Introduction
Colorectal cancer (CRC) is the third most commonly diagnosed cancer and the third leading cause of cancer-related deaths in the United States. CRC is the leading cause of cancer deaths in men aged 50 or younger and the second leading cause in women of the same age group, following breast cancer. CRC is almost always preceded by adenomatous or serrated colon polyps. About 65% of CRC cases are sporadic, 25% are associated with a family history of CRC or adenomatous polyps in first-degree relatives, and the remaining 10% are attributed to hereditary (genetic) CRC syndromes.[1]
The risk of CRC is approximately twice as high in individuals with a family history of CRC compared to those without such a history.[2] Hereditary CRC syndromes result from specific genetic mutations that increase susceptibility to CRC, as well as other malignancies and benign conditions. Familial adenomatous polyposis (FAP) and Lynch syndrome, also referred to as hereditary nonpolyposis CRC (HNPCC), are the most prevalent hereditary CRC syndromes. These conditions are generally more aggressive, have a poorer prognosis, and may be less responsive to chemotherapy compared to sporadic CRC.
A genetic or hereditary cause of CRC should be suspected and investigated if one or more of the following factors are present:
- A significant family history of CRC or premalignant colon polyps in 2 or more first-degree relatives of the proband.
- A family history of other cancers known to be associated with hereditary CRC, particularly endometrial cancer.
- Onset of CRC at an unusually early age.
- Multiple primary cancers in an individual, including CRC.[3]
Etiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Etiology
Hereditary CRC syndromes result from mutations in various genes, which significantly increase the risk of developing CRC and other malignancies. Some of these syndromes are caused by dominant traits, inherited from just one parent, whereas others are recessive and require mutated alleles from both parents. Hereditary CRC syndromes can be categorized into high-penetrance and moderate-penetrance genetic variants, as mentioned below.[4]
High-Penetrance Genetic Syndromes
Lynch syndrome: This condition is also known as HNPCC and is the most common hereditary CRC syndrome, which follows an autosomal dominant inheritance pattern. This condition is primarily caused by mutations in mismatch repair (MMR) genes, listed in descending order of prevalence—PMS2, MLH1, and MutS homologs 6 (MSH6) and 2 (MSH2).[5] CRCs in individuals with Lynch syndrome are often multiple and may occur synchronously or metachronously. Among these genes, MLH1 and MSH2 are associated with the highest CRC risk, while individuals with MSH6 and PMS2 mutations have a lower risk and tend to develop Lynch syndrome–related cancers later in life.[6] Additionally, a deletion in the epithelial cellular adhesion molecule (EPCAM) gene can result in epigenetic silencing of MSH2 in tissues expressing EPCAM, leading to a loss of MSH2 expression.[7][8]
When CRC is tested for MMR mutations by immunohistochemistry (IHC; refer to the Pathophysiology section) and shows loss of MLH1 or PMS2 staining, further testing for a BRAF V600E mutation is performed. If the BRAF V600E mutation is positive, the CRC is classified as sporadic rather than Lynch syndrome and no further evaluation for an inherited CRC diagnosis is required. The same conclusion applies if BRAF mutation testing is negative but MLH1 promoter methylation testing is positive. In contrast, CRCs associated with Lynch syndrome tend to develop earlier, typically in the mid-40s, are more likely to originate on the right side of the colon and exhibit more aggressive behavior than sporadic lesions.
Lynch syndrome significantly increases the risk of developing various cancers, including the below-mentioned malignancies.
- Colorectal cancer: Approximately 80% lifetime risk.
- Endometrial cancer: Approximately 40% lifetime risk.
- Ovarian adenocarcinoma.
- Gastric adenocarcinoma.
- Small bowel adenocarcinoma.
- Pancreaticobiliary adenocarcinomas.
- Upper urinary tract transitional cell cancers: Particularly associated with MSH2 mutations.
- Prostate cancer.
- Skin lesions: Associated with Muir-Torre syndrome, particularly in individuals with MSH2 mutations.
- Brain cancers: Associated with Turcot syndrome type 1. Please see StatPearls' companion resource, "Turcot Syndrome," for more information.
- Muir-Torre syndrome: Lynch syndrome is associated with an increased risk of multiple cancers, including those seen in Muir-Torre syndrome. This subtype of Lynch syndrome involves skin neoplasms such as sebaceous adenomas, sebaceous carcinomas, and keratoacanthomas. Muir-Torre syndrome is most commonly linked to mutations in the MSH2 gene and has also been reported in individuals with mutations in the MLH1, MSH6, and PMS2 genes. The likelihood of Muir-Torre syndrome is higher in patients with CRC and skin neoplasms if the onset occurs earlier in life, there is a large number of skin lesions, or there is a personal or family history of Lynch-associated cancers.[9]
- Turcot syndrome type 1: This condition is associated with CRC, lipomas, cafe-au-lait spots on the skin, basal cell skin cancers, and various brain and spinal cord tumors. Turcot syndrome type 1 is an autosomal recessive trait usually due to mutations of the MLH1 and PMS2 genes. This subtype accounts for approximately 20% to 25% of all Turcot syndrome cases.[10] Please see StatPearls' companion resource, "Turcot Syndrome," for more information.
Adenomatous polyposis syndromes: This syndrome is characterized by hundreds to thousands of adenomas and should be considered in patients with the following criteria:
- More than 10 adenomas detected during a single colonoscopy.
- More than 20 adenomas identified throughout their lifetime.
- Onset of CRC in individuals aged 50 or younger.
- Presence of extracolonic manifestations associated with any of the polyposis syndromes (eg, Gardner syndrome mentioned below).
- A family history of FAP or MUTYH-associated polyposis (MAP) in probands.
Familial adenomatous polyposis,[11] which is associated with mutations in the adenomatous polyposis coli (APC) gene, significantly increases the risk of several cancers and conditions, as mentioned below.
- Colorectal cancer: Patients typically develop hundreds to thousands of adenomatous colorectal polyps. Without a subtotal or total colectomy, nearly 90% of affected individuals will develop CRC by age 45.[12]
- Gastric and duodenal adenomatous polyps and adenocarcinomas: Duodenal polyposis severity is classified using the Spigelman staging system (stages 0-IV).[13]
- Hepatoblastomas
- Thyroid cancer
- Desmoid tumors
- Pancreaticobiliary cancers
- Adrenal gland adenocarcinomas
- Brain tumors
- Congenital hypertrophy of the retinal pigment epithelium
Attenuated familial adenomatous polyposis: Attenuated FAP (AFAP) is an autosomal dominant inherited condition characterized by fewer polyps and a lower risk of CRC compared to classic FAP.[14] This is caused by a mutation in the APC I1307K gene, most commonly found in individuals of Ashkenazi Jewish descent, leading to an approximately 70% lifetime risk of CRC.[15] Non-Jewish White individuals carrying the APC I1307K mutation face an increased risk of melanoma and cancers of the stomach, small intestine, liver, thyroid, breast, and prostate.[16]
Among Ashkenazi Jewish individuals in the United States, the APC c.3920T>A (p.I1307K) mutation is found in 11.5% of individuals with CRC compared to 7.2% without CRC.[17] This condition typically manifests later in life than classic FAP and is diagnosed when fewer than 100 adenomas are identified in the colon at presentation. While mutations in the APC gene cause both conditions, the specific mutations in AFAP differ from those found in classic FAP cases.[2][3][9][10]
The median age for CRC diagnosis in individuals with AFAP is between 55 and 58, which is approximately 10 to 20 years earlier than in typical FAP. However, cases have been reported in individuals aged between 29 and 81.[18] Regular colonoscopy and polypectomy can delay the need for colectomy in these patients, who may also benefit from aspirin chemoprophylaxis.[19]
Gardner syndrome: Gardner syndrome, associated with mutations in the APC gene, presents with distinct clinical features, as outlined below.
- Hundreds to thousands of adenomas and cancers of the colorectum and small intestine.
- Benign lesions of the skin (pilomatrixoma and epidermal inclusion cysts), soft tissue (desmoid tumors, lipomas, and fibromatosis), and bones (osteomas).
- Supernumerary teeth
- Adrenal adenomas
- Congenital hypertrophy of the retinal pigment epithelium.
Total or subtotal colectomy is typically required for patients with Gardner syndrome who develop CRC or have too many adenomas to manage endoscopically. However, desmoid tumors of the mesentery and abdominal wall may still occur after colectomy.[20] Please see StatPearls' companion resource, "Gardner Syndrome," for more information.
Turcot syndrome type 2: Turcot syndrome type 2 is an autosomal dominant condition associated with mutations in the APC gene and is distinct from a variant of FAP.[21] In addition to CRC, this trait predisposes individuals to CHRPE, spinal cord tumors, and medulloblastomas of the brain.[22]
MUTYH-associated polyposis: MAP is caused by biallelic variants of the MUTYH gene and is characterized by the presence of 10 to a few 100 colonic adenomas (average of 50 polyps). However, CRC can also develop in some individuals without polyposis. The average age of CRC onset is 46. First described in 2002, MAP is inherited in an autosomal recessive pattern and is considered an attenuated polyposis syndrome.[23]
Polyps may include serrated adenomas, hyperplastic or sessile serrated polyps, and mixed hyperplastic-adenomatous polyps. MAP is also associated with malignancies of the small bowel, ovary, and bladder, and there may be an increased risk for breast and endometrial cancer. Benign conditions associated with MAP include thyroid nodules, benign adrenal lesions, jawbone cysts, and CHRPE.[24] MAP shares many clinical features of FAP and AFAP, necessitating genetic testing for differentiation in many instances.
MAP should be suspected in individuals with the following clinical findings and family history:
- A lifetime history of at least 10 colorectal adenomas in individuals aged 60 or younger.
- A lifetime history of at least 20 colorectal adenomas in individuals of any age.
- A lifetime history of at least 20 or more colorectal polyps, including adenomas, hyperplastic polyps (proximal to the sigmoid colon), and sessile serrated polyps.[25]
Oligopolyposis syndromes (with 10-100 polyps): The POLE exonuclease mutation is a rare genetic alteration that causes polymerase proofreading-associated polyposis (PPAP) syndrome. This syndrome increases the risk of developing colorectal, gastric, and small bowel polyps, which in turn heightens the likelihood of CRC and small bowel cancers. Additional risks include brain tumors, endometrial cancer, and ovarian cancer. These mutations are typically somatic rather than hereditary, and CRCs associated with PPAP are always MSI-S.[26]
While the POLE mutation typically occurs de novo, children of affected individuals have a 50% chance of inheriting it. A study involving 47 POLE mutation carriers from 20 families reported an average of 19 colonic adenomas (range 1–68). Among these carriers, 81.8% had at least 2 adenomas, and 73.9% had 5 or more. CRC was diagnosed in 63.8% of carriers, with the average age at first diagnosis being 40.7. Additionally, 50% of carriers developed duodenal adenomas, and 6.4% were diagnosed with brain tumors at an average age of 30.6.[25]
Clinical and colonoscopic findings include:
- Attenuated adenomatous polyposis (20-100 adenomas).
- Amsterdam I criteria (for CRC only).
- CRC and 5 to 20 adenomas, both diagnosed in individuals before age 50.
- CRC or 5 to 20 adenomas, with a first-degree relative diagnosed with CRC before age 50.
- CRC or 5 to 20 adenomas, with at least 2 first- or second-degree relatives with CRC, diagnosed at any age.[27]
POLD1 gene mutation: A study of 22 carriers across 8 families revealed that over 55% of POLD1 mutation carriers had an average of 12.3 adenomas (range 0–45), with at least 2 adenomas in 63.6% and at least 5 adenomas in 55.5%. CRC was diagnosed at an average age of 35.9, whereas endometrial cancer was identified in 57.1% of female carriers at a mean age of 51.4.[27]
Clinical and colonoscopic findings include:
- Attenuated adenomatous polyposis (20–100 adenomas).
- Amsterdam II criteria (specific for CRC and endometrial cancer only).
- CRC diagnosed before age 50 or endometrial cancer diagnosed before age 60, with 5 to 20 adenomas detected before age 50.
- CRC, endometrial cancer, or 5 to 20 adenomas, along with a first-degree relative with CRC diagnosed before age 50 or endometrial cancer diagnosed before age 60.
- CRC, endometrial cancer, or 5 to 20 adenomas, accompanied by at least 2 first- or second-degree relatives with CRC or endometrial cancer at any age.
Note: The presence of brain tumors and/or breast cancer in the context of CRC, polyposis, and/or endometrial cancer may also suggest PPAP syndrome, which is more commonly associated with POLE mutations.
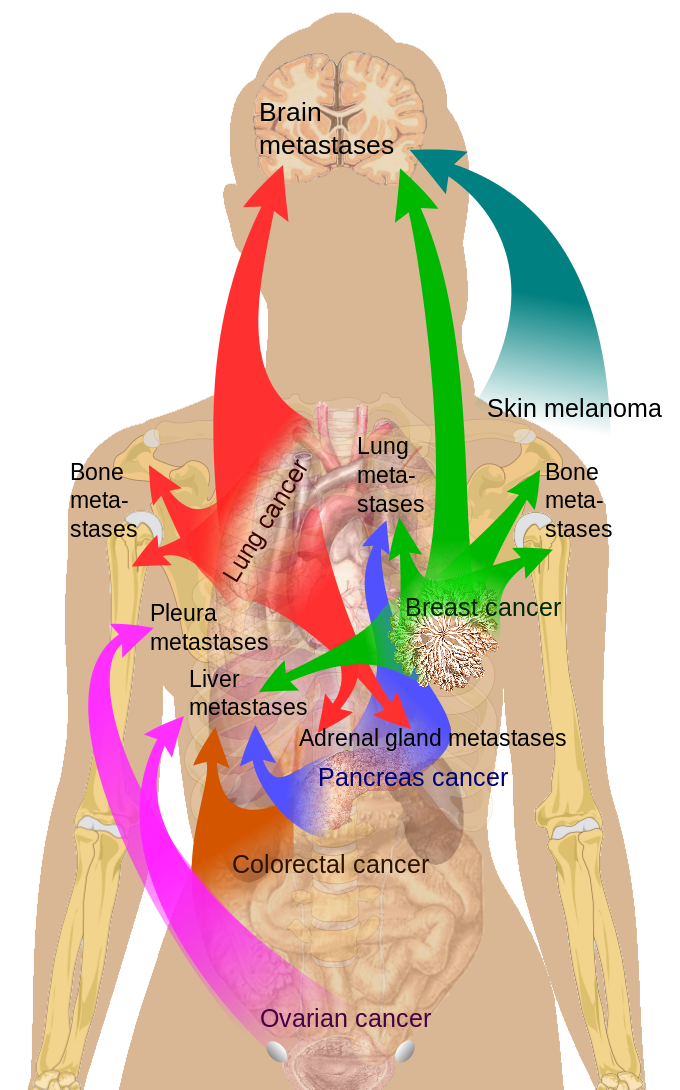
GREM1 gene mutation: This mutation may lead to hereditary mixed polyposis syndrome (HMPS), which is characterized by the development of numerous adenomatous, hyperplastic, hamartomatous, and juvenile polyps at a young age. HMPS increases the lifetime risk of developing CRC to 10% to 25%, nearly double the risk associated with sporadic CRC. This also raises the risk of melanoma, uterine sarcomas, and cancers of the lung, bladder, breast, and melanoma (see Image. Common Sites of Breast Cancer Metastasis).[28]
Sessile serrated polyposis syndrome: Previously known as hyperplastic polyposis syndrome, serrated polyposis syndrome is the most common colorectal polyposis syndrome characterized by the accumulation of serrated polyps and adenomas, which are associated with an elevated risk of CRC.[29]
The diagnosis is based on the total lifetime count of hyperplastic polyps, traditional serrated adenomas, and sessile serrated polyps in patients meeting 1 of the 2 World Health Organization (WHO) 2019 criteria:
- Five serrated polyps proximal to the rectum, all at least 5 mm in size, with 2 measuring at least 10 mm.
- Twenty or more serrated polyps of any size throughout the colon, with five proximal to the rectum.[30]
Duodenal polyps or cancer may also occur in serrated polyposis syndrome. While a family history of CRC or polyps consistent with autosomal recessive inheritance has been noted,[31] no specific genetic abnormality has been identified. Enhanced colonoscopic screening and surveillance are essential due to the potential number of polyps and the increased cancer risk.[32]
Surveillance recommendations for individuals with a first-degree relative diagnosed with serrated polyposis include a baseline colonoscopy at the earliest of the following—age 40, the age at which the youngest FDR was diagnosed with serrated polyposis without cancer, or 10 years before the earliest CRC diagnosis in the family associated with serrated polyposis. Following the baseline exam, colonoscopy should be repeated every 5 years if no polyps are found or every 1 to 3 years if proximal serrated polyps or multiple adenomas are detected.
