Introduction
Electrophysiology (EP) has become an essential cardiology subspecialty in recent decades.[1] Detailed electrophysiologic studies (EPS) have provided researchers and clinicians with insights into the mechanisms of cardiac arrhythmias, leading to the development of very effective ablative therapies. EPS represents a precise method for identifying the origin and mechanism of arrhythmias by mapping the heart's electrical activity with intracardiac electrodes.[2] Therefore, EPS are particularly valuable in evaluating patients with unexplained syncope, assessing the risk of sudden cardiac death, and determining the need for interventions such as catheter ablation or implantable devices (eg, pacemakers or defibrillators).
This approach is essential for personalized arrhythmia management, as it improves diagnostic accuracy and optimizes treatment strategies to enhance patient outcomes. The performance and interpretation of an EPS present a considerable challenge due to the technical demands inherent in the procedure and the sophisticated analytical approach required to integrate data derived from intracardiac electrograms, surface electrocardiograms (ECGs), and, in some instances, advanced imaging modalities. This review offers a concise introduction to the fundamental findings observed in EPS and examines the role of EPS interpretation in diagnosing and managing prevalent arrhythmias.
Anatomy and Physiology
Register For Free And Read The Full Article
Search engine and full access to all medical articles
10 free questions in your specialty
Free CME/CE Activities
Free daily question in your email
Save favorite articles to your dashboard
Emails offering discounts
Learn more about a Subscription to StatPearls Point-of-Care
Anatomy and Physiology
The EPS starts with the placement of bipolar or multipolar catheters in the heart for the acquisition of intracardiac electrical signals. These signals are properly amplified, filtered, digitized, and finally displayed along with standard ECG recordings. The intracardiac electrograms (EGMs) are broadly divided into 2 types: “unipolar” and “bipolar.” A unipolar signal is the recording of the voltage difference between an intracardiac (cathode, located at the tip of the catheter) and an extracardiac (anode, usually in the inferior vena cava) electrode. On the contrary, bipolar signals are electrical activities recorded from one of the close-spaced (2-3 mm apart) electrode pairs of an electrophysiologic catheter within the heart cavity.
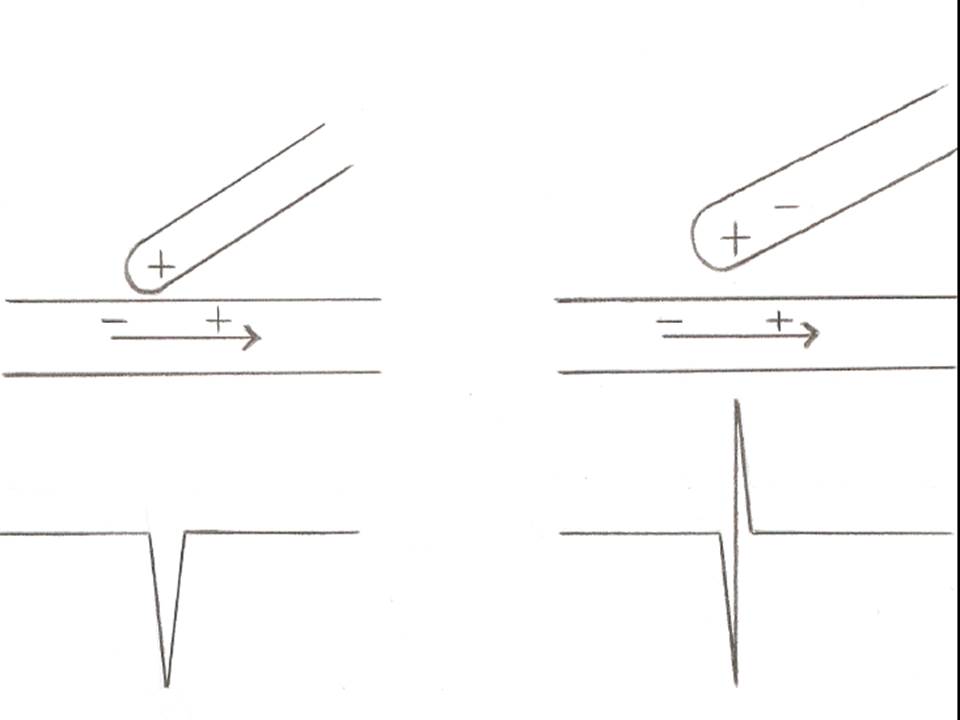
In EPS, bipolar recordings are most commonly used because they are far less susceptible to sensing signals originating far away from the recording site (far-field signals) compared to unipolar recordings, which display a voltage difference over a considerable distance. However, unipolar recordings are instrumental when clinicians want to locate and ablate an arrhythmogenic focus accurately. The reason is that a unipolar electrode records only 1 electrical impulse, which passes through the point of contact with cardiac tissue. In contrast, a bipolar electrode displays the summation of 2 recordings, each from the proximal and the distal pole of a pair of electrodes. Since a negative deflection is always inscribed when an electrical pulse is moving away from the recording electrode, a sharp negative unipolar signal denotes the point of origin of the impulse, which then spreads away to the rest of the myocardial tissue (see Image. Unipolar and Bipolar Electrogram Recordings).[3]
In a baseline EPS, the EGMs are usually obtained from catheters located in the following anatomical structures:
- High right atrium near the sinus node (HRA)
- His-bundle (His)
- Coronary sinus (CS)
- The apex of the right ventricle (RV)
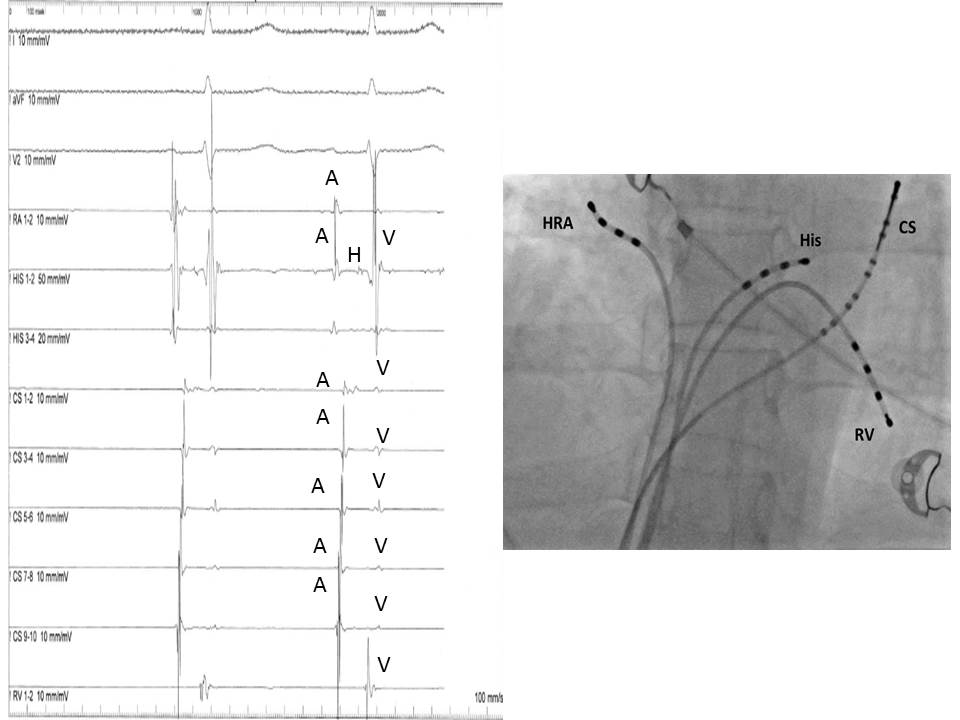
The most common displayed format follows the sequence of the normal impulse during sinus rhythm and from top to bottom includes: 2 or 3 ECG leads (usually I, III, and V1), HRA, His distal, His proximal, CS 1-10, and RV (see Image. Electrophysiologic Recording).
