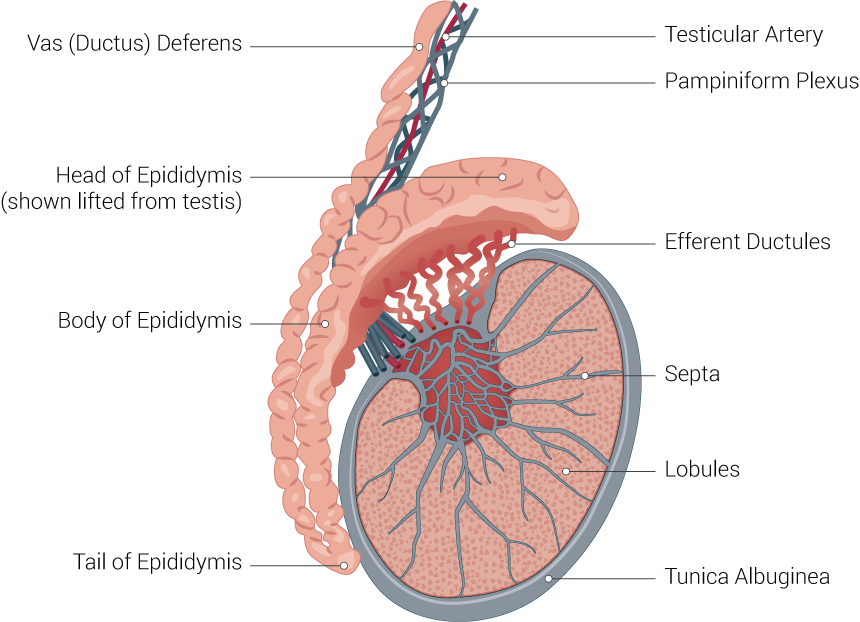
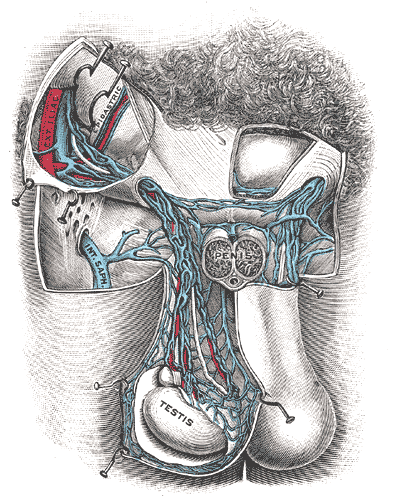
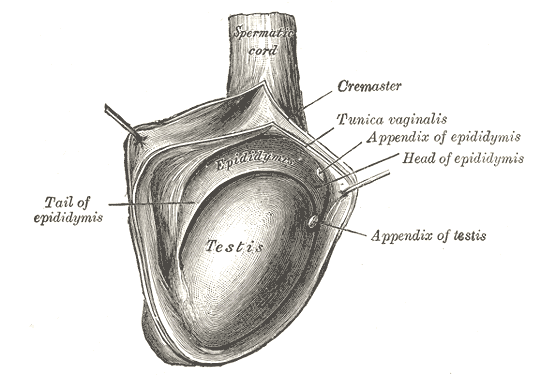
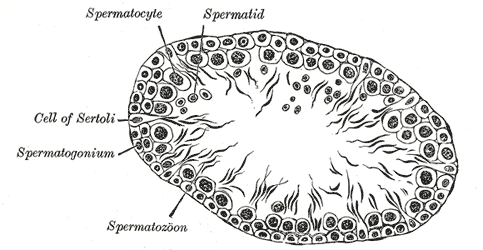
[1]
Tsili AC, Sofikitis N, Stiliara E, Argyropoulou MI. MRI of testicular malignancies. Abdominal radiology (New York). 2019 Mar:44(3):1070-1082. doi: 10.1007/s00261-018-1816-5. Epub
[PubMed PMID: 30386879]
[2]
Mäkelä JA, Koskenniemi JJ, Virtanen HE, Toppari J. Testis Development. Endocrine reviews. 2019 Aug 1:40(4):857-905. doi: 10.1210/er.2018-00140. Epub
[PubMed PMID: 30590466]
[3]
Yang Y, Workman S, Wilson M. The molecular pathways underlying early gonadal development. Journal of molecular endocrinology. 2018 Jul 24:():. pii: JME-17-0314. doi: 10.1530/JME-17-0314. Epub 2018 Jul 24
[PubMed PMID: 30042122]
[4]
Avellar MCW, Ribeiro CM, Dias-da-Silva MR, Silva EJR. In search of new paradigms for epididymal health and disease: innate immunity, inflammatory mediators, and steroid hormones. Andrology. 2019 Sep:7(5):690-702. doi: 10.1111/andr.12654. Epub 2019 Jun 17
[PubMed PMID: 31207127]
[5]
Acién P. Embryological observations on the female genital tract. Human reproduction (Oxford, England). 1992 Apr:7(4):437-45
[PubMed PMID: 1522183]
[6]
Gropp A, Ohno S. The presence of a common embryonic blastema for ovarian and testicular parenchymal (follicular, interstitial and tubular) cells in cattle Bos taurus. Zeitschrift fur Zellforschung und mikroskopische Anatomie (Vienna, Austria : 1948). 1966:74(4):505-28
[PubMed PMID: 5988471]
[7]
Gill ME, Hu YC, Lin Y, Page DC. Licensing of gametogenesis, dependent on RNA binding protein DAZL, as a gateway to sexual differentiation of fetal germ cells. Proceedings of the National Academy of Sciences of the United States of America. 2011 May 3:108(18):7443-8. doi: 10.1073/pnas.1104501108. Epub 2011 Apr 19
[PubMed PMID: 21504946]
[9]
Sinclair AH, Berta P, Palmer MS, Hawkins JR, Griffiths BL, Smith MJ, Foster JW, Frischauf AM, Lovell-Badge R, Goodfellow PN. A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature. 1990 Jul 19:346(6281):240-4
[PubMed PMID: 1695712]
[10]
Gubbay J, Collignon J, Koopman P, Capel B, Economou A, Münsterberg A, Vivian N, Goodfellow P, Lovell-Badge R. A gene mapping to the sex-determining region of the mouse Y chromosome is a member of a novel family of embryonically expressed genes. Nature. 1990 Jul 19:346(6281):245-50
[PubMed PMID: 2374589]
[11]
Albrecht KH, Eicher EM. Evidence that Sry is expressed in pre-Sertoli cells and Sertoli and granulosa cells have a common precursor. Developmental biology. 2001 Dec 1:240(1):92-107
[PubMed PMID: 11784049]
[12]