Juvenile polyposis coli results from germline mutations in the BMPR1A and SMAD4 genes. The most common phenotype includes multiple gastrointestinal hamartomatous polyps and an increased risk of gastric, small bowel, and CRCs, inherited in an autosomal dominant pattern. Less commonly, attenuated adenomatous polyposis may occur, with at least 10 cumulative colorectal adenomas or CRC without hamartomatous polyps. These mutations are typically identified through multigene panel testing (MGPT) for hereditary cancer-related genes in cases of unexplained polyposis.[33][34] Mutations are more frequently de novo rather than inherited.[35]
Screening typically begins in childhood, with earlier endoscopic evaluation warranted if symptoms arise (refer to the Table in the Evaluation section). The median age of onset for CRC is 36, whereas gastric cancer has a median onset of 41.[34] Gastric lesions and gastrointestinal cancers occur more frequently with pathogenic variants of the SAMAD4 gene compared to those of the BMPR1A gene. SMAD4 mutations may also be associated with hereditary hemorrhagic telangiectasia,[36] mitral valve disease, and aortic dilatation, which may result in aortic aneurysms or dissection.
Gastrointestinal diagnostic features of juvenile polyposis syndrome include one or more of the following:
- At least 5 juvenile polyps in the colorectum.
- Juvenile polyps located elsewhere in the gastrointestinal tract.
- Any number of juvenile polyps accompanied by at least one affected family member.
Peutz-Jeghers syndrome: Peutz-Jeghers syndrome, caused by mutations in the STK11 gene, is a rare disorder characterized by mucocutaneous pigmentation and hamartomatous polyposis throughout the gastrointestinal tract, excluding the esophagus, with the highest frequency in the duodenum and proximal jejunum. Germline variants in the STK11 gene are inherited in an autosomal dominant pattern with universal penetrance. Approximately half of newly diagnosed patients inherit the mutation from a parent, while the rest result from de novo mutations in one copy of the gene. Gastrointestinal lesions may manifest in childhood, and serious complications can significantly affect the quality of life.
Hamartomatous polyps in the small bowel are usually diagnosed with small-bowel capsule endoscopy or double-balloon enteroscopy. Double-balloon enteroscopy also enables therapeutic interventions, such as polypectomy. These lesions may lead to several complications, such as bleeding, obstruction, and intussusception. The risk of colorectal, gastric, small bowel, and pancreatic cancers is elevated,[37][38] with the incidence of cancer rising significantly after age 50.[39] Screening for pancreatic cancer using endoscopic ultrasound or magnetic resonance imaging (MRI) or magnetic resonance cholangiopancreatography is recommended in adults.[40]
The diagnosis is established based on the following criteria:[41]
- Mucocutaneous pigmentations: Pigmented macules measuring 1 to 5 mm on the lips, oral mucosa, and fingertips.
- Endoscopic findings: Upper endoscopy, colonoscopy, and small bowel endoscopy show hamartomatous polyps throughout the gastrointestinal tract, excluding the esophagus.
- Pathological findings: Hamartomatous polyps exhibit mucosal hyperplasia and dendritic growth of smooth muscle fiber bundles within the muscularis mucosae.
- Differential diagnoses: Conditions to consider include FAP, juvenile polyposis syndrome, Cowden syndrome or PTEN (phosphatase and tensin homolog) hamartoma tumor syndrome (PHTS), tuberous sclerosis, inflammatory polyposis, serrated polyposis syndrome, Cronkhite-Canada syndrome, HMPS, and Laugier-Hunziker-Baran syndrome.
- Genetic testing: Identification of germline pathogenic variants in the STK11 gene.
Peutz-Jeghers syndrome is classified into the following diagnostic categories:
- Presence of mucocutaneous pigmentations with typical endoscopic and pathological findings after excluding the differential diagnoses listed above.
- Mucocutaneous pigmentations in an individual with a family history of Peutz-Jeghers syndrome, excluding the differential diagnoses listed above.
- Typical endoscopic and pathological findings in an individual with a family history of Peutz-Jeghers syndrome in a close relative after ruling out the differential diagnoses listed above.
- Typical endoscopic findings with corresponding pathology in multiple lesions, excluding the differential diagnoses listed above.
- Identification of a pathogenic mutation in the STK11 gene.
PHTS is caused by mutations in the PTEN gene.[42][43] This multisystem condition includes Cowden syndrome, Bannayan-Riley-Ruvalcaba syndrome, and Proteus-like syndrome. Affected individuals may develop several to numerous colorectal polyps, which can include a combination of hamartomas, adenomas, ganglioneuromas, serrated polyps, juvenile polyps, inflammatory polyps, and lymphoid aggregates. Upper gastrointestinal polyps with similar histologies are common among these patients; however, esophageal and gastric cancers are rare.[44]
Individuals with PTEN mutations face a 2- to 3-fold increased lifetime risk of CRC, along with an elevated risk of breast, thyroid, and renal cancers. These mutations may also contribute to a higher likelihood of endometrial cancer and melanoma.[45] Additionally, PTEN mutations have been linked to various benign conditions, including macrocephaly, Hashimoto's thyroiditis, mucocutaneous hamartomas, vascular malformations, and neurodevelopmental disorders such as autism.[46] An increased risk of immunological dysregulation, including autoimmunity and immune deficiencies,[43] has also been associated with these mutations.
Non-colorectal cancer–associated syndromes and cancer risks: A smaller fraction of hereditary cancer cases is attributable to non-CRC–associated syndromes, which carry a lower risk of CRC. Mutations in BRCA1, BRCA2, and PALB2 genes significantly increase the risk of breast and ovarian cancer. Although BRCA2 mutation carriers may have a slightly higher incidence of CRC, a definitive cause-and-effect relationship has not been established. The incidence of CRC in BRCA1 mutation carriers does not exceed that of the wild type. However, BRCA2 mutation carriers show stronger evidence of increased risk for pancreatic ductal adenocarcinoma, with possible involvement in BRCA1.[47] PALB2, a partner gene of BRCA2 involved in DNA repair, has been identified as a marker for shorter disease-free and overall survival (OS) in CRC patients.[48]
Li-Fraumeni syndrome: Mutations in the TP53 tumor suppressor gene significantly increase the risk of multiple cancers in children and young adults, with a 50% risk by age 40 and 80%–90% by age 60. Colonoscopic screening begins in young adults, with a median age of CRC onset being 33 to 38 (refer to the Table in the Evaluation section).[49][50] Patients with Li-Fraumeni syndrome frequently develop more than one type of cancer, which are listed below.
- Colorectal cancer: Colonoscopic screening begins when the CRC risk reaches or exceeds 0.6% (refer to the Table in the Evaluation section).
- Pancreatic ductal adenocarcinoma: Lifetime risk is increased 7-fold, with 50% diagnosed by age 30 and up to 90% by age 60.
- Anaplastic rhabdomyosarcoma
- Breast cancer: Females have a significantly increased risk, with nearly 100% lifetime risk of developing cancer.
- Brain tumors: Includes sonic hedgehog medulloblastoma, choroid plexus carcinoma, glioma, and neuroblastoma.
- Adrenocortical carcinoma
- Hypodiploid acute lymphoblastic leukemia
- Melanoma
- Lung adenocarcinoma
- Kidney cancer
- Thyroid cancer
- Gonadal germ cell tumors: This includes ovarian, testicular, and prostate cancers.
Gastric adenocarcinoma and proximal polyposis of the stomach: Gastric adenocarcinoma and proximal polyposis of the stomach (GAPPS) is a rare autosomal dominant condition caused by germline pathogenic variants in the APC gene's promoter 1B region. It is primarily associated with gastric body fundic gland polyps and gastric adenocarcinoma. Large deletions in the promoter 1B region can result in a FAP phenotype. GAPPS shows an inverse relationship with Helicobacter pylori infection.[51][52]
Cystic fibrosis: Cystic fibrosis (CF) is a recessive genetic disorder caused by mutations in the CF transmembrane conductance regulator (CFTR) gene. Patients with CF have a 5-fold increased risk of CRC, with a smaller increase in CF carriers. The risk is further elevated in individuals with severe CFTR genotypes, reaching up to a 20-fold increase after solid organ transplants, particularly lung transplants, when immunosuppressive therapy is used. In addition, well-recognized increased risks of esophageal, small bowel, hepatobiliary, and pancreatic cancers exist. As life expectancy for CF patients improves with advances in medical care, the risk of developing cancer continues to rise.[53][54][55]
Moderate-Penetrance Genetic Syndromes
Monoallelic mutations in the MUTYH gene: Autosomal dominant inheritance of a single allele from one parent may slightly increase the risk of developing CRC. However, no specific guidelines recommend more frequent screening compared to average-risk individuals. Some studies have suggested an increased risk of gastric, liver, and endometrial cancers, while there is no clear evidence of an elevated risk for breast or liver cancer.[56]
Checkpoint kinase 2 gene: CHEK2 is a tumor suppressor gene that affects MMR and has been linked with Li-Fraumeni syndrome. Mutations in CHEK2 minimally increase the risk of CRC while significantly raising the risk of breast and prostate cancers, brain tumors, and sarcomas.[57]
ATM is a gene involved in DNA repair, and mutations are commonly observed in CRC, especially in patients with Lynch syndrome. Promoter hypermethylation has been identified in 40% of CRCs and adenomas in genes such as APC and ATM. These mutations also increase the risk of stomach, lung, and breast cancers.[58]
Epidemiology
CRC is the third most commonly diagnosed cancer and the third leading cause of cancer deaths in the United States. CRC is the leading cause of cancer deaths in men aged 50 or younger and the second leading cause in women of the same age group. In 2024, an estimated 46,200 rectal cancers and 106,590 other colon cancers will be diagnosed, with approximately 53,010 CRC-related deaths.[59]
The incidence and mortality rates from CRC have steadily decreased in the United States during the 21st century,[60] although this reduction is primarily seen in individuals aged 65 and older. Rates remain stable in individuals aged 55 to 64, while the incidence and death rates have increased by 1% per year in individuals aged 55 or younger since the mid-1990s. Overall mortality rates have decreased from 29.2 per 100,000 individuals in 1970 to 12.8 per 100,000 individuals in 2021. This reduction is attributed to a lower incidence, earlier detection through screening, and advancements in cancer treatment modalities.[59]
Hereditary CRC syndromes are identified in 6% to 10% of all CRC cases diagnosed before age 50, with an incidence of approximately 2 per 100,000 individuals worldwide.[1][61] Lynch syndrome is the most common hereditary colon cancer, accounting for 3% of newly diagnosed CRC cases in the United States [4] and 2% to 3% of endometrial cancers.[62] FAP accounts for less than 1% of CRC cases, with other hereditary causes of CRC being even rarer.[63]
Pathophysiology
CRC develops through the adenoma-carcinoma sequence, initially described by Fearon and Vogelstein in 1990.[64] This process follows 2 major pathogenic pathways, driven by the accumulation of multiple mutations. The first pathway involves genetic defects in DNA MMR genes, evaluated using microsatellite instability (MSI) testing and IHC. The second pathway involves APC/β-catenin alterations characterized by chromosomal instability, leading to the stepwise accumulation of mutations in various oncogenes and tumor suppressor genes. The molecular evolution of this pathway aligns with Knudson's "2-hit" theory, proposed in 1971 to explain the development of retinoblastomas.[65]
The loss of the APC tumor suppressor gene is the first event in adenoma formation, requiring the inactivation of both copies to stimulate adenoma development. Under normal conditions, the APC gene promotes the degradation of β-catenin. When APC is lost, β-catenin accumulates, translocates to the nucleus, and activates the transcription of several genes, including MYC and cyclin D1. This is followed by KRAS mutations, mutations in a putative tumor suppressor gene on 18q21, and alterations in TP53, ultimately leading to cancer. Additional mutations further contribute to progression. While the sequence of mutations is less critical, their cumulative effect is essential for colorectal carcinogenesis. This adenoma-carcinoma sequence accounts for approximately 80% of sporadic CRC cases.[66]
Mutational Mechanisms Associated With Colorectal Cancer
Genomic instability: This is a hallmark of CRCs and encompasses the below-mentioned mechanisms, driving tumorigenesis through the accumulation of DNA repair defects and mutations.
- Microsatellite instability: MSI testing is used to detect defects in the MMR genes, often indicating Lynch syndrome. Tumor biopsies from CRC and endometrial cancers are classified as MSI-high (MSI-H), MSI-low (MSI-L), and microsatellite stable (MSI-S). MSI-H is present in over 90% of Lynch syndrome tumors and 10% to 15% of sporadic CRCs, often due to MLH1 promoter methylation.[67] MSI arises from errors in the DNA repair system, primarily a failure in base complementarity, leading to the expansion of short tandem repeats (microsatellites) and increased mutations. The MMR system comprises 7 genes—hMLH1, hMLH3, hMSH2, hMSH3, hMSH6, hPMS1, and hPMS2. Currently, more than 500 different mutations have been identified.[68] The cumulative incidence of CRC by age 75 is 46% for MLH1 carriers, 43% for MSH2 carriers, and 15% for MSH6 carriers.[69] MSI testing is carried out vis Polymerase Chain Reaction (PCR) technology. IHC is an alternative to MSI testing for CRC or endometrial cancer tissue screening. Similar to that seen with an MSI-H reading, more than 90% of Lynch syndrome tumors will show a lack of expression of at least one of the MMR proteins by IHC testing, and concordance with MSI testing is 99.1%. MSI is slightly more sensitive in the setting of Lynch syndrome, but a larger amount of tissue is necessary to do MSI testing, so IHC testing is performed in this case.[70]
- Chromosomal instability: This is the most common genetic alteration, present in 85% of sporadic CRC cases.[67] This mutation involves the loss of heterozygosity in tumor suppressor genes and mutations in proto-oncogenes,[71] including APC, TP53, KRAS, allelic loss of 18q, and aneuploidy. FAP progresses to cancer through this pathway.[72] Numerous mutations in the APC gene (a large "housekeeping" gene) have been identified, including microdeletions and frameshift mutations. Over 60% of these mutations are located at the 5′ end of the exon on the 5q21 chromosome.[73]
Epigenetic (DNA methylation): The CpG island methylator phenotype (CIMP) pathway is observed in 15% of sporadic CRC cases. While it impacts the expression of MMR genes, it remains distinct from Lynch syndrome.[74][75][66] Hypomethylation typically occurs in the advanced stages of tumor development and has a greater impact on the genome than hypermethylation.
CIMP is characterized by the methylation of tumor suppressor genes, with tumorigenesis potentially occurring through progressive genetic silencing without genetic mutations. Approximately 30% to 40% of proximal CRCs and 3% to 12% of distal CRCs are classified as CIMP.[76] CIMP is considered a distinct subgroup of CRC, fundamentally differing from other types of colon cancers. CIMP tumors display unique pathological features, including high mutation rates in KRAS or BRAF, a proximal location, wild-type p53, mucinous histology, poor differentiation, and a higher occurrence in female and older patients.[77]
History and Physical
History
The signs and symptoms of hereditary CRC are similar to those of sporadic CRC. Patients may present with symptoms such as bleeding, abdominal pain, weight loss, and changes in bowel habits, which can progress to intestinal obstruction if the cancer is advanced by the time of diagnosis. A comprehensive family history is crucial for diagnosing hereditary CRC. A thorough review of systems may also uncover other conditions, both cancerous and benign, associated with certain hereditary CRC syndromes.
Family history: Meeting the Amsterdam II or modified Bethesda criteria should prompt an evaluation for Lynch syndrome. A family history of CRC, other malignancies, or benign conditions associated with hereditary CRC syndromes should raise suspicion for these conditions.
- Amsterdam I criteria: These older criteria are similar to the Amsterdam II criteria but apply only to CRC and not other Lynch syndrome-associated cancers.
- Amsterdam II criteria: These criteria follow the "3-2-1" rule, as mentioned below.
- "Three" or more relatives affected by a neoplasm associated with HNPCC (CRC, cancer of the endometrium, small intestine, ureter, or renal pelvis), with 1 being a first-degree relative of the other 2.
- "Two" or more successive generations were affected.
- "One" or more relatives affected by CRC were diagnosed before age 50.
- Exclusion of FAP in cases of CRC.[78]
- Modified Bethesda criteria:
- Patients with CRC in families that meet the Amsterdam criteria.