Indications
In conjunction with a noninvasive diagnostic assessment, the evaluation of cardiac arrhythmias is the main indication for conducting an EPS.[4] More specifically, EPS can provide valuable diagnostic and prognostic information in the assessment of:
- Sinus node dysfunction
- Atrioventricular conduction abnormalities
- Supraventricular tachyarrhythmias
- Ventricular tachyarrhythmias
- Result of an ablation procedure
Contraindications
An EPS is generally contraindicated in patients with:
- Acute coronary syndromes and/or decompensated heart failure since rapid atrial pacing can significantly deteriorate their symptoms, causing hemodynamic instability
- High risk of bleeding due to the administration of anticoagulation (heparin) during the procedure [5]
- Presence of arrhythmogenic conditions which can significantly decrease its diagnostic accuracy (eg, electrolyte abnormalities, drug toxicity, hyperthyroidism)
- Pregnancy due to radiation exposure (although in patients with arrhythmias resistant to medical management an ablation procedure with specific precautions can be performed) [6]
Equipment
A fully equipped EP laboratory includes:
- C-arm fluoroscopy with a radiographic table and an image intensifier
- EP data acquisition system and a cardiac stimulator
- Hemodynamic monitoring equipment
- External defibrillator and temporary pacing system
- Fully equipped resuscitation cart
- Intravenous infusion systems with the necessary drugs and solutions
- Standby equipment not necessarily present in the room, but immediately available in the case of suspected or verified complications (eg, echocardiographer, pericardial aspiration set, ventilator)
Personnel
The specialized arrhythmia team required for an EPS includes:
- An electrophysiologist who inserts and manipulates the EP catheters
- A second electrophysiologist or a technician for the data acquisition
- A nurse responsible for the care of the patient (eg, hemodynamic monitoring, respiratory function, intravenous drug administration)
- Standby personnel: anesthetist, interventional cardiologist, cardiac surgeon (to manage rare severe complications)
Preparation
The patient must be fasting for at least 6 hours before the procedure. Antiarrhythmic therapy must be withheld for at least 4 half-lives. Hypoglycemic therapy must be appropriately adjusted. Anticoagulation therapy is usually interrupted before the procedure for 3 to 4 days for warfarin and 1 to 2 days for novel anticoagulant regimens. "Bridging therapy" with low molecular weight heparin is generally not indicated.[7] On the other hand, uninterrupted therapy is an option with the novel anticoagulants.[8] However, the approach should be individualized, considering the patient's thromboembolic and bleeding risk. Intravenous access should be secured before arrival in the EP laboratory, and fluids should be administered during the procedure to avoid dehydration.
Upon arrival in the EP laboratory, the patient is connected to a 12-lead surface ECG (usually obtained through the EPS recording system), a pulse oximeter, a noninvasive blood pressure measurement cuff, and external defibrillation pads. Of note, the administration of intravenous sedation should be generally avoided since it can impede the induction of arrhythmias. Nevertheless, light sedation with the combination of benzodiazepines, opiates, and antiemetics is given in the great majority of patients to relieve pain from the initial puncture and anxiety.[9]
Technique or Treatment
The interpretation of an EPS includes the assessment of both the morphology and the timing of EGMs at baseline and after programmed electrical stimulation of the heart in relation to 2 or more simultaneously recorded surface ECG signals. Notably, the recording speed commonly used in EPS is 100 to 200 mm/sec instead of the usual 25 mm/sec of normal ECG recording. This sweep speed is adequate for a clear display of EGMs and for accurate measurements of the time intervals among the various deflections. While atrial and ventricular signals are easy to obtain and recognize due to their simultaneous inscription with the surface P and QRS complexes, the His bundle EGM requires specific maneuvers of the recording catheter, which must be oriented septally at the level of the tricuspid ring. In this area, His bundle EGM can be recorded as a biphasic or triphasic sharp deflection interpolated between the atrial and ventricular signals.
Another important point is the recordings obtained from the multipolar CS catheter. Since CS runs in the atrioventricular groove, the EP catheter records both atrial and ventricular signals. More importantly, the signals obtained from the distal (near the tip located) pairs of electrodes originate from the left atrium and the left ventricle, while those inscribed from the more proximal pairs of electrodes are coming from the right side. This way, the CS recording makes the distinction between right and left atrial signals possible.
Beyond the identification of EGMs, the interpretation of an EPS requires the understanding of the following terms:
- Cycle length
- Cycle length is measured between 2 consecutive events in milliseconds (ms), ie, between 2 consecutive normal or paced atrial or ventricular signals. The shorter the cycle length, the faster the rate. For instance, a cycle length of 600 ms corresponds to a heart rate of 100 beats/min, while a cycle length of 400 ms corresponds to a heart rate of 150 beats/min.