Sekido R, Lovell-Badge R. Sex determination involves synergistic action of SRY and SF1 on a specific Sox9 enhancer. Nature. 2008 Jun 12:453(7197):930-4. doi: 10.1038/nature06944. Epub 2008 May 4
[PubMed PMID: 18454134]
[13]
Warr N, Greenfield A. The molecular and cellular basis of gonadal sex reversal in mice and humans. Wiley interdisciplinary reviews. Developmental biology. 2012 Jul-Aug:1(4):559-77. doi: 10.1002/wdev.42. Epub 2012 Feb 28
[PubMed PMID: 23801533]
[14]
Larney C, Bailey TL, Koopman P. Switching on sex: transcriptional regulation of the testis-determining gene Sry. Development (Cambridge, England). 2014 Jun:141(11):2195-205. doi: 10.1242/dev.107052. Epub
[PubMed PMID: 24866114]
[15]
Hutson JM. A biphasic model for the hormonal control of testicular descent. Lancet (London, England). 1985 Aug 24:2(8452):419-21
[PubMed PMID: 2863447]
[16]
Emmen JM, McLuskey A, Grootegoed JA, Brinkmann AO. Androgen action during male sex differentiation includes suppression of cranial suspensory ligament development. Human reproduction (Oxford, England). 1998 May:13(5):1272-80
[PubMed PMID: 9647559]
[17]
Wensing CJ. Testicular descent in some domestic mammals. 3. Search for the factors that regulate the gubernacular reaction. Proceedings of the Koninklijke Nederlandse Akademie van Wetenschappen. Series C. Biological and medical sciences. 1973:76(2):196-202
[PubMed PMID: 4267621]
[18]
Heyns CF, Human HJ, De Klerk DP. Hyperplasia and hypertrophy of the gubernaculum during testicular descent in the fetus. The Journal of urology. 1986 May:135(5):1043-7
[PubMed PMID: 2421019]
[19]
Heyns CF, Human HJ, Werely CJ, De Klerk DP. The glycosaminoglycans of the gubernaculum during testicular descent in the fetus. The Journal of urology. 1990 Mar:143(3):612-7
[PubMed PMID: 2106043]
[20]
Kumagai J, Hsu SY, Matsumi H, Roh JS, Fu P, Wade JD, Bathgate RA, Hsueh AJ. INSL3/Leydig insulin-like peptide activates the LGR8 receptor important in testis descent. The Journal of biological chemistry. 2002 Aug 30:277(35):31283-6
[PubMed PMID: 12114498]
[21]
Heyns CF. The gubernaculum during testicular descent in the human fetus. Journal of anatomy. 1987 Aug:153():93-112
[PubMed PMID: 2892824]
[22]
Tanyel FC. Obliteration of processus vaginalis: aberrations in the regulatory mechanism result in an inguinal hernia, hydrocele or undescended testis. The Turkish journal of pediatrics. 2004:46 Suppl():18-27
[PubMed PMID: 15499794]
[23]
Patel AP. Anatomy and physiology of chronic scrotal pain. Translational andrology and urology. 2017 May:6(Suppl 1):S51-S56. doi: 10.21037/tau.2017.05.32. Epub
[PubMed PMID: 28725619]
[24]