- Patients with 2 neoplasms associated with HNPCC, including synchronous or metachronous CRC and extracolonic cancers (endometrium, ovary, gastric, hepatobiliary, small intestine, ureter, or renal pelvis).
- Patients with CRC and a first-degree relative with CRC, an extracolonic neoplasm associated with HNPCC, or colorectal adenoma; with one of the cancers diagnosed before age 50 and the adenoma before age 40.
- Patients with CRC or endometrial cancer were diagnosed before age 50.
- Patients with CRC in the right colon, histologically undifferentiated, and diagnosed before age 50.
- Patients with chemokine receptor (CCR)-type cells in the signet ring (comprising more than 50% of signet ring cells) and diagnosed before age 50.
- Patients with colorectal adenoma diagnosed before age 40.[79]
Physical Examination
The physical examination should include an assessment for adenopathy, particularly in the supraclavicular, cervical, and axillary regions. It should also involve a digital rectal examination, evaluation for abdominal tenderness, palpation to detect masses or ascites, and checking for hepatomegaly, which may suggest metastatic involvement. Physical examination findings are often nonspecific, and no abnormal findings may be directly related to the diagnosis. When abnormalities are present, they can include abdominal tenderness, blood in the stool, and, more rarely, a palpable abdominal or pelvic mass, which could represent the primary tumor, a metastatic lesion, or hepatomegaly if the liver is involved.
Laboratory Studies
Common laboratory findings that may raise suspicion for CRC include anemia, iron deficiency (often first indicated by low transferrin saturation, but more definitively confirmed by a low ferritin level), and markers suggestive of metastatic disease, such as elevated alkaline phosphatase or carcinoembryonic antigen levels.
Evaluation
Evaluation for Colorectal Cancer and Associated Syndromes
Diagnostic approaches for hereditary CRC include:
- Clinical findings
- Family history
- Genetic testing (blood or saliva)
- Additional blood tests
- Endoscopic evaluation, including one or more of colonoscopy, sigmoidoscopy, esophagogastroduodenoscopy (EGD), endoscopic retrograde cholangiopancreatography, and endoscopic ultrasound
- Diagnostic imaging for cancer staging
Total Colonoscopy
Total colonoscopy is the gold standard for diagnosing colorectal polyps and cancer, as it enables both biopsy and lesion resection during the procedure.[80] If colonoscopy is refused, unsuccessful, not tolerated, or contraindicated due to the inability to administer sedation, alternative diagnostic studies such as computed tomography (CT) colonography or virtual colonoscopy (or a barium enema) may be performed. However, if a colon mass is diagnosed using a test other than colonoscopy, biopsy confirmation of cancer will require an additional procedure.
Genetic Counseling
Genetic counseling should always be conducted before genetic testing is initiated in individuals with CRC, those with a family history of CRC, or when there is a family history of Lynch syndrome or another hereditary CRC syndrome. This process helps gather essential information about the proband and their family members and assists in determining whether the patient should undergo genetic testing and if they wish to proceed with it.
Molecular and Genetic Testing
If a Lynch syndrome-related gene mutation is identified in a family, testing should be performed on any newly diagnosed family member with CRC or endometrial cancer to check for the same mutation. In the absence of a known gene mutation in the family, it is now recommended that tumor tissue from all CRCs be evaluated for MSI or IHC.[81] If either or both are abnormal, genetic testing for a possible Lynch syndrome diagnosis should be conducted. The relevant MMR genes associated with Lynch syndrome include MLH1, MSH2/EPCAM, MSH6, and PMS2.
Microsatellite instability testing by PCR: Microsatellites are short, repetitive DNA sequences. Abnormal patterns of microsatellite repeats (MSI) are observed when DNA from cancer tissue with defective MMR is amplified and compared to DNA from surrounding normal tissue. PCR testing detects microsatellite instability by analyzing short tandem repeat (STR) sequences in tumor and normal tissue. A panel of 5 mononucleotide markers is used to classify tumors as MSI-High (≥2 unstable markers), MSI-Low (1 unstable marker), or Microsatellite Stable (no instability).
Immunohistochemistry: Immunohistochemistry testing assesses the presence or absence of MMR gene protein expression. If a protein from one of the MMR genes is absent, it suggests that the corresponding gene may be mutated or methylated. In such cases, genetic testing should begin with the specific gene associated with the absent protein.
Methylation: In patients with CRC and absent MLH1 expression on IHC, approximately 75% of cases are sporadic due to a BRAF gene mutation, rather than Lynch syndrome. Sporadic MSI-H CRCs typically exhibit epigenetic loss of MLH1 gene expression caused by hypermethylation of the MLH1 promoter. The presence of a BRAF mutation is often associated with this somatic loss of MLH1. If both BRAF mutation and MLH1 promoter hypermethylation tests are negative, germline mutation testing for Lynch syndrome is warranted. However, if either test result is positive, the likelihood of Lynch syndrome is extremely low.[82]
Lynch syndrome gene panel (MLH1, MSH2/EPCAM, MSH6, and PMS2): Blood testing for germline mutation in all genes associated with Lynch syndrome is essential when no previously identified MMR mutation is present in the family. Although MLH1 and MSH2 mutations are the most commonly affected genes in Lynch syndrome, initial MMR testing can be limited to these 2 genes. Testing for the other genes, including MSH6 and PMS2, can be conducted only if mutations in MLH1 and MSH2 are not detected.
Therefore, a positive genetic test (MSI), meeting the Bethesda or Amsterdam criteria, and having cancer located on the right side of the colon are strong indicators of Lynch syndrome.[83] Universal MMR-IHC, routinely performed on all CRC cases, has proven to be a crucial and effective tool for identifying individuals who may have Lynch syndrome.[70] While expanding eligibility for MGPT in CRC could reduce the need for MMR-IHC testing as part of Lynch syndrome screening,[84] MMR-IHC or MSI testing would still be necessary for therapeutic decision-making, including determining eligibility for immunotherapy.[85]
MGPT has been recommended for identifying gene mutations in affected families that lack a known mutation despite its limitations. It can detect a high rate of pathogenic germline variants, regardless of the patient's age, race, ethnicity, or the number of genes included in the panel. Although limited eligibility criteria for MGPT should be considered in patients with CRC, insurers may impose more restrictive coverage criteria.[84] When colonic polyposis presents in a single individual with a negative family history, testing for a de novo APC mutation should be considered. If the APC mutation is negative, MUTYH genetic testing should be performed. In cases where colonic polyposis is present only in siblings, recessive inheritance should be considered, and MUTYH testing should be prioritized.[86]
Diagnostic Imaging
A CT scan with contrast of the chest, abdomen, and pelvis is typically performed to stage newly diagnosed CRCs. A positron emission tomography (PET) scan, a nuclear medicine study, is often ordered after a histologic diagnosis of CRC to detect cancer in areas not visible on a contrast CT scan. In this procedure, a radioactive tracer, usually 18F-fluorodeoxyglucose (FDG), is injected intravenously. Increased uptake of the tracer in tissues beyond normal levels appears as brighter spots on the PET scan, indicating potential cancer. However, elevated FDG uptake can also result from infection, inflammation, post-operative or post-radiation healing, and other benign conditions. PET/CT scans combine both imaging techniques into a study for enhanced diagnostic accuracy.[87]
Specific aspects of an individual's family history of CRC influence the age at which screening should begin and the frequency at which it should continue. The family history of the proband’s first-degree relatives should be carefully considered when planning the screening schedule (see Image. Comparison of Guidelines for Colorectal Cancer Screening Methods and Intervals).
- Established diagnosis of CRC
- Advanced polyps that harbor any of the following characteristics:
- Adenomas measuring at least 1 cm in size
- Presence of high-grade dysplasia
- Adenomas with a villous component
- Advanced sessile serrated lesions with any of the following characteristics:
- Sessile polyps with a diameter of at least 1 cm
- Serrated adenomas with a diameter of at least 1 cm
- Presence of dysplasia in a sessile serrated polyp
Colonoscopic Findings
Lynch syndrome—Hereditary nonpolyposis colorectal cancer: Lynch syndrome typically presents with flat, premalignant polyps predominantly located in the right colon. Diagnosing Lynch syndrome can be challenging due to the lack of pathognomonic clinical signs. A definitive diagnosis requires genetic testing, and it is recommended to first identify patients for further evaluation using the Amsterdam or Bethesda criteria.[88]
Familial adenomatous polyposis: FAP is characterized by the development of hundreds to thousands of precancerous adenomatous polyps. Extracolonic manifestations include desmoid tumors, osteomas, epidermoid cysts, papillary thyroid carcinoma, pancreatic carcinoma, gastric cancer, duodenal cancer, and hepatobiliary and central nervous system tumors. These manifestations, along with CRC, result from mutations in the APC gene, which regulates cell proliferation and the expression of a novel long noncoding RNA.[86]
FAP is clinically classified in 2 ways, as mentioned below.
- Classic FAP: This condition is characterized by extensive involvement of the colon, presenting with hundreds to thousands of adenomatous polyps. The onset of polyps typically occurs between the ages of 10 and 20, with CRC diagnosis commonly in the fourth decade of life.[89][90]
- Attenuated FAP: This condition often involves the right colon and presents with 10 to 100 adenomatous polyps. Polyps usually develop between the ages of 20 and 30, with CRC diagnosis generally occurring between the ages of 40 and 50. This form is frequently associated with gastric, duodenal, and thyroid cancers.[91][92]
High-Risk Syndrome Screenings
Screening schedules for individuals with Lynch-associated cancers should be tailored based on the following factors:
- Personal and family history of cancer
- Specific gene affected
- The method used to identify the familial mutation
Expert opinions suggest that screening recommendations should account for the significantly lower lifetime cancer risk in carriers of germline PMS2 mutations, with schedules customized accordingly. Additionally, the later average age of CRC onset in individuals with germline MSH6 mutations necessitates adjustments to the screening schedule (see Table below).
Screening is recommended for individuals at risk for Lynch-associated cancers, including:
- Individuals with a pathogenic germline mutation in DNA MMR genes or deletions in EPCAM that can inactivate MSH2.
- Individuals at risk for Lynch syndrome, including those from families meeting the Amsterdam I or II criteria, and individuals meeting the revised Bethesda criteria.
- High-risk individuals with at least 2.5% to 5% risk for MMR mutation, as determined by prediction models such as PREMM5.[93]
Table. Colorectal Cancer Screening Intervals
| Syndrome | Age to Start CRC Screening | Frequency of CRC Screening | Other Factors |
| Lynch syndrome (MLH1) | 20-25 yo or 10 years before the age of the youngest affected relative | Every 1-2 years | TAH-BSO should be considered at age 40; EGD to the distal duodenum at 30-40 yo, then every 2-4 years. |
| Lynch syndrome (MSH2/EPCAM) | 20-25 yo or 10 years before the age of the youngest affected relative | Every 1-2 years | EGD to the distal duodenum at 30-40 yo, then every 2-4 years. |
| Lynch syndrome (MSH6) | 30-35 yo or 10 years before the age of the youngest affected relative | Every 1-3 years | TAH-BSO should be considered at age 40; EGD to the distal duodenum at 30-40 yo, then every 2-4 years. |
| Lynch syndrome (PMS2) | 30-35 yo or 10 years before the age of the youngest affected relative | Every 1-3 years | |
| Lynch syndrome (MSH3 biallelic pathogenic variant) | 25-30 yo or 2 years before the age of the youngest affected relative (if the relative is <25 yo) | Every 2-3 years (if no adenomas are found); every 1-2 years (if adenomas are found) | |
| Lynch syndrome (NTHL biallelic pathogenic variant) | 25-30 yo or 2 years before the age of the youngest affected relative (if the relative is <25 yo) | Every 2-3 years (if no adenomas are found); every 1-2 years (if adenomas are found) | |
| FAP | 10-12 yo | Annually |
Subtotal or total colectomy must be performed when the number of polyps is endoscopically unmanageable. EGD should be performed starting at 25-30 yo, ideally with a side-viewing scope, and repeated every 1-3 years, depending on the Spigelman stage of duodenal polyposis. |
| AFAP | Late teens | Every 1-2 years; endoscopic evaluation of the rectum every 6-12 months (depending on the extent of rectal polyposis) after subtotal colectomy and ileorectal anastomosis | Endoscopy with a side-viewing endoscope at 20-25 yo, or sooner if there is a family history of duodenal cancer or advanced duodenal adenomas. Subsequent screenings are every 6 months to 4 years, depending on the Spigelman stage of duodenal polyposis. |
| Turcot I | 10-12 yo | Annually | |
| Gardner syndrome | 10-12 yo if known APC mutation | Annually | EGD should be done at the time of the initial colonoscopy. |
| POLE | 25-30 yo or 2 years before the age of the youngest affected relative (if the relative is <25 yo) | Every 2-3 years (if no adenomas are found); every 1 to 2 years (if adenomas are found) | |
| POLD-1 | 25-30 yo or 2-5 years before the age of the youngest affected relative (if the relative is <25 yo) | Every 2-3 years (if no adenomas are found); every 1-2 years (if adenomas are found) | Subtotal or total colectomy must be done when the number of polyps is endoscopically unmanageable. EGD starts at 25-30 yo, ideally with a side-viewing scope, and repeats every 1-3 years. |
| GREM-1 | 25-30 yo or 2 years before the age of the youngest affected relative (if the relative is <25 yo) | Every 2-3 years (if no adenomas are found); every 1-2 years (if adenomas are found) | |
| MAP | 25-30 yo | Every 1-2 years | EGD at 30-35 yo. This should be repeated based on the number of gastric and duodenal polyps, with the latter based on the Spigelman stage of duodenal polyposis. Gastric fundic gland polyps should be biopsied for dysplasia. |
| Juvenile polyposis syndrome | 12 yo or sooner if symptomatic | Annually | EGD at the time of the first colonoscopy, then every 1-3 years |
| Peutz-Jeghers syndrome | 8 yo | Every 1-3 years (if polyps are found). If no polyps are found, this should be repeated at 18 yo (or sooner if symptoms develop) and every 3 years until age 50. Screening should be considered every 1-2 years after the age of 50. |
EGD should be done concurrently with colonoscopy. Small bowel evaluation by capsule endoscopy, magnetic resonance or CT enterography, double balloon enteroscopy, or small bowel series should be done at age 8 (sooner if symptoms), then every 1-3 years if many or larger polyps are found, or at age 18 if few or no polyps are found at age 8. |
| PHTS | 35-40 yo or 5-10 years before the age of the youngest affected relative | Every 5 years or sooner, based on colonoscopic findings. No published guidelines | |
| BRCA1, BRCA2, or PALB2 mutations | Same as for average-risk individuals | Same as for average-risk individuals | Increased risk of pancreatic ductal adenocarcinoma. |
| Li-Fraumeni syndrome | 25 yo, 10 years before the age of youngest relative with CRC or gastric cancer, or 5 years after abdominal or whole-body radiation therapy | Every 2-5 years | Increased risk of pancreatic ductal adenocarcinoma. |
| Serrated polyposis syndrome | Once polyps are found at screening | Every 1-2 years after larger polyps are cleared with more frequent colonoscopies | First-degree relatives should begin colonoscopy at age 40 and every 5 years if serrated polyposis syndrome is not found. |
| Monoallelic MUTYH mutation | Same as for average-risk individuals | Same as for average-risk individuals | |
| CHEK2 | Same as for average-risk individuals or based on family history | Same as for average-risk individuals or based on family history | |
| ATM | As per the family history | As per the family history | |
| APC I1370K mutation | 40 yo or 10 years before the age of the youngest affected relative | Every 5 years | |
| GAPPS | At the time of the first EGD to rule out colonic polyposis | Same as average risk screening (if there is no evidence of colorectal polyposis); annual colonoscopy (if colonoscopy shows evidence of FAP) | As for FAP, subtotal or total colectomy must be done if the number of colorectal polyps is endoscopically unmanageable. |
| Cystic fibrosis | 40 yo or at age 30 within 2 years of the transplantation if the patient has had a lung or other solid organ transplant | Every 5 years or every 3 years after transplant | The risk of other gastrointestinal and hepatobiliary malignancies is well-recognized, and screening recommendations are provided in the references above. |
Abbreviations: AFAP, attenuated familial adenomatous polyposis; APC, adenomatous polyposis coli gene; CHEK2, checkpoint kinase 2 gene; CRC, colorectal cancer; EGD, esophagogastroduodenoscopy; EPCAM, epithelial cellular adhesion molecule; FAP, familial adenomatous polyposis; GAPPS, gastric adenocarcinoma and proximal polyposis of the stomach; MAP, MUTYH-associated polyposis; MSH, MutS homolog; PHTS, PTEN hamartoma tumor syndrome; TAH-BSO, total abdominal hysterectomy with bilateral salpingo-oophorectomy; yo, years old.