- Programmed stimulation
- The pacing of cardiac tissue through an EP catheter
- Incremental pacing (or burst pacing)
- Introducing a specific number of pacing stimuli (train) at a progressively shorter cycle length
- Extra stimulus pacing
- This introduces 1 or more (more commonly up to 3) premature impulses at specific coupling intervals. The extra stimuli are introduced after a native impulse or a train of 8 impulses paced at a fixed cycle.
- Coupling interval
- The time measured in ms between a normal impulse and a premature impulse introduced through programmed stimulation (pacing).
- Effective refractory period
- An effective refractory period refers to a particular tissue or structure, such as the ERP of the ventricle or the atrioventricular node. ERP is the longest coupling interval at which a premature impulse fails to propagate through the tissue or the structure.
The baseline analysis of intracardiac recordings starts with the measurement of the time intervals between consecutive signals in relation to the normal activation sequence of the heart in sinus rhythm. Since the sinus node is located high in the right atrium, the first atrial bipolar EGM is recorded from the HRA catheter, followed by the atrial signals in the His catheter and the proximal poles of the CS catheter. The most delayed atrial signal is normally inscribed from the distal pole of the CS catheter (CS 1,2) located at the left atrium's lateral wall. The atrial signals are followed by the His signal and, after that, by the RV signal. The baseline time intervals (conduction intervals) measured after the identification of EGMs are:
- PA interval
- This interval is measured from the onset of the P wave to the onset of the low atrial signal in the His bundle recording and represents the intraatrial conduction time (normally 25-55 ms).
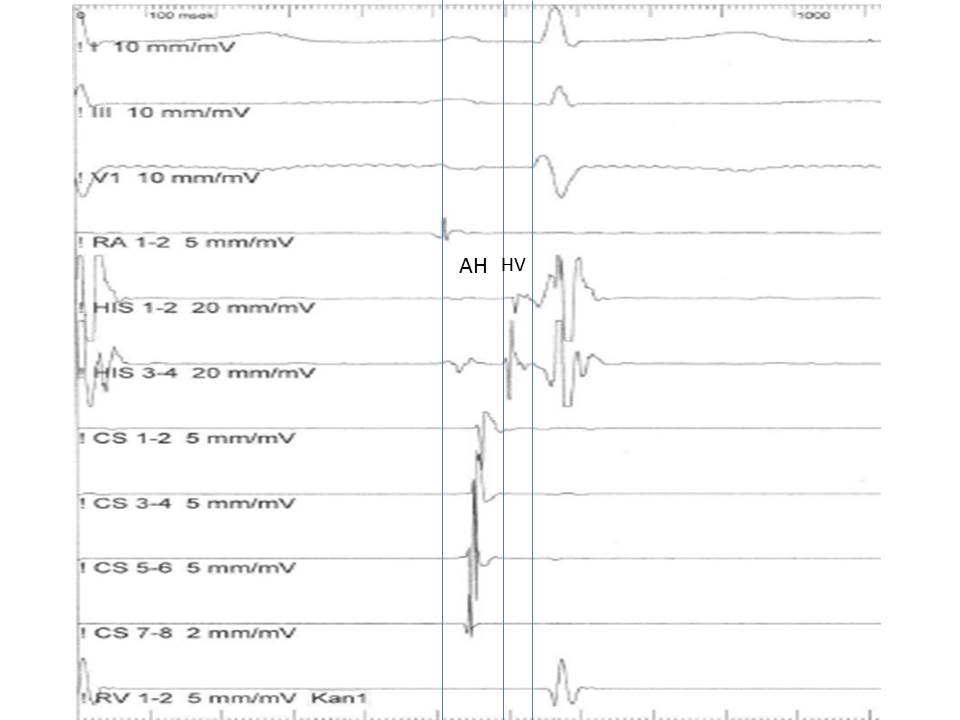
- AH interval
- This interval is measured from the onset of the atrial signal to the onset of the His bundle signal in His catheter recording and represents the conduction time through the AV node (normally 55-125 msec).
- H time
- This is measured from the onset to the end of His bundle recording and represents the conduction time through the His bundle (normally <30 ms).
- HV interval
- This interval is measured from the onset of His deflection to the onset of the QRS in the surface ECG. This represents the conduction time from the His bundle to the ventricular myocardium (normally 35-55 msec) (see Image. His Bundle Recording).
Complications
The complication rate of EPS is relatively low when performed by an experienced operator with the support of a well-trained arrhythmia team. Radiofrequency ablation and advanced age are the most important risk factors associated with an increased rate of complications.[10] The most common complication is the induction of sustained ventricular tachycardia requiring cardioversion.[11]
Other less frequent complications include local vascular injury, thrombophlebitis, pulmonary embolism, systemic arterial embolism, cardiac perforation and tamponade, catheter-induced atrioventricular block, and induction of nonclinical atrial fibrillation. The risk of life-threatening complications such as myocardial infarction, stroke, or sudden death is generally less than 0.5%. Nevertheless, the patient must be thoroughly informed about all the possible risks and benefits associated with the procedure, and undersigned consent must always be obtained from the clinician responsible for the patient's care.
Clinical Significance
Concerning the procedure's clinical significance, the evaluation of sinus node dysfunction, atrioventricular conduction abnormalities, supraventricular or ventricular tachyarrhythmias, and EP findings must be addressed after successful ablation of an arrhythmia.