Malas MA, Sulak O, Oztürk A. The growth of the testes during the fetal period. BJU international. 1999 Oct:84(6):689-92
[PubMed PMID: 10510117]
[25]
Nemec SF, Nemec U, Weber M, Kasprian G, Brugger PC, Krestan CR, Rotmensch S, Rimoin DL, Graham JM Jr, Prayer D. Male sexual development in utero: testicular descent on prenatal magnetic resonance imaging. Ultrasound in obstetrics & gynecology : the official journal of the International Society of Ultrasound in Obstetrics and Gynecology. 2011 Dec:38(6):688-94. doi: 10.1002/uog.8964. Epub 2011 Feb 17
[PubMed PMID: 21337441]
[26]
Barrionuevo F, Burgos M, Jiménez R. Origin and function of embryonic Sertoli cells. Biomolecular concepts. 2011 Dec 1:2(6):537-47. doi: 10.1515/BMC.2011.044. Epub
[PubMed PMID: 25962053]
[27]
Chojnacka K, Zarzycka M, Mruk DD. Biology of the Sertoli Cell in the Fetal, Pubertal, and Adult Mammalian Testis. Results and problems in cell differentiation. 2016:58():225-51. doi: 10.1007/978-3-319-31973-5_9. Epub
[PubMed PMID: 27300181]
[28]
Cheng CY, Mruk DD. Cell junction dynamics in the testis: Sertoli-germ cell interactions and male contraceptive development. Physiological reviews. 2002 Oct:82(4):825-74
[PubMed PMID: 12270945]
[29]
Silva FR, Leite LD, Wassermann GF. Rapid signal transduction in Sertoli cells. European journal of endocrinology. 2002 Sep:147(3):425-33
[PubMed PMID: 12213681]
[30]
Ni FD, Hao SL, Yang WX. Multiple signaling pathways in Sertoli cells: recent findings in spermatogenesis. Cell death & disease. 2019 Jul 17:10(8):541. doi: 10.1038/s41419-019-1782-z. Epub 2019 Jul 17
[PubMed PMID: 31316051]
[31]
Shi JF, Li YK, Ren K, Xie YJ, Yin WD, Mo ZC. Characterization of cholesterol metabolism in Sertoli cells and spermatogenesis (Review). Molecular medicine reports. 2018 Jan:17(1):705-713. doi: 10.3892/mmr.2017.8000. Epub 2017 Nov 7
[PubMed PMID: 29115523]
[32]
Makanji Y, Zhu J, Mishra R, Holmquist C, Wong WP, Schwartz NB, Mayo KE, Woodruff TK. Inhibin at 90: from discovery to clinical application, a historical review. Endocrine reviews. 2014 Oct:35(5):747-94. doi: 10.1210/er.2014-1003. Epub 2014 Jul 22
[PubMed PMID: 25051334]
[33]
O'Connor AE, De Kretser DM. Inhibins in normal male physiology. Seminars in reproductive medicine. 2004 Aug:22(3):177-85
[PubMed PMID: 15319820]
[34]
Shiratsuchi A, Osada Y, Nakanishi Y. Differences in the mode of phagocytosis of bacteria between macrophages and testicular Sertoli cells. Drug discoveries & therapeutics. 2013 Apr:7(2):73-7
[PubMed PMID: 23715505]
[35]