The recommendations mentioned above are based on references from this article and the National Comprehensive Cancer Network® (NCCN) 2024 guidelines. These should be considered recommendations rather than definitive practice guidelines. Readers should review individual references to assess the strength of supporting evidence.
Guidelines From the National Comprehensive Cancer Network
Testing for Lynch syndrome should be carried out if there is:
- A known Lynch syndrome pathogenic variant in the family.
- Personal history of a tumor with MMR deficiency diagnosed at any age. The NCCN recommends tumor screening for MMR deficiency diagnosed in all cases of CRCs and endometrial cancers, regardless of age. Tumor screening for MMR deficiency should also be considered for small bowel, gastric, pancreatic, biliary tract, brain, bladder, urothelial, and adrenocortical adenocarcinomas.
An individual with CRC or endometrial cancer meeting any of the following criteria:
- Diagnosis of cancer before the age of 50.
- Presence of synchronous or metachronous Lynch syndrome-related cancer at any age.
- A first- or second-degree relative diagnosed with a Lynch syndrome-related cancer before the age of 50.
- At least 2 first- or second-degree relatives diagnosed with Lynch syndrome-related cancers, regardless of age.
Family history of any of the following:
- At least 1 first-degree relative with CRC or endometrial cancer diagnosed before the age of 50.
- At least 1 first-degree relative with CRC or endometrial cancer and a synchronous or metachronous Lynch syndrome–related cancer, regardless of age.
- At least 2 first- or second-degree relatives with Lynch syndrome–related cancers, including one or more diagnosed before the age of 50.
- At least 3 first- or second-degree relatives with Lynch syndrome-related cancers, regardless of age.
- Increased model-predicted risk for Lynch syndrome.
- An individual with 5% or more risks of carrying an MMR gene pathogenic variant, as indicated by predictive models, such as PREMM5, MMRpro, and MMRpredict.
- Individuals with a personal history of colorectal or endometrial cancer and a PREMM5 score of at least 2.5% should be considered for MGPT.
PREMM5 Model for Risk Assessment of Lynch Syndrome
For individuals without a personal history of either CRC or endometrial cancer, some data suggest using a PREMM5 score threshold of 2.5% to determine eligibility for MMR genetic testing.[94] Refer to the study by Kastrinos et al for the complete PREMM5 model equation.[95] This model considers multiple risk factors for Lynch syndrome, including sex, age at evaluation, presence of CRC, endometrial cancer, or other Lynch syndrome-associated cancers in the patient, and the presence or absence of these cancers in first- and second-degree relatives.
Treatment / Management
The treatment of patients with hereditary CRC is multidisciplinary and mainly based on the patient's specific clinical and genetic findings.
Chemotherapy and Targeted therapy
Hereditary CRCs may respond differently to traditional chemotherapy regimens depending on the genetic mutation involved. MSI often reflects distinct biological characteristics of the tumor, with MSI-H tumors potentially showing unique susceptibility to immunotherapy.[96][97] High-risk rectal cancers require neoadjuvant chemotherapy along with radiotherapy.[98] (B3)
- The current guidelines from the NCCN, American Society of Clinical Oncology (ASCO), and European Society for Medical Oncology (ESMO) strongly recommend adjuvant therapy for selected stage II colorectal cancers with high-risk features and proficient MMR or MSI-S disease, as well as for all stage III (node-positive) cases. These guidelines advocate initiating adjuvant chemotherapy (FOLFOX or CAPOX) within 6 to 8 weeks after surgical resection, provided the patient has sufficiently recovered.
-
FOLFOX (a combination of 5-fluorouracil, leucovorin, and oxaliplatin) is the first-line chemotherapy for metastatic CRC and has demonstrated superior efficacy compared to single-agent 5-fluorouracil therapy. Clinical trials have shown that FOLFOX significantly improves progression-free survival (PFS; HR: 0.59; 95% CI: 0.49-0.73; based on 2 randomized controlled trials [RCTs]) and OS (HR: 0.69; 95% CI: 0.51-0.94; based on 1 RCT).[99]
(A1)
-
FOLFIRI (a combination of 5-fluorouracil, leucovorin, and irinotecan) is another effective chemotherapy regimen for metastatic CRC and can be used as either a first- or second-line treatment. Clinical trials have demonstrated its efficacy, with a significant improvement in OS (HR: 0.58; 95% CI: 0.43-0.80; based on 1 RCT).Irinotecan-based combination therapies demonstrated greater effectiveness compared to irinotecan monotherapy, significantly improving PFS (HR: 0.68; 95% CI: 0.60–0.76; based on 6 RCTs).[99]
-
Targeted agents further enhance the effectiveness of conventional chemotherapy. Adding bevacizumab (an anti-VEGF monoclonal antibody) to chemotherapy for metastatic CRC has been shown to improve PFS and OS in multiple phase III trials. The AVF2107g trial demonstrated a significant OS benefit when bevacizumab was added to irinotecan, fluorouracil, and leucovorin (IFL).[100] The NO16966 trial showed that adding bevacizumab to FOLFOX/XELOX significantly improved PFS (9.4 vs. 8.0 months, p = 0.0023) in metastatic colorectal cancer, though OS benefit was not significant due to crossover. Subsequent studies confirmed efficacy with other chemo regimens, making bevacizumab a standard first-line treatment in metastatic CRC.[101]
(A1)
- In metastatic CRC, monoclonal antibodies such as bevacizumab, cetuximab, and panitumumab are commonly used in combination with chemotherapy. Adding bevacizumab to chemotherapy provides a notable benefit for right-sided tumors, while cetuximab enhances outcomes for left-sided tumors in RAS/RAF wild-type cases. These agents can be incorporated into oxaliplatin- and irinotecan-based regimens, making them valuable options for first-line therapy.[102]
- A meta-analysis, including the TAILOR and PRIME studies, indicates that primary tumor location predicts the survival benefit of adding an anti-epidermal growth factor receptor antibody to the FOLFOX regimen in patients with KRAS/NRAS/BRAF wild-type metastatic CRC. The analysis showed a significant benefit for OS for left-sided tumors (HR for left-sided: 0.71; 95% CI: 0.59-0.85; P=.0002), unlike right-sided tumors (HR for right-sided: 0.90; 95% CI: 0.65-1.25; P=.53). The efficacy was similar in patients with wild-type RAS and BRAF molecular characteristics.[103]
- Recently the Phase III BREAKWATER trial demonstrated that adding encorafenib and cetuximab to the chemotherapy regimen FOLFOX significantly improved outcomes for patients with previously untreated BRAF V600E–mutant metastatic CRC. The combination achieved a 60.9% overall response rate compared to 40% with standard chemotherapy, and a median duration of response of 13.9 months versus 11.1 months, respectively. These findings led to the FDA's accelerated approval of this regimen in December 2024, establishing a new first-line treatment standard for this patient population.[104] (A1)
Differential Diagnosis
Solamen Syndrome
Solamen syndrome refers to segmental overgrowth, lipomatosis, arteriovenous malformation, and epidermal nevus. This condition is considered part of Cowden syndrome, with patients carrying germline mutations in the PTEN homologs.[105]
Bannayan-Riley-Ruvalcaba Syndrome
Bannayan-Riley-Ruvalcaba syndrome is a congenital disorder often characterized by hamartomatous intestinal polyps, lipomas, macrocephaly, and genital lentiginosis. Patients with Bannayan-Riley-Ruvalcaba syndrome who carry a PTEN mutation share the same cancer risk as those with Cowden syndrome.[106]
Proteus Syndrome
Proteus syndrome is a disorder characterized by progressive excessive growth in the skeleton, skin, fat, and central nervous system, with a few hamartomatous polyps. Complications include hemimegalencephaly, bullous lung disease, pulmonary embolism, and deep vein thrombosis. Although the polyps are typically benign, rare cases of malignancy have been reported.[107]
Surgical Oncology
Surgical management should be tailored to the specific hereditary CRC. For patients with AFAP and a significant number of rectal adenomas, the preferred treatment is proctocolectomy with ileostomy or ileal-pouch anal anastomosis. However, the timing of the procedure should be individualized based on the patient’s condition and clinical factors. If all rectal adenomas larger than 5 mm can be removed endoscopically, a rectal-sparing alternative, such as subtotal abdominal colectomy and total abdominal colectomy with ileorectal anastomosis (TAC-IRA), is a viable and commonly used alternative. Additionally, in patients with more than 20 lifetime adenomas, a diagnosis of MAP should be considered. For patients with biallelic MYH mutations, annual colonoscopy and endoscopic polypectomy are recommended for managing removable polyps.[108]
Surgery
The appropriate surgical option for patients with colon cancer and Lynch syndrome, such as subtotal abdominal colectomy or TAC-IRA, will depend on various risk factors. The choice of surgery should also consider the patient's preferences, with particular attention to their age and ability to adhere to intensive surveillance.[109] Currently, subtotal colectomy is the most commonly performed surgical method for these patients.[109]
For Lynch syndrome patients with MLH1, MSH2, or MLH6 mutations, where the risks of endometrial and ovarian cancers are significantly elevated, it is recommended that patients complete their families and undergo prophylactic total abdominal hysterectomy with bilateral salpingo-oophorectomy (TAH-BSO) starting at age 40. This procedure should be considered concurrently with colorectal resection or other abdominal surgery, if needed.[110][111][112]
In FAP, prophylactic colectomy is a critical intervention to prevent colorectal cancer, as nearly all patients with FAP will develop cancer by age 30 unless a total or subtotal colectomy is performed early.[113] For patients with attenuated FAP, the preventive surgical approach is more limited.[114] However, surveillance and surgical planning should be guided by a comprehensive assessment of familial cancer risk.[115]
Radiation Oncology
Radiation therapy is commonly used in the management of rectal cancer but is less effective for treating the colon, as the colon's mobility within the abdomen prevents targeted application. Preoperative hypofractionated radiotherapy combined with chemotherapy is recommended for patients with stage II or III rectal cancer or when there is an unresectable tumor burden, such as positive surgical margins following resection.[116] Additionally, radiotherapy is typically reserved for palliative treatment in selected cases of metastasis, such as to the bone or brain.[117]
Medical Oncology
Microsatellite Instability-High (MSI-H) and Its Implications for Treatment in Hereditary Colorectal Cancer
MSI-H, a hallmark of colorectal cancer linked to Lynch syndrome, significantly impact treatment strategies, particularly in adjuvant chemotherapy and immunotherapy.
For patients with MSI-H tumors, fluoropyrimidine-based chemotherapy alone has shown limited benefit and may even be detrimental in the adjuvant setting. However, combination therapy with leucovorin calcium (folinic acid), fluorouracil, and oxaliplatin (FOLFOX) has demonstrated positive outcomes particularly in adjuvant treatment of Stage III MSI-H CRC.[118] Notably, these negative effects of single-agent fluorouracil or FOLFOX have not been observed in the metastatic setting.[119]
Immunotherapy with immune checkpoint inhibitors targeting programmed death receptor-1 (PD-1) has been highly effective for both front-line treatment of MSI-H metastatic CRC and cases that have progressed despite conventional chemotherapy. The KEYNOTE-177 trial established pembrolizumab as more effective than chemotherapy, demonstrating significantly longer progression-free survival and sustained responses in previously untreated metastatic MSI-H/dMMR colorectal cancer.[120] Additionally, dual blockade of PD-1 and cytotoxic T lymphocyte-associated protein 4 (CTLA-4) has shown improved disease control without increasing toxicity.[121] Ongoing clinical investigations are evaluating the role of immunotherapy with anti-PD-1/PD-L1 and anti-CTLA-4 monoclonal antibodies in multiple settings, including adjuvant therapy, maintenance therapy, and even preventive strategies in Lynch syndrome patients without a cancer diagnosis.[122] Moreover, MSI-H tumors outside of CRC have also exhibited strong responsiveness to PD-1/PD-L1 blockade.
Neoadjuvant chemotherapy in early-stage colon cancer has shown limited efficacy, particularly in MSI-H/dMMR tumors (FOxTROT trial: 7% regression rate). In contrast, neoadjuvant immunotherapy has demonstrated remarkable pathologic responses, with NICHE-1 and NICHE-2 showing 97% and 95% MPR rates, respectively, and no recurrence.[123][124][123] Emerging trials (PICC, NICHE-3, IMHOTEP) further support this approach, suggesting potential for organ preservation, though optimizing treatment duration may enhance outcomes.
Prognosis
In a study by Lee et al, patients with sporadic CRC who had a family history of CRC demonstrated better OS compared to those without a family history. However, cancer recurrence rates were similar between the 2 groups.[125]
CRC survivors (5-10 years post-diagnosis) generally reported good health-related quality of life. However, they experienced significantly poorer social functioning and reported more issues with dyspnea, constipation, diarrhea, and financial challenges compared to controls, particularly among younger individuals.[126]
Complications
Severe complications during colonoscopy are a significant concern in CRC screening programs. Immediate complications, including perforation and hemorrhagic events, occur in approximately 3.3% of cases. Several factors increase the risk of early and late complications, such as a history of colon disease, anticoagulation therapy, pelvic surgery, abdominal radiotherapy, or a history of perforation.[127]
After a total colectomy, systemic complications occur in approximately 22%, particularly in patients aged 65 and older with significant comorbidities. Surgical complications are reported in 8% of cases and include anastomotic leakage, major wound infections, and postoperative hemorrhage.[128]
Deterrence and Patient Education
Educational efforts are essential to raise awareness about CRC, especially among younger individuals. Improving adherence to screening guidelines for eligible young patients and emphasizing the early evaluation of symptoms are crucial steps in reducing the burden of CRC in this population.[129]
Adopting a healthy lifestyle and dietary habits is recommended to mitigate the key factors contributing to colorectal carcinogenesis, which often begins in the early stages of life.[130]
Enhancing Healthcare Team Outcomes
An interprofessional approach involves a gastroenterologist, oncologist, colorectal surgeon, primary care provider, mid-level practitioners, geneticist, genetic counselor, nursing staff, and pharmacist. Genetic testing should be offered to individuals with suspected hereditary CRC and their family members after genetic counseling.[131] For individuals with a first-degree family history of classical FAP, the recommended screening strategy is an annual colonoscopy starting between ages 12 and 15 and continuing until 30 to 35. After that, screening should occur every 5 years.[132][133]
Effective screening and chemoprevention strategies are essential for reducing CRC incidence and mortality in high-risk populations, with specific approaches tailored to genetic risk factors and associated extracolonic cancers.
-
For individuals with a family history or suspicion of Lynch syndrome, it is recommended to undergo a colonoscopy every 2 years, starting at ages 20 to 25, or 5 to 10 years before the youngest diagnosed case in the family.[134][135]
-
Studies consistently show that early registration and regular screening significantly reduce the incidence and mortality of colorectal cancer (CRC) in patients with FAP and Lynch syndrome.[136]
-
The MSI test is effective for screening Lynch syndrome, but must align with the appropriate marker panel or thresholds to be considered a positive test.[137]
-
The studies indicate that APC promoter hypermethylation is an early event in CRC carcinogenesis in APC patients, potentially serving as a valuable diagnostic marker for early-stage CRC.[138][139]
- Current evidence supports the use of nonsteroidal anti-inflammatory drugs, sulindac, and low-dose aspirin as chemopreventive agents, which help reduce the progression of polyps and the long-term incidence of CRCs in patients with FAP and HNPCC, respectively.[140]
- Close monitoring is essential for syndromes linked to CRC and extracolonic manifestations, particularly Lynch syndrome, which carries an elevated risk of endometrial cancer, along with other conditions outlined in the Differential Diagnosis section.[141]
- Risk factors associated with increased mortality in CRC include advanced age, male sex, African-American race, elevated carcinoembryonic antigen levels, and the absence of curative surgery. Additionally, CRC patients face a heightened risk of cardiovascular death, particularly within the first year following diagnosis.[142]
-
Overall, FOLFOX with or without bevacizumab is the most effective first-line treatment for metastatic CRC. In contrast, FOLFIRI plus cetuximab is the most effective regimen for left-sided KRAS/NRAS/BRAF wild-type tumors.[143]
- Bevacizumab offers hope for improved survival and the possibility of metastasectomy in patients with metastatic CRC. However, its use is associated with several adverse events, including severe hypertension and gastrointestinal perforation, which require careful monitoring.[144]
- The treating physician should recommend a diet rich in dietary fiber, as it may reduce the risk of CRC and lower the likelihood of postoperative relapses.[145][146][147]
Regardless of the treatment path (such as chemotherapy and surgery), all members of the interprofessional healthcare team must closely monitor the patient, provide counsel, and notify other team members when changes in the patient's condition occur, such as adverse drug events or signs of therapeutic failure or relapse. These observations should be documented in the patient's medical record to enable timely adjustments to the treatment plan, optimizing patient outcomes. An interprofessional approach to patient care will enhance the patient's well-being and ensure the delivery of appropriate therapeutic interventions.