Evaluation of Sinus Node Dysfunction
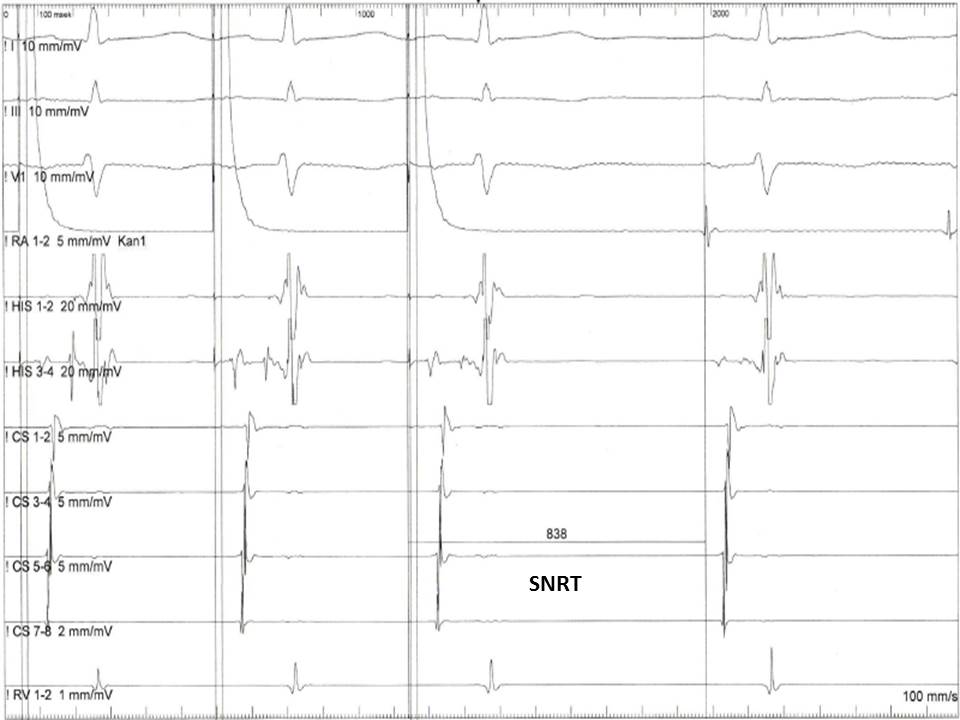
The EP evaluation of sinus node dysfunction includes measuring the sinus node recovery time (SNRT) and the sinoatrial conduction time (SACT). The SNRT is the most important of these 2 parameters and is defined as the interval between the last paced atrial signal and the first spontaneous atrial EGM in the HRA recording (see Image. Measurement of the Sinus Node Recovering Time). The protocol includes the stimulation of the atria at a fixed rate for 30 seconds at various cycle lengths (600, 500, and 400 ms). The stimulation is then abruptly terminated, and the time needed for the sinus node to regain its automaticity is measured. Normally, the SNRT must be less than 1600 ms. The sinus cycle length is usually subtracted from the SNRT to standardize the measurement. The corrected SNRT should not exceed 525 ms.
The SACT estimates the time needed from the sinus impulse to reach the perinodal atrial tissue and is rarely measured nowadays in clinical practice. A prolonged SACT indicates some degree of sinus exit block, and is measured after the delivery of premature atrial stimuli (Strauss method) or after continuous atrial pacing at a rate slightly faster than the basic sinus cycle. The impulse is always delivered from the HRA catheter, which is located near the sinus node. Similarly to the SNRT measurement, the time from the last paced atrial signal to the first spontaneous sinus signal is measured. This return interval represents the sum of time the paced impulse needs to conduct from the HRA into the sinus node, the time needed for the subsequent spontaneous sinus node beat to conduct through the perinodal tissue, and the spontaneous sinus cycle length. Assuming that the conduction times in and out of the sinus node, ie, the SACT, are equal, then the SACT can be calculated from the formula:
Return interval = Spontaneous cycle length + 2 x SACT
Notably, the role of EP evaluation of sinus node dysfunction has substantially subsided in recent years. The various noninvasive monitoring methods have shown a better diagnostic yield. Nevertheless, the method can still be useful for assessing patients with syncope and asymptomatic bradycardia or sinoatrial block documented in surface ECG.[12]
Evaluation of Atrioventricular Conduction Abnormalities
His bundle recording is the focus of interest for assessing atrioventricular (AV) conduction abnormalities. A prolonged AV interval in the His EGM denotes a block within the compact AV node, which usually does not need further evaluation or treatment. On the other hand, longer duration or splitting of the His EGM into 2 components reflects a conduction delay within the His bundle. At the same time, a significant prolongation of the HV time (more than 100 ms) indicates a block below the AV node in the His-Purkinje system.[13] Both these abnormalities are an indication for the implantation of a permanent pacemaker since they can suddenly progress to a complete AV block, causing syncope and even sudden cardiac death.
If the patient presents with symptoms suggesting an AV conduction abnormality but the baseline intervals in the His EGM are within normal limits, a further assessment of the AV conduction system under "stress" is required. This is accomplished by delivering extra atrial stimuli at progressively shorter intervals or through incremental atrial pacing. A gradual prolongation of the AH interval at progressively earlier premature extra atrial stimuli or progressively faster atrial pacing rates is a normal response of the AV node called "decremental conduction." This normal conduction delay of the incoming impulses at the level of the AV node is a protective mechanism against the development of rapid and potentially dangerous ventricular rates.
The pacing protocols aim to determine the occurrence of an AV block and whether it is at the level of the AV node (supra-His block) or the His-Purkinje system (infra-His block). In the first case, an atrial signal will be recorded in the His bundle electrogram (EGM) without a subsequent His or ventricular signal. On the contrary, an atrial signal followed by a His deflection in the absence of a subsequent ventricular signal indicates a block distal to the AV node and establishes the indication of pacemaker implantation.[14]
Evaluation of Supraventricular Tachyarrhythmias
The assessment of supraventricular tachyarrhythmias is the most challenging but also the most important part of an EPS since it offers invaluable information about the mechanism of the arrhythmia and guides the therapy. Abnormal automaticity and reentry are the 2 main EP abnormalities causing tachyarrhythmias. Abnormal automaticity refers to the abnormal initiation of an impulse from 1 myocardial focus (cell or group of cells) different from the sinus node. The focal tachyarrhythmias are characterized by a rate acceleration at the beginning and a rate deceleration at the end of the arrhythmia, the so-called "warm-up" and "cool down" phenomenon. Focal atria tachycardia (FAT) is the most common supraventricular tachyarrhythmia due to abnormal automaticity.[15]
On the other hand, the reentry mechanism is not associated with the initiation but with the propagation of the impulse. A nonuniformly propagated impulse results in late activation of myocardial regions (conduction delay). Other previously excited regions have recovered excitability and, consequently, can be reexcited retrogradely from the same impulse. This EP abnormality can lead to the establishment of a continuous circuit. At the same time, the propagation of the rapid circulating impulse to the rest of the myocardium results in a tachyarrhythmia with an abrupt onset. More importantly, any block (eg, pharmaceutical or invasive) at any limb of the reentry circuit can abruptly terminate the arrhythmia. The most common reentrant supraventricular tachyarrhythmias are the AV nodal reentrant tachycardia (AVNRT), the orthodromic and antidromic AV reentrant tachycardias (AVRT), and the macroreentry atrial tachycardias (MRAT). In AVNRT, the most common reentrant arrhythmia is the reentrant circuit located within the AV node. This circuit is usually composed of 2 pathways, a fast (faster conduction and longer refractory period) and a slow pathway (slower conduction and shorter refractory period).