Svechnikov K, Landreh L, Weisser J, Izzo G, Colón E, Svechnikova I, Söder O. Origin, development and regulation of human Leydig cells. Hormone research in paediatrics. 2010:73(2):93-101. doi: 10.1159/000277141. Epub 2010 Feb 9
[PubMed PMID: 20190545]
[36]
Davidoff MS, Schulze W, Middendorff R, Holstein AF. The Leydig cell of the human testis--a new member of the diffuse neuroendocrine system. Cell and tissue research. 1993 Mar:271(3):429-39
[PubMed PMID: 8472301]
[37]
Mahran AM, Elgamal DA, Ghafeer HH, Abdel-Maksoud SA, Farrag AA. Histological alterations in Leydig cells and macrophages in azoospermic men. Andrologia. 2017 Oct:49(8):. doi: 10.1111/and.12714. Epub 2016 Oct 6
[PubMed PMID: 27709649]
[38]
Chen H, Wang Y, Ge R, Zirkin BR. Leydig cell stem cells: Identification, proliferation and differentiation. Molecular and cellular endocrinology. 2017 Apr 15:445():65-73. doi: 10.1016/j.mce.2016.10.010. Epub 2016 Oct 12
[PubMed PMID: 27743991]
[39]
Su H, Lau YF. Identification of the transcriptional unit, structural organization, and promoter sequence of the human sex-determining region Y (SRY) gene, using a reverse genetic approach. American journal of human genetics. 1993 Jan:52(1):24-38
[PubMed PMID: 8434602]
[40]
Hossain A, Saunders GF. The human sex-determining gene SRY is a direct target of WT1. The Journal of biological chemistry. 2001 May 18:276(20):16817-23
[PubMed PMID: 11278460]
[41]
Massardier J, Roth P, Michel-Calemard L, Rudigoz RC, Bouvier R, Dijoud F, Arnould P, Combourieu D, Gaucherand P. Campomelic dysplasia: echographic suspicion in the first trimester of pregnancy and final diagnosis of two cases. Fetal diagnosis and therapy. 2008:24(4):452-7. doi: 10.1159/000176299. Epub 2008 Nov 26
[PubMed PMID: 19033726]
Level 3 (low-level) evidence
[42]
Wagner T, Wirth J, Meyer J, Zabel B, Held M, Zimmer J, Pasantes J, Bricarelli FD, Keutel J, Hustert E, Wolf U, Tommerup N, Schempp W, Scherer G. Autosomal sex reversal and campomelic dysplasia are caused by mutations in and around the SRY-related gene SOX9. Cell. 1994 Dec 16:79(6):1111-20
[PubMed PMID: 8001137]
[43]
Kim Y, Kobayashi A, Sekido R, DiNapoli L, Brennan J, Chaboissier MC, Poulat F, Behringer RR, Lovell-Badge R, Capel B. Fgf9 and Wnt4 act as antagonistic signals to regulate mammalian sex determination. PLoS biology. 2006 Jun:4(6):e187
[PubMed PMID: 16700629]
[44]
De Santa Barbara P, Bonneaud N, Boizet B, Desclozeaux M, Moniot B, Sudbeck P, Scherer G, Poulat F, Berta P. Direct interaction of SRY-related protein SOX9 and steroidogenic factor 1 regulates transcription of the human anti-Müllerian hormone gene. Molecular and cellular biology. 1998 Nov:18(11):6653-65
[PubMed PMID: 9774680]
[45]