Media
(Click Image to Enlarge)
Common Sites of Breast Cancer Metastasis.
Medical Gallery of Mikael Häggström, Public Domain, via Wikimedia Commons
(Click Image to Enlarge)
References
Valle L, Vilar E, Tavtigian SV, Stoffel EM. Genetic predisposition to colorectal cancer: syndromes, genes, classification of genetic variants and implications for precision medicine. The Journal of pathology. 2019 Apr:247(5):574-588. doi: 10.1002/path.5229. Epub 2019 Feb 20 [PubMed PMID: 30584801]
Baglietto L, Jenkins MA, Severi G, Giles GG, Bishop DT, Boyle P, Hopper JL. Measures of familial aggregation depend on definition of family history: meta-analysis for colorectal cancer. Journal of clinical epidemiology. 2006 Feb:59(2):114-24 [PubMed PMID: 16426946]
Level 1 (high-level) evidencePDQ Cancer Genetics Editorial Board. Genetics of Colorectal Cancer (PDQ®): Health Professional Version. PDQ Cancer Information Summaries. 2002:(): [PubMed PMID: 26389505]
Level 3 (low-level) evidenceKastrinos F, Samadder NJ, Burt RW. Use of Family History and Genetic Testing to Determine Risk of Colorectal Cancer. Gastroenterology. 2020 Jan:158(2):389-403. doi: 10.1053/j.gastro.2019.11.029. Epub 2019 Nov 21 [PubMed PMID: 31759928]
Win AK, Jenkins MA, Dowty JG, Antoniou AC, Lee A, Giles GG, Buchanan DD, Clendenning M, Rosty C, Ahnen DJ, Thibodeau SN, Casey G, Gallinger S, Le Marchand L, Haile RW, Potter JD, Zheng Y, Lindor NM, Newcomb PA, Hopper JL, MacInnis RJ. Prevalence and Penetrance of Major Genes and Polygenes for Colorectal Cancer. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2017 Mar:26(3):404-412. doi: 10.1158/1055-9965.EPI-16-0693. Epub 2016 Oct 31 [PubMed PMID: 27799157]
Level 3 (low-level) evidenceBonadona V, Bonaïti B, Olschwang S, Grandjouan S, Huiart L, Longy M, Guimbaud R, Buecher B, Bignon YJ, Caron O, Colas C, Noguès C, Lejeune-Dumoulin S, Olivier-Faivre L, Polycarpe-Osaer F, Nguyen TD, Desseigne F, Saurin JC, Berthet P, Leroux D, Duffour J, Manouvrier S, Frébourg T, Sobol H, Lasset C, Bonaïti-Pellié C, French Cancer Genetics Network. Cancer risks associated with germline mutations in MLH1, MSH2, and MSH6 genes in Lynch syndrome. JAMA. 2011 Jun 8:305(22):2304-10. doi: 10.1001/jama.2011.743. Epub [PubMed PMID: 21642682]
Porkka N, Lahtinen L, Ahtiainen M, Böhm JP, Kuopio T, Eldfors S, Mecklin JP, Seppälä TT, Peltomäki P. Epidemiological, clinical and molecular characterization of Lynch-like syndrome: A population-based study. International journal of cancer. 2019 Jul 1:145(1):87-98. doi: 10.1002/ijc.32085. Epub 2019 Jan 7 [PubMed PMID: 30575961]
Level 2 (mid-level) evidenceKempers MJ, Kuiper RP, Ockeloen CW, Chappuis PO, Hutter P, Rahner N, Schackert HK, Steinke V, Holinski-Feder E, Morak M, Kloor M, Büttner R, Verwiel ET, van Krieken JH, Nagtegaal ID, Goossens M, van der Post RS, Niessen RC, Sijmons RH, Kluijt I, Hogervorst FB, Leter EM, Gille JJ, Aalfs CM, Redeker EJ, Hes FJ, Tops CM, van Nesselrooij BP, van Gijn ME, Gómez García EB, Eccles DM, Bunyan DJ, Syngal S, Stoffel EM, Culver JO, Palomares MR, Graham T, Velsher L, Papp J, Oláh E, Chan TL, Leung SY, van Kessel AG, Kiemeney LA, Hoogerbrugge N, Ligtenberg MJ. Risk of colorectal and endometrial cancers in EPCAM deletion-positive Lynch syndrome: a cohort study. The Lancet. Oncology. 2011 Jan:12(1):49-55. doi: 10.1016/S1470-2045(10)70265-5. Epub 2010 Dec 8 [PubMed PMID: 21145788]
Roberts ME, Riegert-Johnson DL, Thomas BC, Rumilla KM, Thomas CS, Heckman MG, Purcell JU, Hanson NB, Leppig KA, Lim J, Cappel MA. A clinical scoring system to identify patients with sebaceous neoplasms at risk for the Muir-Torre variant of Lynch syndrome. Genetics in medicine : official journal of the American College of Medical Genetics. 2014 Sep:16(9):711-6. doi: 10.1038/gim.2014.19. Epub 2014 Mar 6 [PubMed PMID: 24603434]
Hu XR, Xu C, Kang Y, Wang T, Zhang Y, Yang XH. [Correlation between mismatch-repair protein expression and clinicopathologic features in 658 colorectal cancers]. Zhonghua bing li xue za zhi = Chinese journal of pathology. 2018 Nov 8:47(11):827-833. doi: 10.3760/cma.j.issn.0529-5807.2018.11.003. Epub [PubMed PMID: 30423605]
Karstensen JG, Burisch J, Pommergaard HC, Aalling L, Højen H, Jespersen N, Schmidt PN, Bülow S. Colorectal Cancer in Individuals With Familial Adenomatous Polyposis, Based on Analysis of the Danish Polyposis Registry. Clinical gastroenterology and hepatology : the official clinical practice journal of the American Gastroenterological Association. 2019 Oct:17(11):2294-2300.e1. doi: 10.1016/j.cgh.2019.02.008. Epub 2019 Feb 8 [PubMed PMID: 30743005]
Poylin VY, Shaffer VO, Felder SI, Goldstein LE, Goldberg JE, Kalady MF, Lightner AL, Feingold DL, Paquette IM, Clinical Practice Guidelines Committee of the American Society of Colon and Rectal Surgeons. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Management of Inherited Adenomatous Polyposis Syndromes. Diseases of the colon and rectum. 2024 Feb 1:67(2):213-227. doi: 10.1097/DCR.0000000000003072. Epub 2023 Sep 8 [PubMed PMID: 37682806]
Level 1 (high-level) evidenceSpigelman AD, Williams CB, Talbot IC, Domizio P, Phillips RK. Upper gastrointestinal cancer in patients with familial adenomatous polyposis. Lancet (London, England). 1989 Sep 30:2(8666):783-5 [PubMed PMID: 2571019]
Knudsen AL, Bisgaard ML, Bülow S. Attenuated familial adenomatous polyposis (AFAP). A review of the literature. Familial cancer. 2003:2(1):43-55 [PubMed PMID: 14574166]
Yang J, Gurudu SR, Koptiuch C, Agrawal D, Buxbaum JL, Abbas Fehmi SM, Fishman DS, Khashab MA, Jamil LH, Jue TL, Law JK, Lee JK, Naveed M, Qumseya BJ, Sawhney MS, Thosani N, Wani SB, Samadder NJ. American Society for Gastrointestinal Endoscopy guideline on the role of endoscopy in familial adenomatous polyposis syndromes. Gastrointestinal endoscopy. 2020 May:91(5):963-982.e2. doi: 10.1016/j.gie.2020.01.028. Epub 2020 Mar 10 [PubMed PMID: 32169282]
Forkosh E, Bergel M, Hatchell KE, Nielsen SM, Heald B, Benson AA, Friedman E, Esplin ED, Katz LH. Ashkenazi Jewish and Other White APC I1307K Carriers Are at Higher Risk for Multiple Cancers. Cancers. 2022 Nov 29:14(23):. doi: 10.3390/cancers14235875. Epub 2022 Nov 29 [PubMed PMID: 36497357]
Valle L, Katz LH, Latchford A, Mur P, Moreno V, Frayling IM, Heald B, Capellá G, InSiGHT Council. Position statement of the International Society for Gastrointestinal Hereditary Tumours (InSiGHT) on APC I1307K and cancer risk. Journal of medical genetics. 2023 Nov:60(11):1035-1043. doi: 10.1136/jmg-2022-108984. Epub 2023 Apr 19 [PubMed PMID: 37076288]
Sokic-Milutinovic A. Appropriate Management of Attenuated Familial Adenomatous Polyposis: Report of a Case and Review of the Literature. Digestive diseases (Basel, Switzerland). 2019:37(5):400-405. doi: 10.1159/000497207. Epub 2019 Mar 5 [PubMed PMID: 30836352]
Level 3 (low-level) evidenceBurn J, Bishop DT, Chapman PD, Elliott F, Bertario L, Dunlop MG, Eccles D, Ellis A, Evans DG, Fodde R, Maher ER, Möslein G, Vasen HF, Coaker J, Phillips RK, Bülow S, Mathers JC, International CAPP consortium. A randomized placebo-controlled prevention trial of aspirin and/or resistant starch in young people with familial adenomatous polyposis. Cancer prevention research (Philadelphia, Pa.). 2011 May:4(5):655-65. doi: 10.1158/1940-6207.CAPR-11-0106. Epub [PubMed PMID: 21543343]
Level 1 (high-level) evidenceSoravia C, Berk T, McLeod RS, Cohen Z. Desmoid disease in patients with familial adenomatous polyposis. Diseases of the colon and rectum. 2000 Mar:43(3):363-9 [PubMed PMID: 10733118]
Buecher B. Colorectal adenomatous polyposis syndromes: Genetic determinism, clinical presentation and recommendations for care. Bulletin du cancer. 2016 Feb:103(2):199-209. doi: 10.1016/j.bulcan.2015.10.019. Epub 2016 Jan 19 [PubMed PMID: 26805944]
Velter C, Caussade P, Fricker JP, Cribier B. [Muir-Torre syndrome and Turcot syndrome]. Annales de dermatologie et de venereologie. 2017 Aug-Sep:144(8-9):525-529. doi: 10.1016/j.annder.2017.01.017. Epub 2017 Feb 27 [PubMed PMID: 28256262]
Al-Tassan N, Chmiel NH, Maynard J, Fleming N, Livingston AL, Williams GT, Hodges AK, Davies DR, David SS, Sampson JR, Cheadle JP. Inherited variants of MYH associated with somatic G:C--}T:A mutations in colorectal tumors. Nature genetics. 2002 Feb:30(2):227-32 [PubMed PMID: 11818965]
Sutcliffe EG, Bartenbaker Thompson A, Stettner AR, Marshall ML, Roberts ME, Susswein LR, Wang Y, Klein RT, Hruska KS, Solomon BD. Multi-gene panel testing confirms phenotypic variability in MUTYH-Associated Polyposis. Familial cancer. 2019 Apr:18(2):203-209. doi: 10.1007/s10689-018-00116-2. Epub [PubMed PMID: 30604180]
Kantor M, Sobrado J, Patel S, Eiseler S, Ochner C. Hereditary Colorectal Tumors: A Literature Review on MUTYH-Associated Polyposis. Gastroenterology research and practice. 2017:2017():8693182. doi: 10.1155/2017/8693182. Epub 2017 Sep 25 [PubMed PMID: 29147111]
Bikhchandani M, Amersi F, Hendifar A, Gangi A, Osipov A, Zaghiyan K, Atkins K, Cho M, Aguirre F, Hazelett D, Alvarez R, Zhou L, Hitchins M, Gong J. POLE-Mutant Colon Cancer Treated with PD-1 Blockade Showing Clearance of Circulating Tumor DNA and Prolonged Disease-Free Interval. Genes. 2023 May 8:14(5):. doi: 10.3390/genes14051054. Epub 2023 May 8 [PubMed PMID: 37239414]
Bellido F, Pineda M, Aiza G, Valdés-Mas R, Navarro M, Puente DA, Pons T, González S, Iglesias S, Darder E, Piñol V, Soto JL, Valencia A, Blanco I, Urioste M, Brunet J, Lázaro C, Capellá G, Puente XS, Valle L. POLE and POLD1 mutations in 529 kindred with familial colorectal cancer and/or polyposis: review of reported cases and recommendations for genetic testing and surveillance. Genetics in medicine : official journal of the American College of Medical Genetics. 2016 Apr:18(4):325-32. doi: 10.1038/gim.2015.75. Epub 2015 Jul 2 [PubMed PMID: 26133394]
Level 3 (low-level) evidenceJaeger E, Leedham S, Lewis A, Segditsas S, Becker M, Cuadrado PR, Davis H, Kaur K, Heinimann K, Howarth K, HMPS Collaboration, East J, Taylor J, Thomas H, Tomlinson I. Hereditary mixed polyposis syndrome is caused by a 40-kb upstream duplication that leads to increased and ectopic expression of the BMP antagonist GREM1. Nature genetics. 2012 May 6:44(6):699-703. doi: 10.1038/ng.2263. Epub 2012 May 6 [PubMed PMID: 22561515]
Mankaney G, Rouphael C, Burke CA. Serrated Polyposis Syndrome. Clinical gastroenterology and hepatology : the official clinical practice journal of the American Gastroenterological Association. 2020 Apr:18(4):777-779. doi: 10.1016/j.cgh.2019.09.006. Epub 2019 Sep 11 [PubMed PMID: 31520728]
Rosty C, Brosens LAA. Pathology of Gastrointestinal Polyposis Disorders. Gastroenterology clinics of North America. 2024 Mar:53(1):179-200. doi: 10.1016/j.gtc.2023.09.006. Epub 2023 Oct 20 [PubMed PMID: 38280747]
Viel A, Bruselles A, Meccia E, Fornasarig M, Quaia M, Canzonieri V, Policicchio E, Urso ED, Agostini M, Genuardi M, Lucci-Cordisco E, Venesio T, Martayan A, Diodoro MG, Sanchez-Mete L, Stigliano V, Mazzei F, Grasso F, Giuliani A, Baiocchi M, Maestro R, Giannini G, Tartaglia M, Alexandrov LB, Bignami M. A Specific Mutational Signature Associated with DNA 8-Oxoguanine Persistence in MUTYH-defective Colorectal Cancer. EBioMedicine. 2017 Jun:20():39-49. doi: 10.1016/j.ebiom.2017.04.022. Epub 2017 Apr 13 [PubMed PMID: 28551381]
Gupta V, East JE. Optimal Endoscopic Treatment and Surveillance of Serrated Polyps. Gut and liver. 2020 Jul 15:14(4):423-429. doi: 10.5009/gnl19202. Epub [PubMed PMID: 31581390]
Latchford AR, Neale K, Phillips RK, Clark SK. Juvenile polyposis syndrome: a study of genotype, phenotype, and long-term outcome. Diseases of the colon and rectum. 2012 Oct:55(10):1038-43. doi: 10.1097/DCR.0b013e31826278b3. Epub [PubMed PMID: 22965402]
Matsumoto T, Umeno J, Jimbo K, Arai M, Iwama I, Kashida H, Kudo T, Koizumi K, Sato Y, Sekine S, Tanaka S, Tanakaya K, Tamura K, Hirata K, Fukahori S, Esaki M, Ishikawa H, Iwama T, Okazaki Y, Saito Y, Matsuura N, Mutoh M, Tomita N, Akiyama T, Yamamoto T, Ishida H, Nakayama Y. Clinical Guidelines for Diagnosis and Management of Juvenile Polyposis Syndrome in Children and Adults-Secondary Publication. Journal of the anus, rectum and colon. 2023:7(2):115-125. doi: 10.23922/jarc.2023-002. Epub 2023 Apr 25 [PubMed PMID: 37113581]
Gilad O, Rosner G, Fliss-Isakov N, Aharon-Kaspi S, Strul H, Gluck N, Kariv R. Clinical and Histologic Overlap and Distinction Among Various Hamartomatous Polyposis Syndromes. Clinical and translational gastroenterology. 2019 May 22:10(5):1-9. doi: 10.14309/ctg.0000000000000035. Epub [PubMed PMID: 31107726]