On the other hand, in AVRT, the AV conduction system is 1 of the 2 limbs of the reentrant circuit, with the other being an accessory pathway conducting either retrograde from the ventricles to the atria (orthodromic AVRT) or anterograde from the atria to the ventricles (antidromic AVNRT). Finally, in MRAT, the AV conduction system is not part of the reentry circuit confined within the atrial tissue. They are further divided into cavotricuspid isthmus-dependent when the circuit passes through the anatomical area confined by the inferior vena cava and the tricuspid ring (ie, common and reverse atrial flutter) and noncavotricuspid isthmus-dependent or atypical flutter if the circuit is located somewhere else in the right or the left atrium.
EPS aims to initially make an appropriate differential diagnosis of a spontaneously running or induced supraventricular arrhythmia (through programmed atrial or ventricular stimulation).[16] Beyond observing the specific EGM pattern of the arrhythmia, additional pacing maneuvers may be needed to establish the diagnosis. After the diagnosis has been confirmed, therapeutic intervention with the application of radiofrequency energy through specifically designed ablation catheters in critical areas within the heart usually follows. Finally, a control EPS is performed 30 to 60 minutes after the ablation procedure to evaluate the effectiveness of the therapy.
Observing EGMs recorded in sinus rhythm can offer important initial information about the arrhythmia mechanism. The presence of an accessory pathway makes the diagnosis of AVRT highly likely. Overt preexcitation on the ECG, fusion of the A and V signals and a negative HV interval—indicating that the QRS onset precedes the His signal—confirms the diagnosis of an accessory pathway. On the other hand, the absence of retrograde conduction during ventricular pacing in sinus rhythm rules out a retrograde conducting accessory pathway and accordingly excludes the diagnosis of AVRT.
The initiation of the tachycardia is the first point that needs to be addressed during the EPS. All supraventricular tachyarrhythmias can be induced with atrial stimulation, while ventricular stimulation can induce an AVRT, less commonly an AVNRT, and rarely a FAT or an MRAT. Moreover, a reentry tachycardia is induced more quickly when an extra stimulus is delivered at the right moment (extra stimulus pacing). In contrast, the induction of a focal tachycardia is facilitated mainly by incremental pacing. Also, a significant conduction delay of the last beat before the onset of the tachycardia strongly indicates a reentry mechanism, since it implies a shift of the impulse propagation from the fast conducting limb to the slow one.
At the level of the AV node, a gradual increase in the AH interval with progressively shorter coupling intervals of the extra atrial stimuli is usually expected due to the decremental conduction property of the AV. A sudden increase ("jump") in the AH interval of at least 50 ms, though, indicates that the ERP of the fast pathway is reached, and the impulse is conducted through the slow pathway. Suppose the fast pathway's excitability has recovered when the impulse reaches the end of the slow pathway. In that case, retrograde conduction ("echo beat ") through the fast pathway can happen (see Image. Induction of an Atrioventricular Nodal Reentry Tachycardia).
Almost simultaneous activation of the atria and the ventricles is recorded with a ventriculoatrial (VA) interval, measured for the earliest QRS deflection to the earliest atrial deflection in the His bundle EGM, typically less than 60 ms.[17] This pattern of anterograde and retrograde conduction can be interrupted after the first "echo beat" if there is a change in the refractoriness of the 2 limbs of the reentry circuit, or can continue as AVNRT. On the other hand, if the limbs of the reentrant circuit include the AV node-His-Purkinje system and an accessory pathway, a VA interval is expected to be more than 65 ms in the case of an orthodromic AVRT, since the retrograde conduction of the impulse through the ventricular tissue and the accessory pathway back to atria requires more time.
The mode of termination of the arrhythmia can also help delineate its mechanism. Suppose the tachycardia terminates with an atrial EGM with AV block. In that case, the diagnosis of FAT is unlikely since there is no reason for the last atrial impulse initiated from the ectopic focus not to be conducted to the ventricles. On the other hand, any supraventricular tachycardia can terminate with a ventricular EGM, a pattern which does not provide any diagnostic information regarding the mechanism of the arrhythmia.
The atrial activation pattern during the tachycardia is another important diagnostic feature of the EPS. In MRATs, for example, atrial signals are recorded throughout the tachycardia cycle length. In contrast, atrial EGMs take up only a small portion of the tachycardia cycle in the other types of supraventricular tachycardia. Moreover, the earliest atrial EGM can indicate the site of origin of the arrhythmia. If the earliest signal is recorded in the high right atrium, a right-sided FAT or an AVRT through a right-sided accessory pathway are the most likely diagnoses. On the contrary, the earliest atrial activation signal recorded in the distal poles of the CS catheter ("eccentric activation pattern") indicates an AVRT with retrograde atrial activation through a left-sided accessory pathway or a left FAT.
Finally, the tachycardia response to atrial and ventricular stimulation provides further clues for the diagnosis. An atrial extra stimulus, for example, can penetrate a reentrant circuit, collide with the circulating impulse, and terminate the arrhythmia. Such a response is less likely if a focal activity is the arrhythmia mechanism. Ventricular stimulation, however, is even more informative of the arrhythmia mechanism. An appropriately timed ventricular extra stimulus can penetrate the atria through the His bundle and reset the tachycardia, changing its cycle length.