Oba K, Yanase T, Nomura M, Morohashi K, Takayanagi R, Nawata H. Structural characterization of human Ad4bp (SF-1) gene. Biochemical and biophysical research communications. 1996 Sep 4:226(1):261-7
[PubMed PMID: 8806624]
[46]
Shen WH, Moore CC, Ikeda Y, Parker KL, Ingraham HA. Nuclear receptor steroidogenic factor 1 regulates the müllerian inhibiting substance gene: a link to the sex determination cascade. Cell. 1994 Jun 3:77(5):651-61
[PubMed PMID: 8205615]
[47]
Cate RL, Mattaliano RJ, Hession C, Tizard R, Farber NM, Cheung A, Ninfa EG, Frey AZ, Gash DJ, Chow EP. Isolation of the bovine and human genes for Müllerian inhibiting substance and expression of the human gene in animal cells. Cell. 1986 Jun 6:45(5):685-98
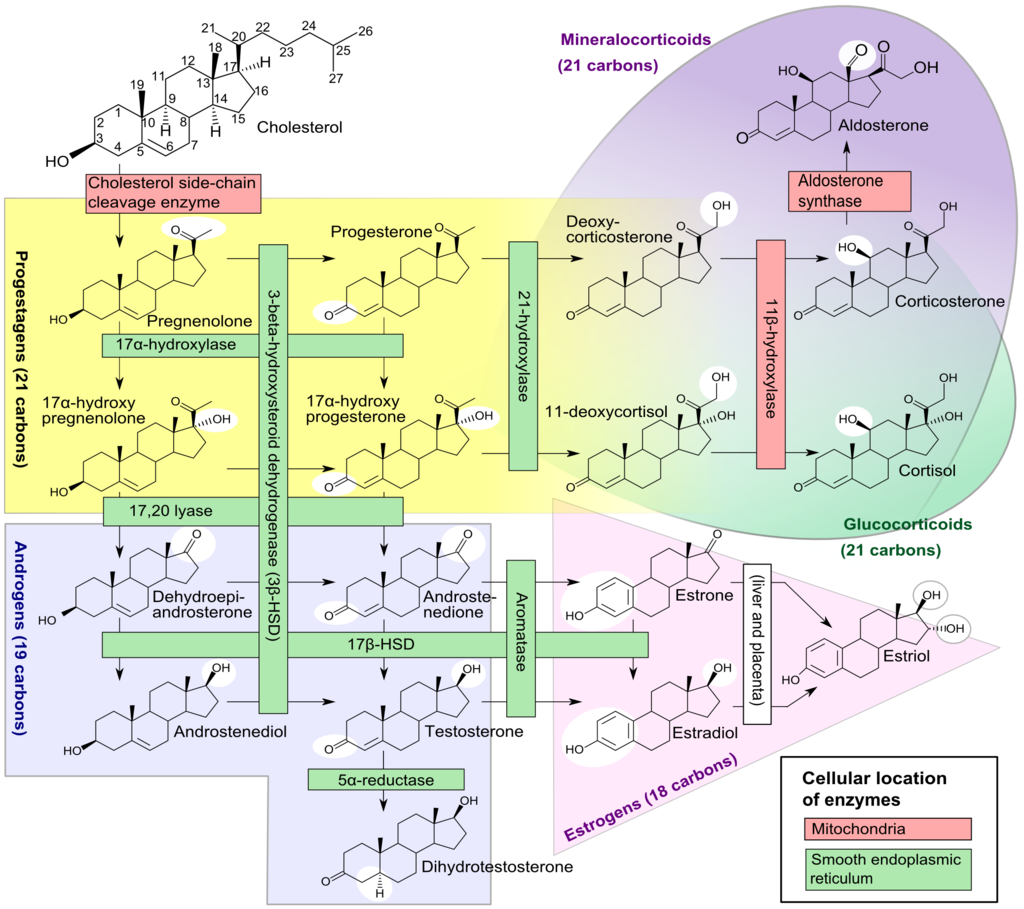
[PubMed PMID: 3754790]
Level 3 (low-level) evidence
[48]
Cohen-Haguenauer O, Picard JY, Mattéi MG, Serero S, Nguyen VC, de Tand MF, Guerrier D, Hors-Cayla MC, Josso N, Frézal J. Mapping of the gene for anti-müllerian hormone to the short arm of human chromosome 19. Cytogenetics and cell genetics. 1987:44(1):2-6
[PubMed PMID: 3028714]
[49]
Behringer RR. The in vivo roles of müllerian-inhibiting substance. Current topics in developmental biology. 1994:29():171-87
[PubMed PMID: 7828438]
[50]
Wang PY, Koishi K, McGeachie AB, Kimber M, Maclaughlin DT, Donahoe PK, McLennan IS. Mullerian inhibiting substance acts as a motor neuron survival factor in vitro. Proceedings of the National Academy of Sciences of the United States of America. 2005 Nov 8:102(45):16421-5
[PubMed PMID: 16260730]
[51]
Wang PY, Protheroe A, Clarkson AN, Imhoff F, Koishi K, McLennan IS. Müllerian inhibiting substance contributes to sex-linked biases in the brain and behavior. Proceedings of the National Academy of Sciences of the United States of America. 2009 Apr 28:106(17):7203-8. doi: 10.1073/pnas.0902253106. Epub 2009 Apr 9
[PubMed PMID: 19359476]
[52]
Burkhardt E, Adham IM, Brosig B, Gastmann A, Mattei MG, Engel W. Structural organization of the porcine and human genes coding for a Leydig cell-specific insulin-like peptide (LEY I-L) and chromosomal localization of the human gene (INSL3). Genomics. 1994 Mar 1:20(1):13-9
[PubMed PMID: 8020942]
[53]
Adham IM, Burkhardt E, Benahmed M, Engel W. Cloning of a cDNA for a novel insulin-like peptide of the testicular Leydig cells. The Journal of biological chemistry. 1993 Dec 15:268(35):26668-72
[PubMed PMID: 8253799]
[54]
Klonisch T, Fowler PA, Hombach-Klonisch S. Molecular and genetic regulation of testis descent and external genitalia development. Developmental biology. 2004 Jun 1:270(1):1-18
[PubMed PMID: 15136137]
[55]