McDonald NM, Ramos GP, Sweetser S. SMAD4 mutation and the combined juvenile polyposis and hereditary hemorrhage telangiectasia syndrome: a single center experience. International journal of colorectal disease. 2020 Oct:35(10):1963-1965. doi: 10.1007/s00384-020-03670-3. Epub 2020 Jun 15 [PubMed PMID: 32556653]
Spoto CPE, Gullo I, Carneiro F, Montgomery EA, Brosens LAA. Hereditary gastrointestinal carcinomas and their precursors: An algorithm for genetic testing. Seminars in diagnostic pathology. 2018 May:35(3):170-183. doi: 10.1053/j.semdp.2018.01.004. Epub 2018 Jan 31 [PubMed PMID: 29397239]
Sengupta S, Bose S. Peutz-Jeghers Syndrome. The New England journal of medicine. 2019 Jan 31:380(5):472. doi: 10.1056/NEJMicm1806623. Epub [PubMed PMID: 30699321]
Chen HY, Jin XW, Li BR, Zhu M, Li J, Mao GP, Zhang YF, Ning SB. Cancer risk in patients with Peutz-Jeghers syndrome: A retrospective cohort study of 336 cases. Tumour biology : the journal of the International Society for Oncodevelopmental Biology and Medicine. 2017 Jun:39(6):1010428317705131. doi: 10.1177/1010428317705131. Epub [PubMed PMID: 28653895]
Level 2 (mid-level) evidenceKorsse SE, Harinck F, van Lier MG, Biermann K, Offerhaus GJ, Krak N, Looman CW, van Veelen W, Kuipers EJ, Wagner A, Dekker E, Mathus-Vliegen EM, Fockens P, van Leerdam ME, Bruno MJ. Pancreatic cancer risk in Peutz-Jeghers syndrome patients: a large cohort study and implications for surveillance. Journal of medical genetics. 2013 Jan:50(1):59-64. doi: 10.1136/jmedgenet-2012-101277. Epub [PubMed PMID: 23240097]
Yamamoto H, Sakamoto H, Kumagai H, Abe T, Ishiguro S, Uchida K, Kawasaki Y, Saida Y, Sano Y, Takeuchi Y, Tajika M, Nakajima T, Banno K, Funasaka Y, Hori S, Yamaguchi T, Yoshida T, Ishikawa H, Iwama T, Okazaki Y, Saito Y, Matsuura N, Mutoh M, Tomita N, Akiyama T, Yamamoto T, Ishida H, Nakayama Y. Clinical Guidelines for Diagnosis and Management of Peutz-Jeghers Syndrome in Children and Adults. Digestion. 2023:104(5):335-347. doi: 10.1159/000529799. Epub 2023 Apr 13 [PubMed PMID: 37054692]
Yauy K, Imbert-Bouteille M, Bubien V, Lindet-Bourgeois C, Rathat G, Perrochia H, MacGrogan G, Longy M, Bessis D, Tinat J, Baert-Desurmont S, Blanluet M, Perre PV, Baudry K, Pujol P, Corsini C. Ovarian Clear Cell Carcinoma in Cowden Syndrome. Journal of the National Comprehensive Cancer Network : JNCCN. 2019 Jan:17(1):7-11. doi: 10.6004/jnccn.2018.7065. Epub [PubMed PMID: 30659124]
Eissing M, Ripken L, Schreibelt G, Westdorp H, Ligtenberg M, Netea-Maier R, Netea MG, de Vries IJM, Hoogerbrugge N. PTEN Hamartoma Tumor Syndrome and Immune Dysregulation. Translational oncology. 2019 Feb:12(2):361-367. doi: 10.1016/j.tranon.2018.11.003. Epub 2018 Nov 30 [PubMed PMID: 30504085]
Heald B, Burke CA, Kalady M, Eng C. ACG Guidelines on Management of PTEN-Hamartoma Tumor Syndrome: Does the Evidence Support so Much so Young? The American journal of gastroenterology. 2015 Dec:110(12):1733-4. doi: 10.1038/ajg.2015.368. Epub [PubMed PMID: 26673503]
Tischkowitz M, Colas C, Pouwels S, Hoogerbrugge N, PHTS Guideline Development Group, European Reference Network GENTURIS. Cancer Surveillance Guideline for individuals with PTEN hamartoma tumour syndrome. European journal of human genetics : EJHG. 2020 Oct:28(10):1387-1393. doi: 10.1038/s41431-020-0651-7. Epub 2020 Jun 12 [PubMed PMID: 32533092]
Yehia L, Ngeow J, Eng C. PTEN-opathies: from biological insights to evidence-based precision medicine. The Journal of clinical investigation. 2019 Feb 1:129(2):452-464. doi: 10.1172/JCI121277. Epub 2019 Jan 7 [PubMed PMID: 30614812]
Kupfer SS, Gupta S, Weitzel JN, Samadder J. AGA Clinical Practice Update on Colorectal and Pancreatic Cancer Risk and Screening in BRCA1 and BRCA2 Carriers: Commentary. Gastroenterology. 2020 Aug:159(2):760-764. doi: 10.1053/j.gastro.2020.03.086. Epub 2020 Apr 23 [PubMed PMID: 32335146]
Level 3 (low-level) evidencePan W, Lu K, Wang W, Yao J, Hou Y. PALB2 as a potential prognostic biomarker for colorectal cancer. Computational biology and chemistry. 2020 May 20:87():107289. doi: 10.1016/j.compbiolchem.2020.107289. Epub 2020 May 20 [PubMed PMID: 32497983]
MacFarland SP, Zelley K, Long JM, McKenna D, Mamula P, Domchek SM, Nathanson KL, Brodeur GM, Rustgi AK, Katona BW, Maxwell KN. Earlier Colorectal Cancer Screening May Be Necessary In Patients With Li-Fraumeni Syndrome. Gastroenterology. 2019 Jan:156(1):273-274. doi: 10.1053/j.gastro.2018.09.036. Epub 2018 Sep 19 [PubMed PMID: 30243621]
Mai PL, Best AF, Peters JA, DeCastro RM, Khincha PP, Loud JT, Bremer RC, Rosenberg PS, Savage SA. Risks of first and subsequent cancers among TP53 mutation carriers in the National Cancer Institute Li-Fraumeni syndrome cohort. Cancer. 2016 Dec 1:122(23):3673-3681. doi: 10.1002/cncr.30248. Epub 2016 Aug 6 [PubMed PMID: 27496084]
PDQ Cancer Genetics Editorial Board. Gastric Adenocarcinoma and Proximal Polyposis of the Stomach (GAPPS) (PDQ®): Health Professional Version. PDQ Cancer Information Summaries. 2002:(): [PubMed PMID: 38630970]
Level 3 (low-level) evidenceTacheci I, Repak R, Podhola M, Benesova L, Cyrany J, Bures J, Kohoutova D. Gastric adenocarcinoma and proximal polyposis of the stomach (GAPPS) - A Helicobacter-opposite point. Best practice & research. Clinical gastroenterology. 2021 Mar-Apr:50-51():101728. doi: 10.1016/j.bpg.2021.101728. Epub 2021 Feb 15 [PubMed PMID: 33975682]
Hoskins B, Wasuwanich P, Scheimann AO, Karnsakul W. Screening strategy for gastrointestinal and hepatopancreatobiliary cancers in cystic fibrosis. World journal of gastrointestinal oncology. 2021 Sep 15:13(9):1121-1131. doi: 10.4251/wjgo.v13.i9.1121. Epub [PubMed PMID: 34616517]
Birch RJ, Peckham D, Wood HM, Quirke P, Konstant-Hambling R, Brownlee K, Cosgriff R, Consortium GER, Burr N, Downing A. The risk of colorectal cancer in individuals with mutations of the cystic fibrosis transmembrane conductance regulator (CFTR) gene: An English population-based study. Journal of cystic fibrosis : official journal of the European Cystic Fibrosis Society. 2023 May:22(3):499-504. doi: 10.1016/j.jcf.2022.10.001. Epub 2022 Oct 15 [PubMed PMID: 36253274]
Hadjiliadis D, Khoruts A, Zauber AG, Hempstead SE, Maisonneuve P, Lowenfels AB, Cystic Fibrosis Colorectal Cancer Screening Task Force. Cystic Fibrosis Colorectal Cancer Screening Consensus Recommendations. Gastroenterology. 2018 Feb:154(3):736-745.e14. doi: 10.1053/j.gastro.2017.12.012. Epub 2017 Dec 29 [PubMed PMID: 29289528]
Level 3 (low-level) evidenceWin AK, Cleary SP, Dowty JG, Baron JA, Young JP, Buchanan DD, Southey MC, Burnett T, Parfrey PS, Green RC, Le Marchand L, Newcomb PA, Haile RW, Lindor NM, Hopper JL, Gallinger S, Jenkins MA. Cancer risks for monoallelic MUTYH mutation carriers with a family history of colorectal cancer. International journal of cancer. 2011 Nov 1:129(9):2256-62. doi: 10.1002/ijc.25870. Epub 2011 Apr 8 [PubMed PMID: 21171015]
Xiang HP, Geng XP, Ge WW, Li H. Meta-analysis of CHEK2 1100delC variant and colorectal cancer susceptibility. European journal of cancer (Oxford, England : 1990). 2011 Nov:47(17):2546-51. doi: 10.1016/j.ejca.2011.03.025. Epub 2011 Jul 30 [PubMed PMID: 21807500]
Level 1 (high-level) evidenceSriramulu S, Ramachandran M, Subramanian S, Kannan R, Gopinath M, Sollano J, Bissi L, Banerjee A, Marotta F, Pathak S. A review on role of ATM gene in hereditary transfer of colorectal cancer. Acta bio-medica : Atenei Parmensis. 2019 Jan 15:89(4):463-469. doi: 10.23750/abm.v89i4.6095. Epub 2019 Jan 15 [PubMed PMID: 30657113]
Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA: a cancer journal for clinicians. 2024 Jan-Feb:74(1):12-49. doi: 10.3322/caac.21820. Epub 2024 Jan 17 [PubMed PMID: 38230766]
Cronin KA, Scott S, Firth AU, Sung H, Henley SJ, Sherman RL, Siegel RL, Anderson RN, Kohler BA, Benard VB, Negoita S, Wiggins C, Cance WG, Jemal A. Annual report to the nation on the status of cancer, part 1: National cancer statistics. Cancer. 2022 Dec 15:128(24):4251-4284. doi: 10.1002/cncr.34479. Epub 2022 Oct 27 [PubMed PMID: 36301149]
Venugopal A, Stoffel EM. Colorectal Cancer in Young Adults. Current treatment options in gastroenterology. 2019 Mar:17(1):89-98. doi: 10.1007/s11938-019-00219-4. Epub [PubMed PMID: 30712157]
Moreira L, Balaguer F, Lindor N, de la Chapelle A, Hampel H, Aaltonen LA, Hopper JL, Le Marchand L, Gallinger S, Newcomb PA, Haile R, Thibodeau SN, Gunawardena S, Jenkins MA, Buchanan DD, Potter JD, Baron JA, Ahnen DJ, Moreno V, Andreu M, Ponz de Leon M, Rustgi AK, Castells A, EPICOLON Consortium. Identification of Lynch syndrome among patients with colorectal cancer. JAMA. 2012 Oct 17:308(15):1555-65. doi: 10.1001/jama.2012.13088. Epub [PubMed PMID: 23073952]
Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, Yen T, Stanich PP, Axell L, Patel SG. APC-Associated Polyposis Conditions. GeneReviews(®). 1993:(): [PubMed PMID: 20301519]
Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990 Jun 1:61(5):759-67 [PubMed PMID: 2188735]
Knudson AG Jr. Hereditary cancer, oncogenes, and antioncogenes. Cancer research. 1985 Apr:45(4):1437-43 [PubMed PMID: 2983882]
Chen JJ, Wang AQ, Chen QQ. DNA methylation assay for colorectal carcinoma. Cancer biology & medicine. 2017 Feb:14(1):42-49. doi: 10.20892/j.issn.2095-3941.2016.0082. Epub [PubMed PMID: 28443202]
Gupta R, Sinha S, Paul RN. The impact of microsatellite stability status in colorectal cancer. Current problems in cancer. 2018 Nov:42(6):548-559. doi: 10.1016/j.currproblcancer.2018.06.010. Epub 2018 Jul 18 [PubMed PMID: 30119911]
De' Angelis GL, Bottarelli L, Azzoni C, De' Angelis N, Leandro G, Di Mario F, Gaiani F, Negri F. Microsatellite instability in colorectal cancer. Acta bio-medica : Atenei Parmensis. 2018 Dec 17:89(9-S):97-101. doi: 10.23750/abm.v89i9-S.7960. Epub 2018 Dec 17 [PubMed PMID: 30561401]
Møller P, Seppälä TT, Bernstein I, Holinski-Feder E, Sala P, Gareth Evans D, Lindblom A, Macrae F, Blanco I, Sijmons RH, Jeffries J, Vasen HFA, Burn J, Nakken S, Hovig E, Rødland EA, Tharmaratnam K, de Vos Tot Nederveen Cappel WH, Hill J, Wijnen JT, Jenkins MA, Green K, Lalloo F, Sunde L, Mints M, Bertario L, Pineda M, Navarro M, Morak M, Renkonen-Sinisalo L, Valentin MD, Frayling IM, Plazzer JP, Pylvanainen K, Genuardi M, Mecklin JP, Moeslein G, Sampson JR, Capella G, Mallorca Group. Cancer risk and survival in path_MMR carriers by gene and gender up to 75 years of age: a report from the Prospective Lynch Syndrome Database. Gut. 2018 Jul:67(7):1306-1316. doi: 10.1136/gutjnl-2017-314057. Epub 2017 Jul 28 [PubMed PMID: 28754778]
Pearlman R, Frankel WL, Swanson BJ, Jones D, Zhao W, Yilmaz A, Miller K, Bacher J, Bigley C, Nelsen L, Goodfellow PJ, Goldberg RM, Paskett E, Shields PG, Freudenheim JL, Stanich PP, Lattimer I, Arnold M, Prior TW, Haut M, Kalady MF, Heald B, Paquette I, Draper DJ, Brell JM, Mahesh S, Weeman K, Bastola S, Zangmeister J, Gowda A, Kencana F, Malcolm A, Liu Y, Cole S, Bane C, Li C, Rehmus E, Pritchard CC, Shirts BH, Jacobson A, Cummings SA, de la Chapelle A, Hampel H. Prospective Statewide Study of Universal Screening for Hereditary Colorectal Cancer: The Ohio Colorectal Cancer Prevention Initiative. JCO precision oncology. 2021:5():. pii: PO.20.00525. doi: 10.1200/PO.20.00525. Epub 2021 May 5 [PubMed PMID: 34250417]
Vacante M, Borzì AM, Basile F, Biondi A. Biomarkers in colorectal cancer: Current clinical utility and future perspectives. World journal of clinical cases. 2018 Dec 6:6(15):869-881. doi: 10.12998/wjcc.v6.i15.869. Epub [PubMed PMID: 30568941]
Level 3 (low-level) evidenceBallester V, Rashtak S, Boardman L. Clinical and molecular features of young-onset colorectal cancer. World journal of gastroenterology. 2016 Feb 7:22(5):1736-44. doi: 10.3748/wjg.v22.i5.1736. Epub [PubMed PMID: 26855533]
Li H, Zhang L, Jiang Q, Shi Z, Tong H. Identification a nonsense mutation of APC gene in Chinese patients with familial adenomatous polyposis. Experimental and therapeutic medicine. 2017 Apr:13(4):1495-1499. doi: 10.3892/etm.2017.4122. Epub 2017 Feb 14 [PubMed PMID: 28413499]
Abbaszadegan MR, Moghbeli M. Genetic and molecular origins of colorectal Cancer among the Iranians: an update. Diagnostic pathology. 2018 Dec 22:13(1):97. doi: 10.1186/s13000-018-0774-0. Epub 2018 Dec 22 [PubMed PMID: 30579343]
Markowitz SD, Bertagnolli MM. Molecular origins of cancer: Molecular basis of colorectal cancer. The New England journal of medicine. 2009 Dec 17:361(25):2449-60. doi: 10.1056/NEJMra0804588. Epub [PubMed PMID: 20018966]
Issa JP. CpG island methylator phenotype in cancer. Nature reviews. Cancer. 2004 Dec:4(12):988-93. doi: 10.1038/nrc1507. Epub [PubMed PMID: 15573120]
Terry MB, Neugut AI, Bostick RM, Sandler RS, Haile RW, Jacobson JS, Fenoglio-Preiser CM, Potter JD. Risk factors for advanced colorectal adenomas: a pooled analysis. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2002 Jul:11(7):622-9 [PubMed PMID: 12101109]