Suppose that happens when the His bundle is in the refractory phase and does not permit conduction through it (His synchronous pacing). In that case, an alternative route must be responsible for the retrograde atrial conduction. This finding establishes the diagnosis of an accessory pathway, which is most likely part of the reentry circuit. Moreover, if this maneuver, instead of resetting, terminates the tachycardia without conducting the extra stimulus to the atrium, then AVRT is ruled out, and the accessory pathway is a necessary part of the arrhythmia and not a possible bystander. However, this finding does not rule out an AVNRT. AV dissociation, which typically happens when a ventricular beat is not conducted back to the atria, can still occur. AVNRT can continue since the AV node defines the reentrant circuit. His Purkinje system is not part of it.
Beyond the resetting with a single ventricular extra stimulus, a continuous resetting of the tachycardia through ventricular pacing at a rate slightly faster than the cycle length of the arrhythmia (overdrive pacing) can also happen. Penetration of the paced beats through an accessory pathway or the AV node can reset and accelerate (entrain) the tachycardia at the paced cycle length. Of importance is the recording immediately after the cessation of pacing. In the case of a reentry tachycardia, the last paced beat is conducted up to the atrium through one of the limbs of the reentrant circuit (AV node or accessory pathway) and travels back to the ventricles through the other limb of the loop, continuing the tachycardia (as so-called VAVA response).
On the other hand, in the case of a FAT, the paced ventricular pulse is conducted retrograde through the AV node and suppresses the ectopic focus through the premature atrial depolarization. This premature atrial impulse cannot be conducted back to the ventricles because the AV node is still refractory from its previous retrograde depolarization. For the tachycardia to continue, a new A wave has to be generated and travel down to the ventricles (a so-called VAAV response).
Evaluation of Ventricular Tachyarrhythmias
Like supraventricular tachycardias, abnormal automaticity and reentry are the 2 main mechanisms of ventricular arrhythmias. Premature ventricular contractions (PVC) are always of focal origin. On the other hand, VT can be either focal or reentrant. The role of baseline EP evaluation is rather limited in the case of ventricular arrhythmias and is practically confined to the confirmation of the diagnosis of VT and the induction of VT through various stimulation protocols. Of note is the inducibility of a VT with ventricular extra stimuli, which highly suggests a reentry mechanism.
AV dissociation, the hallmark of VT, is easy to recognize from the simultaneous recording of atrial and ventricular EGMs. Nevertheless, VT with 1:1 VA conduction can also happen. In that case, diagnosing VT is more difficult since a supraventricular tachycardia with aberrancy (conducting atrial signals to the ventricles with bundle branch block or through an accessory pathway) cannot be excluded. The induction of AV block with the administration of adenosine is the easiest way to dissociate the atria transiently from the ventricles. If the wide complex tachycardia continues, VT is the most likely diagnosis.
The induction of VT with extra stimulus ventricular pacing highly suggests reentry as the underlying mechanism. However, the induction of a VT is not as easy as it is in the case of reentrant supraventricular tachycardias. One important reason is the distance between the pacing site, usually at the apex of the right ventricle, and the reentrant circuit, which is usually located in the epicardium or the subendocardium in the left ventricle. A 'drive train' of 8 paced beats is followed by 1 to 3 premature stimuli to facilitate the penetration of the premature stimulus in the reentrant circuit.
The idea is to shorten the refractory period with fast pacing, increasing the chances of extra stimuli reaching and penetrating the reentrant circuit. Most pacing protocols use a drive CL of 600, 500, and 400 msec. The extra stimuli are then delivered at progressively shortened coupling intervals up to the point of refractoriness (no capture) of the ventricular myocardium.[18] The induction of a sustained monomorphic VT is a clear indication for the implantation of a cardioverter-defibrillator. In contrast, the induction of ventricular fibrillation is considered a nonspecific response without clinical significance and is usually the result of an aggressive pacing protocol.[19]
EP Findings After Successful Ablation of an Arrhythmia
A controlled EPS is always performed after an ablation procedure to assess the immediate result and to predict the long-term outcome. Noninducibility of the ablated arrhythmia after programmed stimulation is the main finding indicating a successful endpoint. More specific findings include:
- After the ablation of an AVNRT:
- Loss of the AV jump indicates the complete elimination of the 'slow pathway' or presence of the AV jump but induction of no more than 1 echo beat, which indicates an effective AV modification.[20]
- After the ablation of an accessory pathway:
- Disappearance of the delta wave in ECG and abolishment of the continuity between the atrial and ventricular signals.[21]
- After the ablation of macroreentry tachycardia:
- The significant prolongation of conduction time from one to the other side of the ablation line or the recording of double potentials along this line. These findings indicate a successful conduction block of the macroreentry circuit in this particular anatomic area.[22]
Enhancing Healthcare Team Outcomes
Interpreting EPS requires a specialized interprofessional team, including electrophysiologists, cardiologists, nurses, and technicians, to ensure accurate diagnosis and optimal patient outcomes. The procedure involves an operator performing invasive maneuvers while another team member manages the recording system and external stimulator. Highly trained nurses or technicians fulfill this role, ensuring precise data collection and analysis.