Shalet SM. Normal testicular function and spermatogenesis. Pediatric blood & cancer. 2009 Aug:53(2):285-8. doi: 10.1002/pbc.22000. Epub
[PubMed PMID: 19343782]
[56]
Ulloa-Aguirre A, Reiter E, Crépieux P. FSH Receptor Signaling: Complexity of Interactions and Signal Diversity. Endocrinology. 2018 Aug 1:159(8):3020-3035. doi: 10.1210/en.2018-00452. Epub
[PubMed PMID: 29982321]
[57]
Urbański HF, Kohama SG, Garyfallou VT. Mechanisms mediating the response of GnRH neurones to excitatory amino acids. Reviews of reproduction. 1996 Sep:1(3):173-81
[PubMed PMID: 9414455]
[58]
Kauffman AS, Clifton DK, Steiner RA. Emerging ideas about kisspeptin- GPR54 signaling in the neuroendocrine regulation of reproduction. Trends in neurosciences. 2007 Oct:30(10):504-11
[PubMed PMID: 17904653]
[59]
Wildt L, Häusler A, Marshall G, Hutchison JS, Plant TM, Belchetz PE, Knobil E. Frequency and amplitude of gonadotropin-releasing hormone stimulation and gonadotropin secretion in the rhesus monkey. Endocrinology. 1981 Aug:109(2):376-85
[PubMed PMID: 6788538]
[60]
Savoy-Moore RT, Swartz KH. Several GnRH stimulation frequencies differentially release FSH and LH from isolated, perfused rat anterior pituitary cells. Advances in experimental medicine and biology. 1987:219():641-5
[PubMed PMID: 3124522]
Level 3 (low-level) evidence
[62]
Müller J, Skakkebaek NE. The prenatal and postnatal development of the testis. Bailliere's clinical endocrinology and metabolism. 1992 Apr:6(2):251-71
[PubMed PMID: 1616445]
[63]
Prince FP. Ultrastructural evidence of mature Leydig cells and Leydig cell regression in the neonatal human testis. The Anatomical record. 1990 Dec:228(4):405-17
[PubMed PMID: 2178325]
[64]
Zirkin BR, Papadopoulos V. Leydig cells: formation, function, and regulation. Biology of reproduction. 2018 Jul 1:99(1):101-111. doi: 10.1093/biolre/ioy059. Epub
[PubMed PMID: 29566165]
[65]
Clermont Y. Two classes of spermatogonial stem cells in the monkey (Cercopithecus aethiops). The American journal of anatomy. 1969 Sep:126(1):57-71
[PubMed PMID: 4982042]
[66]
Clermont Y, Antar M. Duration of the cycle of the seminiferous epithelium and the spermatogonial renewal in the monkey Macaca arctoides. The American journal of anatomy. 1973 Feb:136(2):153-65
[PubMed PMID: 4684877]
[67]
Schulze C. Morphological characteristics of the spermatogonial stem cells in man. Cell and tissue research. 1979 May 18:198(2):191-9
[PubMed PMID: 466665]
[68]
Cortes D, Müller J, Skakkebaek NE. Proliferation of Sertoli cells during development of the human testis assessed by stereological methods. International journal of andrology. 1987 Aug:10(4):589-96