Moufid FZ, Bouguenouch L, El Bouchikhi I, Chbani L, Iraqui Houssaini M, Sekal M, Belhassan K, Bennani B, Ouldim K. The First Molecular Screening of MLH1 and MSH2 Genes in Moroccan Colorectal Cancer Patients Shows a Relatively High Mutational Prevalence. Genetic testing and molecular biomarkers. 2018 Aug:22(8):492-497. doi: 10.1089/gtmb.2018.0067. Epub 2018 Jul 25 [PubMed PMID: 30044143]
González ML, Causada-Calo N, Santino JP, Dominguez-Valentin M, Ferro FA, Sammartino I, Kalfayan PG, Verzura MA, Piñero TA, Cajal AR, Pavicic W, Vaccaro C. Universal determination of microsatellite instability using BAT26 as a single marker in an Argentine colorectal cancer cohort. Familial cancer. 2018 Jul:17(3):395-402. doi: 10.1007/s10689-017-0052-4. Epub [PubMed PMID: 29128931]
Hatfield E, Green JS, Woods MO, Warden G, Parfrey PS. Impact of colonoscopic screening in Familial Colorectal Cancer Type X. Molecular genetics & genomic medicine. 2018 Nov:6(6):1021-1030. doi: 10.1002/mgg3.478. Epub 2018 Oct 9 [PubMed PMID: 30300963]
Giardiello FM, Allen JI, Axilbund JE, Boland CR, Burke CA, Burt RW, Church JM, Dominitz JA, Johnson DA, Kaltenbach T, Levin TR, Lieberman DA, Robertson DJ, Syngal S, Rex DK, US Multi-Society Task Force on Colorectal Cancer. Guidelines on genetic evaluation and management of Lynch syndrome: a consensus statement by the US Multi-Society Task Force on colorectal cancer. Gastroenterology. 2014 Aug:147(2):502-26. doi: 10.1053/j.gastro.2014.04.001. Epub [PubMed PMID: 25043945]
Level 3 (low-level) evidenceGiardiello FM, Allen JI, Axilbund JE, Boland CR, Burke CA, Burt RW, Church JM, Dominitz JA, Johnson DA, Kaltenbach T, Levin TR, Lieberman DA, Robertson DJ, Syngal S, Rex DK, American Society for Gastrointestinal Endoscopy. Guidelines on genetic evaluation and management of Lynch syndrome: a consensus statement by the U.S. Multi-Society Task Force on Colorectal Cancer. Gastrointestinal endoscopy. 2014 Aug:80(2):197-220. doi: 10.1016/j.gie.2014.06.006. Epub [PubMed PMID: 25034835]
Level 3 (low-level) evidenceArakawa K, Hata K, Kawai K, Tanaka T, Nishikawa T, Sasaki K, Shuno Y, Kaneko M, Hiyoshi M, Emoto S, Murono K, Nozawa H. Predictors for High Microsatellite Instability in Patients with Colorectal Cancer Fulfilling the Revised Bethesda Guidelines. Anticancer research. 2018 Aug:38(8):4871-4876. doi: 10.21873/anticanres.12800. Epub [PubMed PMID: 30061262]
Coughlin SE, Heald B, Clark DF, Nielsen SM, Hatchell KE, Esplin ED, Katona BW. Multigene Panel Testing Yields High Rates of Clinically Actionable Variants Among Patients With Colorectal Cancer. JCO precision oncology. 2022 Nov:6():e2200517. doi: 10.1200/PO.22.00517. Epub [PubMed PMID: 36370464]
Taieb J, Svrcek M, Cohen R, Basile D, Tougeron D, Phelip JM. Deficient mismatch repair/microsatellite unstable colorectal cancer: Diagnosis, prognosis and treatment. European journal of cancer (Oxford, England : 1990). 2022 Nov:175():136-157. doi: 10.1016/j.ejca.2022.07.020. Epub 2022 Sep 14 [PubMed PMID: 36115290]
Morin PJ. Colorectal cancer: the APC-lncRNA link. The Journal of clinical investigation. 2019 Feb 1:129(2):503-505. doi: 10.1172/JCI125985. Epub 2019 Jan 14 [PubMed PMID: 30640173]
Trotter J, Pantel AR, Teo BK, Escorcia FE, Li T, Pryma DA, Taunk NK. Positron Emission Tomography (PET)/Computed Tomography (CT) Imaging in Radiation Therapy Treatment Planning: A Review of PET Imaging Tracers and Methods to Incorporate PET/CT. Advances in radiation oncology. 2023 Sep-Oct:8(5):101212. doi: 10.1016/j.adro.2023.101212. Epub 2023 Mar 27 [PubMed PMID: 37197709]
Level 3 (low-level) evidenceAdar T, Rodgers LH, Shannon KM, Yoshida M, Ma T, Mattia A, Lauwers GY, Iafrate AJ, Hartford NM, Oliva E, Chung DC. Universal screening of both endometrial and colon cancers increases the detection of Lynch syndrome. Cancer. 2018 Aug 1:124(15):3145-3153. doi: 10.1002/cncr.31534. Epub 2018 May 11 [PubMed PMID: 29750335]
Zhang J, Li Z, Huang X, Ye J. [Clinical and molecular characteristics of a child with familial adenomatous polyposis]. Zhonghua er ke za zhi = Chinese journal of pediatrics. 2016 Mar:54(3):205-8. doi: 10.3760/cma.j.issn.0578-1310.2016.03.010. Epub [PubMed PMID: 26957067]
Giang H, Nguyen VT, Nguyen SD, Nguyen HP, Vo BT, Nguyen TM, Nguyen NH, Truong KD, Do TT, Phan MD, Nguyen HN. Detection of a heterozygous germline APC mutation in a three-generation family with familial adenomatous polyposis using targeted massive parallel sequencing in Vietnam. BMC medical genetics. 2018 Oct 19:19(1):188. doi: 10.1186/s12881-018-0701-y. Epub 2018 Oct 19 [PubMed PMID: 30340471]
Sample DC, Samadder NJ, Pappas LM, Boucher KM, Samowitz WS, Berry T, Westover M, Nathan D, Kanth P, Byrne KR, Burt RW, Neklason DW. Variables affecting penetrance of gastric and duodenal phenotype in familial adenomatous polyposis patients. BMC gastroenterology. 2018 Jul 16:18(1):115. doi: 10.1186/s12876-018-0841-8. Epub 2018 Jul 16 [PubMed PMID: 30012100]
Kallenberg FGJ, Bastiaansen BAJ, Nio CY, Soeters MR, Boermeester MA, Aalfs CM, Bossuyt PMM, Dekker E. Adrenal Lesions in Patients With (Attenuated) Familial Adenomatous Polyposis and MUTYH-Associated Polyposis. Diseases of the colon and rectum. 2017 Oct:60(10):1057-1064. doi: 10.1097/DCR.0000000000000809. Epub [PubMed PMID: 28891849]
Klančar G, Blatnik A, Šetrajčič Dragoš V, Vogrič V, Stegel V, Blatnik O, Drev P, Gazič B, Krajc M, Novaković S. A Novel Germline MLH1 In-Frame Deletion in a Slovenian Lynch Syndrome Family Associated with Uncommon Isolated PMS2 Loss in Tumor Tissue. Genes. 2020 Mar 18:11(3):. doi: 10.3390/genes11030325. Epub 2020 Mar 18 [PubMed PMID: 32197529]
Mittendorf KF, Ukaegbu C, Gilmore MJ, Lindberg NM, Kauffman TL, Eubanks DJ, Shuster E, Allen J, McMullen C, Feigelson HS, Anderson KP, Leo MC, Hunter JE, Sasaki SO, Zepp JM, Syngal S, Wilfond BS, Goddard KAB. Adaptation and early implementation of the PREdiction model for gene mutations (PREMM(5)™) for lynch syndrome risk assessment in a diverse population. Familial cancer. 2022 Apr:21(2):167-180. doi: 10.1007/s10689-021-00243-3. Epub 2021 Mar 23 [PubMed PMID: 33754278]
Kastrinos F, Uno H, Ukaegbu C, Alvero C, McFarland A, Yurgelun MB, Kulke MH, Schrag D, Meyerhardt JA, Fuchs CS, Mayer RJ, Ng K, Steyerberg EW, Syngal S. Development and Validation of the PREMM(5) Model for Comprehensive Risk Assessment of Lynch Syndrome. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2017 Jul 1:35(19):2165-2172. doi: 10.1200/JCO.2016.69.6120. Epub 2017 May 10 [PubMed PMID: 28489507]
Level 1 (high-level) evidenceCox VL, Saeed Bamashmos AA, Foo WC, Gupta S, Yedururi S, Garg N, Kang HC. Lynch Syndrome: Genomics Update and Imaging Review. Radiographics : a review publication of the Radiological Society of North America, Inc. 2018 Mar-Apr:38(2):483-499. doi: 10.1148/rg.2018170075. Epub [PubMed PMID: 29528821]
Walkowska J, Kallemose T, Jönsson G, Jönsson M, Andersen O, Andersen MH, Svane IM, Langkilde A, Nilbert M, Therkildsen C. Immunoprofiles of colorectal cancer from Lynch syndrome. Oncoimmunology. 2019:8(1):e1515612. doi: 10.1080/2162402X.2018.1515612. Epub 2018 Sep 26 [PubMed PMID: 30546958]
McGee SF, AlGhareeb W, Ahmad CH, Armstrong D, Babak S, Berry S, Biagi J, Booth C, Bossé D, Champion P, Colwell B, Finn N, Goel R, Gray S, Green J, Harb M, Hyde A, Jeyakumar A, Jonker D, Kanagaratnam S, Kavan P, MacMillan A, Muinuddin A, Patil N, Porter G, Powell E, Ramjeesingh R, Raza M, Rorke S, Seal M, Servidio-Italiano F, Siddiqui J, Simms J, Smithson L, Snow S, St-Hilaire E, Stuckless T, Tate A, Tehfe M, Thirlwell M, Tsvetkova E, Valdes M, Vickers M, Virik K, Welch S, Marginean C, Asmis T. Eastern Canadian Colorectal Cancer Consensus Conference 2017. Current oncology (Toronto, Ont.). 2018 Aug:25(4):262-274. doi: 10.3747/co.25.4083. Epub 2018 Aug 14 [PubMed PMID: 30111967]
Level 3 (low-level) evidenceMocellin S, Baretta Z, Roqué I Figuls M, Solà I, Martin-Richard M, Hallum S, Bonfill Cosp X. Second-line systemic therapy for metastatic colorectal cancer. The Cochrane database of systematic reviews. 2017 Jan 27:1(1):CD006875. doi: 10.1002/14651858.CD006875.pub3. Epub 2017 Jan 27 [PubMed PMID: 28128439]
Level 1 (high-level) evidenceHurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S, Holmgren E, Ferrara N, Fyfe G, Rogers B, Ross R, Kabbinavar F. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. The New England journal of medicine. 2004 Jun 3:350(23):2335-42 [PubMed PMID: 15175435]
Petrelli F, Coinu A, Ghilardi M, Cabiddu M, Zaniboni A, Barni S. Efficacy of oxaliplatin-based chemotherapy + bevacizumab as first-line treatment for advanced colorectal cancer: a systematic review and pooled analysis of published trials. American journal of clinical oncology. 2015 Apr:38(2):227-33. doi: 10.1097/COC.0b013e3182a2d7b8. Epub [PubMed PMID: 25806713]
Level 1 (high-level) evidenceSnyder M, Bottiglieri S, Almhanna K. Impact of Primary Tumor Location on First-line Bevacizumab or Cetuximab in Metastatic Colorectal Cancer. Reviews on recent clinical trials. 2018:13(2):139-149. doi: 10.2174/1574887113666180328104109. Epub [PubMed PMID: 29595113]
Chen D, Li L, Zhang X, Gao G, Shen L, Hu J, Yang M, Liu B, Qian X. FOLFOX plus anti-epidermal growth factor receptor (EGFR) monoclonal antibody (mAb) is an effective first-line treatment for patients with RAS-wild left-sided metastatic colorectal cancer: A meta-analysis. Medicine. 2018 Mar:97(10):e0097. doi: 10.1097/MD.0000000000010097. Epub [PubMed PMID: 29517682]
Level 2 (mid-level) evidenceKopetz S, Yoshino T, Van Cutsem E, Eng C, Kim TW, Wasan HS, Desai J, Ciardiello F, Yaeger R, Maughan TS, Beyzarov E, Zhang X, Ferrier G, Zhang X, Tabernero J. Encorafenib, cetuximab and chemotherapy in BRAF-mutant colorectal cancer: a randomized phase 3 trial. Nature medicine. 2025 Mar:31(3):901-908. doi: 10.1038/s41591-024-03443-3. Epub 2025 Jan 25 [PubMed PMID: 39863775]
Level 1 (high-level) evidenceCaux F, Plauchu H, Chibon F, Faivre L, Fain O, Vabres P, Bonnet F, Selma ZB, Laroche L, Gérard M, Longy M. Segmental overgrowth, lipomatosis, arteriovenous malformation and epidermal nevus (SOLAMEN) syndrome is related to mosaic PTEN nullizygosity. European journal of human genetics : EJHG. 2007 Jul:15(7):767-73 [PubMed PMID: 17392703]
Level 3 (low-level) evidenceWon HS, Chang ED, Na SJ, Whang IY, Lee DS, You SH, Kim YS, Kim JS. PTEN Mutation Identified in Patient Diagnosed with Simultaneous Multiple Cancers. Cancer research and treatment. 2019 Jan:51(1):402-407. doi: 10.4143/crt.2017.579. Epub 2018 Feb 27 [PubMed PMID: 29510612]
Pal R, Bhattacharjee R, Bhansali A. Protean manifestations of Proteus syndrome. Postgraduate medical journal. 2018 Jul:94(1113):416. doi: 10.1136/postgradmedj-2018-135731. Epub 2018 Apr 6 [PubMed PMID: 29626058]
Tian X, Wang Q, Cai W. A Novel Mutation in MYH Gene Associated with Aggressive Colorectal Cancer in a Child: A Case Report and Review of Literature. OncoTargets and therapy. 2020:13():8557-8565. doi: 10.2147/OTT.S259587. Epub 2020 Aug 25 [PubMed PMID: 32904697]
Level 3 (low-level) evidenceMaeda T, Cannom RR, Beart RW Jr, Etzioni DA. Decision model of segmental compared with total abdominal colectomy for colon cancer in hereditary nonpolyposis colorectal cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2010 Mar 1:28(7):1175-80. doi: 10.1200/JCO.2009.25.9812. Epub 2010 Feb 1 [PubMed PMID: 20124166]
Schneider R, Schneider C, Büttner R, Reinacher-Schick A, Tannapfel A, Fürst A, Rüschoff J, Jakobeit C, Royer-Pokora B, Möslein G. [Colorectal Carcinoma with Suspected Lynch Syndrome: A Multidisciplinary Algorithm]. Zentralblatt fur Chirurgie. 2015 Dec:140(6):591-9. doi: 10.1055/s-0034-1368480. Epub 2014 Nov 5 [PubMed PMID: 25372301]
Herzig DO, Buie WD, Weiser MR, You YN, Rafferty JF, Feingold D, Steele SR. Clinical Practice Guidelines for the Surgical Treatment of Patients With Lynch Syndrome. Diseases of the colon and rectum. 2017 Feb:60(2):137-143. doi: 10.1097/DCR.0000000000000785. Epub [PubMed PMID: 28059909]
Level 1 (high-level) evidenceDominguez-Valentin M, Seppälä TT, Engel C, Aretz S, Macrae F, Winship I, Capella G, Thomas H, Hovig E, Nielsen M, Sijmons RH, Bertario L, Bonanni B, Tibiletti MG, Cavestro GM, Mints M, Gluck N, Katz L, Heinimann K, Vaccaro CA, Green K, Lalloo F, Hill J, Schmiegel W, Vangala D, Perne C, Strauß HG, Tecklenburg J, Holinski-Feder E, Steinke-Lange V, Mecklin JP, Plazzer JP, Pineda M, Navarro M, Vidal JB, Kariv R, Rosner G, Piñero TA, Gonzalez ML, Kalfayan P, Sampson JR, Ryan NAJ, Evans DG, Møller P, Crosbie EJ. Risk-Reducing Gynecological Surgery in Lynch Syndrome: Results of an International Survey from the Prospective Lynch Syndrome Database. Journal of clinical medicine. 2020 Jul 18:9(7):. doi: 10.3390/jcm9072290. Epub 2020 Jul 18 [PubMed PMID: 32708519]