Nurses also play a vital role in patient care by monitoring vital signs, administering oxygen and intravenous medications, and assisting with procedural setup. Their involvement includes early diagnosis and management of complications, postprocedural care, and long-term follow-up for patients with arrhythmias. Additionally, collaboration with clinical cardiologists, particularly those specializing in heart failure, cardiac surgeons, and geneticists, enhances comprehensive patient management. While not extensively studied, this integrated team approach is expected to improve arrhythmia outcomes significantly.[23]
Maintaining high standards in EPS interpretation requires continuous education and hands-on training in high-volume centers. The Heart Rhythm Society and the European Heart Rhythm Association provide updated guidelines for physicians and allied professionals, ensuring competency in arrhythmia diagnosis and treatment. Effective interprofessional communication and care coordination are crucial for integrating findings from EPS into patient management strategies, particularly when planning interventions such as catheter ablation or device implantation. By fostering collaboration among medical professionals, the electrophysiology team can enhance patient safety, optimize treatment decisions, and improve outcomes for individuals with complex cardiac arrhythmias.[24][25]
Media
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
(Click Image to Enlarge)
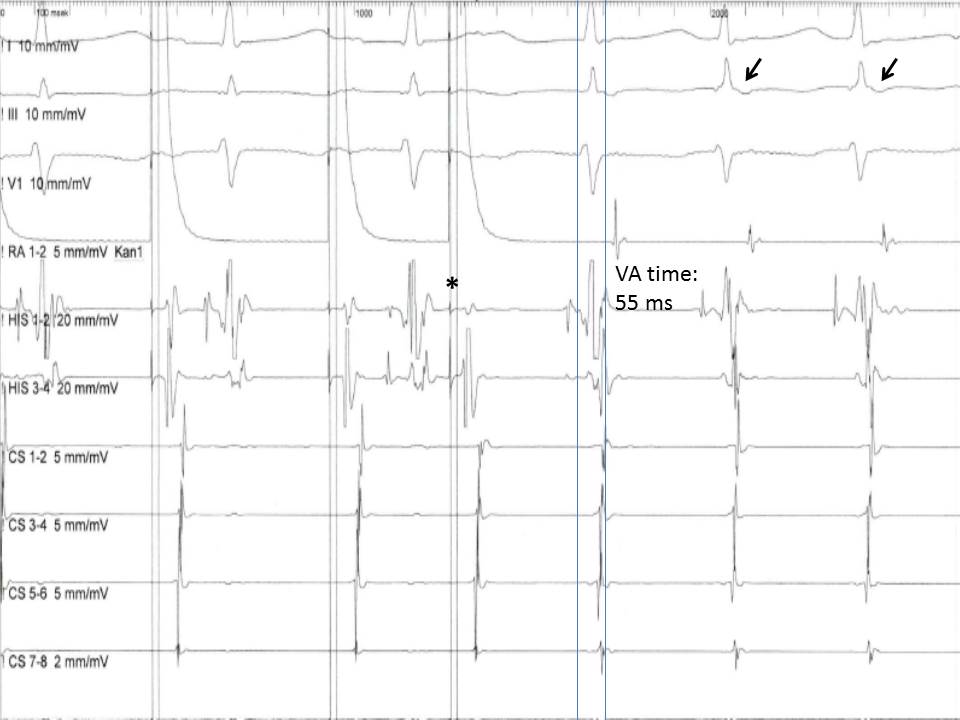
Induction of an Atrioventricular Nodal Reentry Tachycardia. This image shows the induction of an atrioventricular nodal reentry tachycardia (AVNRT) with a premature atrial extra stimulus (asterisk). Note the short ventriculoatrial time (<60 ms) in the His bundle recording and the retrograde P waves in the electrocardiogram leads (arrows).
Contributed by S Koulouris, MD
References
Albert CM, Stevenson WG. The Future of Arrhythmias and Electrophysiology. Circulation. 2016 Jun 21:133(25):2687-96. doi: 10.1161/CIRCULATIONAHA.116.023519. Epub [PubMed PMID: 27324363]
Enriquez A, Gonzalez R, Kumareswaran R, Supple G, Scheinman M. Electrophysiologic diagnosis of narrow and wide complex tachyarrhythmias. Heart rhythm. 2024 Nov:21(11):2215-2225. doi: 10.1016/j.hrthm.2024.05.010. Epub 2024 May 10 [PubMed PMID: 38734227]
Delacretaz E, Soejima K, Gottipaty VK, Brunckhorst CB, Friedman PL, Stevenson WG. Single catheter determination of local electrogram prematurity using simultaneous unipolar and bipolar recordings to replace the surface ECG as a timing reference. Pacing and clinical electrophysiology : PACE. 2001 Apr:24(4 Pt 1):441-9 [PubMed PMID: 11341080]
Muresan L, Cismaru G, Martins RP, Bataglia A, Rosu R, Puiu M, Gusetu G, Mada RO, Muresan C, Ispas DR, Le Bouar R, Diene LL, Rugina E, Levy J, Klein C, Sellal JM, Poull IM, Laurent G, de Chillou C. Recommendations for the use of electrophysiological study: Update 2018. Hellenic journal of cardiology : HJC = Hellenike kardiologike epitheorese. 2019 Mar-Apr:60(2):82-100. doi: 10.1016/j.hjc.2018.09.002. Epub 2018 Sep 29 [PubMed PMID: 30278230]
Anderson R, Harukuni I, Sera V. Anesthetic considerations for electrophysiologic procedures. Anesthesiology clinics. 2013 Jun:31(2):479-89. doi: 10.1016/j.anclin.2013.01.005. Epub 2013 Feb 23 [PubMed PMID: 23711654]
Enriquez AD, Economy KE, Tedrow UB. Contemporary management of arrhythmias during pregnancy. Circulation. Arrhythmia and electrophysiology. 2014 Oct:7(5):961-7. doi: 10.1161/CIRCEP.114.001517. Epub [PubMed PMID: 25336366]
Kuo HC, Liu FL, Chen JT, Cherng YG, Tam KW, Tai YH. Thromboembolic and bleeding risk of periprocedural bridging anticoagulation: A systematic review and meta-analysis. Clinical cardiology. 2020 May:43(5):441-449. doi: 10.1002/clc.23336. Epub 2020 Jan 16 [PubMed PMID: 31944351]
Level 1 (high-level) evidenceCardoso R, Willems S, Gerstenfeld EP, Verma A, Schilling R, Hohnloser SH, Okumura K, Nordaby M, Brouwer MA, Calkins H. Uninterrupted anticoagulation with non-vitamin K antagonist oral anticoagulants in atrial fibrillation catheter ablation: Lessons learned from randomized trials. Clinical cardiology. 2019 Jan:42(1):198-205. doi: 10.1002/clc.23120. Epub 2018 Dec 7 [PubMed PMID: 30460702]
Level 1 (high-level) evidenceBenzoni T, Cascella M. Procedural Sedation. StatPearls. 2025 Jan:(): [PubMed PMID: 31869149]
Chen SA, Chiang CE, Tai CT, Cheng CC, Chiou CW, Lee SH, Ueng KC, Wen ZC, Chang MS. Complications of diagnostic electrophysiologic studies and radiofrequency catheter ablation in patients with tachyarrhythmias: an eight-year survey of 3,966 consecutive procedures in a tertiary referral center. The American journal of cardiology. 1996 Jan 1:77(1):41-6 [PubMed PMID: 8540455]
Level 3 (low-level) evidenceHorowitz LN, Kay HR, Kutalek SP, Discigil KF, Webb CR, Greenspan AM, Spielman SR. Risks and complications of clinical cardiac electrophysiologic studies: a prospective analysis of 1,000 consecutive patients. Journal of the American College of Cardiology. 1987 Jun:9(6):1261-8 [PubMed PMID: 3584718]
