubMed PMID: 35169032]
Okahara M, Kiyosue H, Mori H, Tanoue S, Sainou M, Nagatomi H. Anatomic variations of the cerebral arteries and their embryology: a pictorial review. European radiology. 2002 Oct:12(10):2548-61 [PubMed PMID: 12271398]
Kashtiara A, Beldé S, Schollaert J, Menovsky T. Anatomical Variations and Anomalies of the Middle Cerebral Artery. World neurosurgery. 2024 Mar:183():e187-e200. doi: 10.1016/j.wneu.2023.12.052. Epub 2023 Dec 14 [PubMed PMID: 38101539]
Brzegowy P, Polak J, Wnuk J, Łasocha B, Walocha J, Popiela TJ. Middle cerebral artery anatomical variations and aneurysms: a retrospective study based on computed tomography angiography findings. Folia morphologica. 2018:77(3):434-440. doi: 10.5603/FM.a2017.0112. Epub 2017 Dec 13 [PubMed PMID: 29235088]
Level 2 (mid-level) evidenceHeros RC. Microneurosurgical management of anterior choroidal artery aneurysms. World neurosurgery. 2010 May:73(5):459-60. doi: 10.1016/j.wneu.2010.03.013. Epub [PubMed PMID: 20920922]
Barber PA, Demchuk AM, Zhang J, Buchan AM. Validity and reliability of a quantitative computed tomography score in predicting outcome of hyperacute stroke before thrombolytic therapy. ASPECTS Study Group. Alberta Stroke Programme Early CT Score. Lancet (London, England). 2000 May 13:355(9216):1670-4 [PubMed PMID: 10905241]
Decavel P, Vuillier F, Moulin T. Lenticulostriate infarction. Frontiers of neurology and neuroscience. 2012:30():115-9. doi: 10.1159/000333606. Epub 2012 Feb 14 [PubMed PMID: 22377876]
Ulm AJ, Fautheree GL, Tanriover N, Russo A, Albanese E, Rhoton AL Jr, Mericle RA, Lewis SB. Microsurgical and angiographic anatomy of middle cerebral artery aneurysms: prevalence and significance of early branch aneurysms. Neurosurgery. 2008 May:62(5 Suppl 2):ONS344-52; discussion ONS352-3. doi: 10.1227/01.neu.0000326018.22434.ed. Epub [PubMed PMID: 18596514]
Lastilla M. Lacunar infarct. Clinical and experimental hypertension (New York, N.Y. : 1993). 2006 Apr-May:28(3-4):205-15 [PubMed PMID: 16833026]
Mills MT, Zafar A, Choudhari KA, Smith A, Coley S, Jankowski S, Randall M, Patel UJ. Management of Concomitant Moyamoya Disease, Arterial Venous Malformation, and Intracranial Aneurysm: Case Illustration, Literature Review, and Management Algorithm. World neurosurgery. 2018 Nov:119():262-266. doi: 10.1016/j.wneu.2018.08.017. Epub 2018 Aug 11 [PubMed PMID: 30107248]
Level 3 (low-level) evidenceMuscari A, Faccioli L, Lega MV, Lorusso A, Trossello MP, Puddu GM, Spinardi L, Zoli M. Predicting cerebral edema in ischemic stroke patients. Neurological sciences : official journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology. 2019 Apr:40(4):745-752. doi: 10.1007/s10072-019-3717-y. Epub 2019 Jan 19 [PubMed PMID: 30659418]
Pullicino PM, Alexandrov AV, Shelton JA, Alexandrova NA, Smurawska LT, Norris JW. Mass effect and death from severe acute stroke. Neurology. 1997 Oct:49(4):1090-5 [PubMed PMID: 9339695]
MacCallum C, Churilov L, Mitchell P, Dowling R, Yan B. Low Alberta Stroke Program Early CT score (ASPECTS) associated with malignant middle cerebral artery infarction. Cerebrovascular diseases (Basel, Switzerland). 2014:38(1):39-45. doi: 10.1159/000363619. Epub 2014 Sep 16 [PubMed PMID: 25228461]