Level 2 (mid-level) evidenceKalady MF, Church JM. Prophylactic colectomy: Rationale, indications, and approach. Journal of surgical oncology. 2015 Jan:111(1):112-7. doi: 10.1002/jso.23820. Epub 2014 Nov 21 [PubMed PMID: 25418116]
Yamadera M, Ueno H, Kobayashi H, Konishi T, Ishida F, Yamaguchi T, Hinoi T, Inoue Y, Kanemitsu Y, Tomita N, Ishida H, Sugihara K. Current status of prophylactic surgical treatment for familial adenomatous polyposis in Japan. Surgery today. 2017 Jun:47(6):690-696. doi: 10.1007/s00595-016-1431-4. Epub 2016 Oct 21 [PubMed PMID: 27770209]
Kennelly RP, Gryfe R, Winter DC. Familial colorectal cancer: Patient assessment, surveillance and surgical management. European journal of surgical oncology : the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology. 2017 Feb:43(2):294-302. doi: 10.1016/j.ejso.2016.07.008. Epub 2016 Jul 27 [PubMed PMID: 27546013]
Wang X, Yu Y, Meng W, Jiang D, Deng X, Wu B, Zhuang H, Wang C, Shen Y, Yang L, Zhu H, Cheng K, Zhao Y, Li Z, Qiu M, Gou H, Bi F, Xu F, Zhong R, Bai S, Wang Z, Zhou Z. Total neoadjuvant treatment (CAPOX plus radiotherapy) for patients with locally advanced rectal cancer with high risk factors: A phase 2 trial. Radiotherapy and oncology : journal of the European Society for Therapeutic Radiology and Oncology. 2018 Nov:129(2):300-305. doi: 10.1016/j.radonc.2018.08.027. Epub 2018 Oct 28 [PubMed PMID: 30381141]
Level 2 (mid-level) evidenceYu J, Li N, Tang Y, Wang X, Tang Y, Wang SL, Song YW, Liu YP, Li YX, Jin J. Outcomes after hypofractionated stereotactic radiotherapy for colorectal cancer oligometastases. Journal of surgical oncology. 2019 Mar:119(4):532-538. doi: 10.1002/jso.25361. Epub 2019 Jan 4 [PubMed PMID: 30609038]
Oneda E, Zaniboni A. Adjuvant treatment of colon cancer with microsatellite instability - the state of the art. Critical reviews in oncology/hematology. 2022 Jan:169():103537. doi: 10.1016/j.critrevonc.2021.103537. Epub 2021 Nov 18 [PubMed PMID: 34801698]
Sargent DJ, Marsoni S, Monges G, Thibodeau SN, Labianca R, Hamilton SR, French AJ, Kabat B, Foster NR, Torri V, Ribic C, Grothey A, Moore M, Zaniboni A, Seitz JF, Sinicrope F, Gallinger S. Defective mismatch repair as a predictive marker for lack of efficacy of fluorouracil-based adjuvant therapy in colon cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2010 Jul 10:28(20):3219-26. doi: 10.1200/JCO.2009.27.1825. Epub 2010 May 24 [PubMed PMID: 20498393]
André T, Shiu KK, Kim TW, Jensen BV, Jensen LH, Punt CJA, Smith D, Garcia-Carbonero R, Alcaide-Garcia J, Gibbs P, de la Fouchardiere C, Rivera F, Elez E, Le DT, Yoshino T, Zuo Y, Fogelman D, Adelberg D, Diaz LA. Pembrolizumab versus chemotherapy in microsatellite instability-high or mismatch repair-deficient metastatic colorectal cancer: 5-year follow-up from the randomized phase III KEYNOTE-177 study. Annals of oncology : official journal of the European Society for Medical Oncology. 2025 Mar:36(3):277-284. doi: 10.1016/j.annonc.2024.11.012. Epub 2024 Dec 2 [PubMed PMID: 39631622]
Level 1 (high-level) evidenceLe DT, Uram JN, Wang H, Bartlett BR, Kemberling H, Eyring AD, Skora AD, Luber BS, Azad NS, Laheru D, Biedrzycki B, Donehower RC, Zaheer A, Fisher GA, Crocenzi TS, Lee JJ, Duffy SM, Goldberg RM, de la Chapelle A, Koshiji M, Bhaijee F, Huebner T, Hruban RH, Wood LD, Cuka N, Pardoll DM, Papadopoulos N, Kinzler KW, Zhou S, Cornish TC, Taube JM, Anders RA, Eshleman JR, Vogelstein B, Diaz LA Jr. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. The New England journal of medicine. 2015 Jun 25:372(26):2509-20. doi: 10.1056/NEJMoa1500596. Epub 2015 May 30 [PubMed PMID: 26028255]
Overman MJ, Lonardi S, Wong KYM, Lenz HJ, Gelsomino F, Aglietta M, Morse MA, Van Cutsem E, McDermott R, Hill A, Sawyer MB, Hendlisz A, Neyns B, Svrcek M, Moss RA, Ledeine JM, Cao ZA, Kamble S, Kopetz S, André T. Durable Clinical Benefit With Nivolumab Plus Ipilimumab in DNA Mismatch Repair-Deficient/Microsatellite Instability-High Metastatic Colorectal Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2018 Mar 10:36(8):773-779. doi: 10.1200/JCO.2017.76.9901. Epub 2018 Jan 20 [PubMed PMID: 29355075]
Chalabi M, Fanchi LF, Dijkstra KK, Van den Berg JG, Aalbers AG, Sikorska K, Lopez-Yurda M, Grootscholten C, Beets GL, Snaebjornsson P, Maas M, Mertz M, Veninga V, Bounova G, Broeks A, Beets-Tan RG, de Wijkerslooth TR, van Lent AU, Marsman HA, Nuijten E, Kok NF, Kuiper M, Verbeek WH, Kok M, Van Leerdam ME, Schumacher TN, Voest EE, Haanen JB. Neoadjuvant immunotherapy leads to pathological responses in MMR-proficient and MMR-deficient early-stage colon cancers. Nature medicine. 2020 Apr:26(4):566-576. doi: 10.1038/s41591-020-0805-8. Epub 2020 Apr 6 [PubMed PMID: 32251400]
Chalabi M, Verschoor YL, Tan PB, Balduzzi S, Van Lent AU, Grootscholten C, Dokter S, Büller NV, Grotenhuis BA, Kuhlmann K, Burger JW, Huibregtse IL, Aukema TS, Hendriks ER, Oosterling SJ, Snaebjornsson P, Voest EE, Wessels LF, Beets-Tan RG, Van Leerdam ME, Schumacher TN, van den Berg JG, Beets GL, Haanen JB. Neoadjuvant Immunotherapy in Locally Advanced Mismatch Repair-Deficient Colon Cancer. The New England journal of medicine. 2024 Jun 6:390(21):1949-1958. doi: 10.1056/NEJMoa2400634. Epub [PubMed PMID: 38838311]
Lee SY, Kim DW, Kang SI, Ihn MH, Oh HK, Kang SB, Kim CH, Kim HR, Kim YJ, Ju JK. Impact of Family History on Prognosis of Patients with Sporadic Colorectal Cancer. Annals of surgical oncology. 2019 Apr:26(4):1118-1126. doi: 10.1245/s10434-019-07179-0. Epub 2019 Jan 25 [PubMed PMID: 30684157]
Thong MSY, Koch-Gallenkamp L, Jansen L, Bertram H, Eberle A, Holleczek B, Waldeyer-Sauerland M, Waldmann A, Zeissig SR, Brenner H, Arndt V. Age-specific health-related quality of life in long-term and very long-term colorectal cancer survivors versus population controls - a population-based study(). Acta oncologica (Stockholm, Sweden). 2019 May:58(5):801-810. doi: 10.1080/0284186X.2018.1557340. Epub 2019 Feb 8 [PubMed PMID: 30736716]
Level 2 (mid-level) evidenceVanaclocha-Espi M, Ibáñez J, Molina-Barceló A, Valverde-Roig MJ, Pérez E, Nolasco A, de la Vega M, de la Lastra-Bosch ID, Oceja ME, Espinàs JA, Font R, Pérez-Riquelme F, Arana-Arri E, Portillo I, Salas D, CRIBEA Group. Risk factors for severe complications of colonoscopy in screening programs. Preventive medicine. 2019 Jan:118():304-308. doi: 10.1016/j.ypmed.2018.11.010. Epub 2018 Nov 8 [PubMed PMID: 30414944]
Carlomagno N, Santangelo ML, Amato B, Calogero A, Saracco M, Cremone C, Miranda A, Dodaro C, Renda A. Total colectomy for cancer: analysis of factors linked to patients' age. International journal of surgery (London, England). 2014:12 Suppl 2():S135-S139. doi: 10.1016/j.ijsu.2014.08.363. Epub 2014 Aug 30 [PubMed PMID: 25181631]
Level 2 (mid-level) evidencePatel SG, Ahnen DJ. Colorectal Cancer in the Young. Current gastroenterology reports. 2018 Mar 28:20(4):15. doi: 10.1007/s11894-018-0618-9. Epub 2018 Mar 28 [PubMed PMID: 29616330]
Nimptsch K, Wu K. Is Timing Important? The Role of Diet and Lifestyle during Early Life on Colorectal Neoplasia. Current colorectal cancer reports. 2018 Feb:14(1):1-11. doi: 10.1007/s11888-018-0396-7. Epub 2018 Jan 18 [PubMed PMID: 30140177]
LaDuca H, McFarland R, Gutierrez S, Yussuf A, Ho N, Pepper J, Reineke P, Cain T, Blanco K, Horton C, Dolinsky JS. Quality of Clinician-Reported Cancer History When Ordering Genetic Testing. JCO clinical cancer informatics. 2018 Dec:2():1-11. doi: 10.1200/CCI.18.00014. Epub [PubMed PMID: 30652589]
Level 2 (mid-level) evidenceMauri G, Sartore-Bianchi A, Russo AG, Marsoni S, Bardelli A, Siena S. Early-onset colorectal cancer in young individuals. Molecular oncology. 2019 Feb:13(2):109-131. doi: 10.1002/1878-0261.12417. Epub 2018 Dec 22 [PubMed PMID: 30520562]
Weinberg BA, Marshall JL. Colon Cancer in Young Adults: Trends and Their Implications. Current oncology reports. 2019 Jan 18:21(1):3. doi: 10.1007/s11912-019-0756-8. Epub 2019 Jan 18 [PubMed PMID: 30659375]
Yurgelun MB, Hampel H. Recent Advances in Lynch Syndrome: Diagnosis, Treatment, and Cancer Prevention. American Society of Clinical Oncology educational book. American Society of Clinical Oncology. Annual Meeting. 2018 May 23:38():101-109. doi: 10.1200/EDBK_208341. Epub [PubMed PMID: 30231390]
Level 3 (low-level) evidenceBiller LH, Syngal S, Yurgelun MB. Recent advances in Lynch syndrome. Familial cancer. 2019 Apr:18(2):211-219. doi: 10.1007/s10689-018-00117-1. Epub [PubMed PMID: 30627969]
Level 3 (low-level) evidenceBarrow P, Khan M, Lalloo F, Evans DG, Hill J. Systematic review of the impact of registration and screening on colorectal cancer incidence and mortality in familial adenomatous polyposis and Lynch syndrome. The British journal of surgery. 2013 Dec:100(13):1719-31. doi: 10.1002/bjs.9316. Epub [PubMed PMID: 24227356]
Level 1 (high-level) evidenceCoelho H, Jones-Hughes T, Snowsill T, Briscoe S, Huxley N, Frayling IM, Hyde C. A systematic review of test accuracy studies evaluating molecular micro-satellite instability testing for the detection of individuals with lynch syndrome. BMC cancer. 2017 Dec 8:17(1):836. doi: 10.1186/s12885-017-3820-5. Epub 2017 Dec 8 [PubMed PMID: 29221446]
Level 1 (high-level) evidenceLiang J, Lin C, Hu F, Wang F, Zhu L, Yao X, Wang Y, Zhao Y. APC polymorphisms and the risk of colorectal neoplasia: a HuGE review and meta-analysis. American journal of epidemiology. 2013 Jun 1:177(11):1169-79. doi: 10.1093/aje/kws382. Epub 2013 Apr 10 [PubMed PMID: 23576677]
Level 1 (high-level) evidenceLiang TJ, Wang HX, Zheng YY, Cao YQ, Wu X, Zhou X, Dong SX. APC hypermethylation for early diagnosis of colorectal cancer: a meta-analysis and literature review. Oncotarget. 2017 Jul 11:8(28):46468-46479. doi: 10.18632/oncotarget.17576. Epub [PubMed PMID: 28515349]
Level 1 (high-level) evidenceWakeman C, Keenan J, Eteuati J, Hollington P, Eglinton T, Frizelle F. Chemoprevention of colorectal neoplasia. ANZ journal of surgery. 2017 Dec:87(12):E228-E232. doi: 10.1111/ans.13392. Epub 2015 Dec 21 [PubMed PMID: 26686322]
Dicks E, Pullman D, Kao K, MacMillan A, Simmonds C, Etchegary H. Universal tumor screening for Lynch syndrome: perspectives of Canadian pathologists and genetic counselors. Journal of community genetics. 2019 Jul:10(3):335-344. doi: 10.1007/s12687-018-0398-9. Epub 2018 Nov 21 [PubMed PMID: 30465127]
Level 3 (low-level) evidenceGaitanidis A, Spathakis M, Tsalikidis C, Alevizakos M, Tsaroucha A, Pitiakoudis M. Risk factors for cardiovascular mortality in patients with colorectal cancer: a population-based study. International journal of clinical oncology. 2019 May:24(5):501-507. doi: 10.1007/s10147-018-01382-x. Epub 2019 Jan 2 [PubMed PMID: 30604158]
Wang J, Luo L, Wang D, Guo B, Li J, Yang Z, Tang D. Combination adjuvant chemotherapy with targeted drugs for treatment of colorectal cancer: A network meta-analysis. Journal of cellular biochemistry. 2018 Feb:119(2):1521-1537. doi: 10.1002/jcb.26312. Epub 2017 Nov 24 [PubMed PMID: 28771807]
Level 1 (high-level) evidenceda Silva WC, de Araujo VE, Lima EMEA, Dos Santos JBR, Silva MRRD, Almeida PHRF, de Assis Acurcio F, Godman B, Kurdi A, Cherchiglia ML, Andrade EIG. Comparative Effectiveness and Safety of Monoclonal Antibodies (Bevacizumab, Cetuximab, and Panitumumab) in Combination with Chemotherapy for Metastatic Colorectal Cancer: A Systematic Review and Meta-Analysis. BioDrugs : clinical immunotherapeutics, biopharmaceuticals and gene therapy. 2018 Dec:32(6):585-606. doi: 10.1007/s40259-018-0322-1. Epub [PubMed PMID: 30499082]
Level 2 (mid-level) evidenceJayedi A, Emadi A, Shab-Bidar S. Dietary Inflammatory Index and Site-Specific Cancer Risk: A Systematic Review and Dose-Response Meta-Analysis. Advances in nutrition (Bethesda, Md.). 2018 Jul 1:9(4):388-403. doi: 10.1093/advances/nmy015. Epub [PubMed PMID: 30032224]
Level 3 (low-level) evidenceMa Y, Hu M, Zhou L, Ling S, Li Y, Kong B, Huang P. Dietary fiber intake and risks of proximal and distal colon cancers: A meta-analysis. Medicine. 2018 Sep:97(36):e11678. doi: 10.1097/MD.0000000000011678. Epub [PubMed PMID: 30200062]
Level 1 (high-level) evidenceGianfredi V, Salvatori T, Villarini M, Moretti M, Nucci D, Realdon S. Is dietary fibre truly protective against colon cancer? A systematic review and meta-analysis. International journal of food sciences and nutrition. 2018 Dec:69(8):904-915. doi: 10.1080/09637486.2018.1446917. Epub 2018 Mar 8 [PubMed PMID: 29516760]
Level 1 (high-level) evidence