Brignole M, Moya A, de Lange FJ, Deharo JC, Elliott PM, Fanciulli A, Fedorowski A, Furlan R, Kenny RA, Martín A, Probst V, Reed MJ, Rice CP, Sutton R, Ungar A, van Dijk JG, ESC Scientific Document Group. 2018 ESC Guidelines for the diagnosis and management of syncope. European heart journal. 2018 Jun 1:39(21):1883-1948. doi: 10.1093/eurheartj/ehy037. Epub [PubMed PMID: 29562304]
Saichin A, Dreifus LS, Michelson EL. Electrophysiologic studies of the AV conduction system and AV nodal arrhythmias. Cardiovascular clinics. 1985:16(1):61-82 [PubMed PMID: 3915716]
European Society of Cardiology (ESC), European Heart Rhythm Association (EHRA), Brignole M, Auricchio A, Baron-Esquivias G, Bordachar P, Boriani G, Breithardt OA, Cleland J, Deharo JC, Delgado V, Elliott PM, Gorenek B, Israel CW, Leclercq C, Linde C, Mont L, Padeletti L, Sutton R, Vardas PE. 2013 ESC guidelines on cardiac pacing and cardiac resynchronization therapy: the task force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA). Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2013 Aug:15(8):1070-118. doi: 10.1093/europace/eut206. Epub 2013 Jun 24 [PubMed PMID: 23801827]
Higa S, Tai CT, Lin YJ, Liu TY, Lee PC, Huang JL, Hsieh MH, Yuniadi Y, Huang BH, Lee SH, Ueng KC, Ding YA, Chen SA. Focal atrial tachycardia: new insight from noncontact mapping and catheter ablation. Circulation. 2004 Jan 6:109(1):84-91 [PubMed PMID: 14691042]
Katritsis DG, Josephson ME. Differential diagnosis of regular, narrow-QRS tachycardias. Heart rhythm. 2015 Jul:12(7):1667-76. doi: 10.1016/j.hrthm.2015.03.046. Epub 2015 Mar 28 [PubMed PMID: 25828600]
Katritsis DG, Josephson ME. Classification, Electrophysiological Features and Therapy of Atrioventricular Nodal Reentrant Tachycardia. Arrhythmia & electrophysiology review. 2016 Aug:5(2):130-5. doi: 10.15420/AER.2016.18.2. Epub [PubMed PMID: 27617092]
Prystowsky EN, Heger JJ, Lloyd EA, Zipes DP. Clinical electrophysiology of ventricular tachycardia. Cardiology clinics. 1983 May:1(2):253-73 [PubMed PMID: 6544638]
Priori SG, Blomström-Lundqvist C, Mazzanti A, Blom N, Borggrefe M, Camm J, Elliott PM, Fitzsimons D, Hatala R, Hindricks G, Kirchhof P, Kjeldsen K, Kuck KH, Hernandez-Madrid A, Nikolaou N, Norekvål TM, Spaulding C, Van Veldhuisen DJ, ESC Scientific Document Group. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: The Task Force for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death of the European Society of Cardiology (ESC). Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC). European heart journal. 2015 Nov 1:36(41):2793-2867. doi: 10.1093/eurheartj/ehv316. Epub 2015 Aug 29 [PubMed PMID: 26320108]
Lindsay BD, Chung MK, Gamache MC, Luke RA, Schechtman KB, Osborn JL, Cain ME. Therapeutic end points for the treatment of atrioventricular node reentrant tachycardia by catheter-guided radiofrequency current. Journal of the American College of Cardiology. 1993 Sep:22(3):733-40 [PubMed PMID: 8354806]
Schwartz SM, Dick M 2nd, Dorostkar PC, Serwer GA, LeRoy S. Electrogram patterns associated with successful radiofrequency ablation of accessory pathways in children. Pediatric cardiology. 1996 May-Jun:17(3):137-42 [PubMed PMID: 8662025]
Level 3 (low-level) evidenceSchmieder S, Ndrepepa G, Dong J, Zrenner B, Schreieck J, Schneider MA, Karch MR, Schmitt C. Acute and long-term results of radiofrequency ablation of common atrial flutter and the influence of the right atrial isthmus ablation on the occurrence of atrial fibrillation. European heart journal. 2003 May:24(10):956-62 [PubMed PMID: 12714027]
Fumagalli S, Chen J, Dobreanu D, Madrid AH, Tilz R, Dagres N. The role of the Arrhythmia Team, an integrated, multidisciplinary approach to treatment of patients with cardiac arrhythmias: results of the European Heart Rhythm Association survey. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2016 Apr:18(4):623-7. doi: 10.1093/europace/euw090. Epub [PubMed PMID: 27174994]
Level 3 (low-level) evidenceZipes DP, Calkins H, Daubert JP, Ellenbogen KA, Field ME, Fisher JD, Fogel RI, Frankel DS, Gupta A, Indik JH, Kusumoto FM, Lindsay BD, Marine JE, Mehta LS, Mendes LA, Miller JM, Munger TM, Sauer WH, Shen WK, Stevenson WG, Su WW, Tracy CM, Tsiperfal A. 2015 ACC/AHA/HRS Advanced Training Statement on Clinical Cardiac Electrophysiology (A Revision of the ACC/AHA 2006 Update of the Clinical Competence Statement on Invasive Electrophysiology Studies, Catheter Ablation, and Cardioversion). Journal of the American College of Cardiology. 2015 Dec 22:66(24):2767-2802. doi: 10.1016/j.jacc.2015.08.040. Epub 2015 Sep 18 [PubMed PMID: 26390945]
Trines SA,Moore P,Burri H,Gonçalves Nunes S,Massoullié G,Merino JL,Paton MF,Porta-Sánchez A,Sommer P,Steven D,Whittaker-Axon S,Yorgun H,Arribas F,Claude Deharo J,Steffel J,Wolpert C, 2024 updated European Heart Rhythm Association core curriculum for physicians and allied professionals: a statement of the European Heart Rhythm Association of the European Society of Cardiology. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2024 Nov 1; [PubMed PMID: 39298665]