[PubMed PMID: 3654012]
[69]
Setchell BP. The Parkes Lecture. Heat and the testis. Journal of reproduction and fertility. 1998 Nov:114(2):179-94
[PubMed PMID: 10070346]
[70]
EHRENBERG L, VON EHRENSTEIN G, HEDGRAN A. Gonad temperature and spontaneous mutation-rate in man. Nature. 1957 Dec 21:180(4599):1433-4
[PubMed PMID: 13493561]
[72]
Ritzén EM. Undescended testes: a consensus on management. European journal of endocrinology. 2008 Dec:159 Suppl 1():S87-90. doi: 10.1530/EJE-08-0181. Epub 2008 Aug 26
[PubMed PMID: 18728121]
Level 3 (low-level) evidence
[73]
Kanemoto K, Hayashi Y, Kojima Y, Maruyama T, Ito M, Kohri K. Accuracy of ultrasonography and magnetic resonance imaging in the diagnosis of non-palpable testis. International journal of urology : official journal of the Japanese Urological Association. 2005 Jul:12(7):668-72
[PubMed PMID: 16045560]
[74]
Sarihan H, Sari A, Abeş M, Dinç H. Nonpalpable undescending testis. Value of magnetic resonance imaging. Minerva urologica e nefrologica = The Italian journal of urology and nephrology. 1998 Dec:50(4):233-6
[PubMed PMID: 9973808]
[75]
Maghnie M, Vanzulli A, Paesano P, Bragheri R, Palladini G, Preti P, Del Maschio A, Severi F. The accuracy of magnetic resonance imaging and ultrasonography compared with surgical findings in the localization of the undescended testis. Archives of pediatrics & adolescent medicine. 1994 Jul:148(7):699-703
[PubMed PMID: 7912611]
[76]
Schreck RR, Distèche C. Karyotyping. Current protocols in human genetics. 2001 May:Appendix 4():Appendix 4A. doi: 10.1002/0471142905.hga04as18. Epub
[PubMed PMID: 18428228]
[77]
Cox MJ, Coplen DE, Austin PF. The incidence of disorders of sexual differentiation and chromosomal abnormalities of cryptorchidism and hypospadias stratified by meatal location. The Journal of urology. 2008 Dec:180(6):2649-52; discussion 2652. doi: 10.1016/j.juro.2008.08.058. Epub 2008 Oct 31
[PubMed PMID: 18951572]
[78]
Skakkebaek NE, Rajpert-De Meyts E, Main KM. Testicular dysgenesis syndrome: an increasingly common developmental disorder with environmental aspects. Human reproduction (Oxford, England). 2001 May:16(5):972-8
[PubMed PMID: 11331648]
[79]
Xing JS, Bai ZM. Is testicular dysgenesis syndrome a genetic, endocrine, or environmental disease, or an unexplained reproductive disorder? Life sciences. 2018 Feb 1:194():120-129. doi: 10.1016/j.lfs.2017.11.039. Epub 2017 Nov 26
[PubMed PMID: 29183799]
[80]
van den Driesche S, Kolovos P, Platts S, Drake AJ, Sharpe RM. Inter-relationship between testicular dysgenesis and Leydig cell function in the masculinization programming window in the rat. PloS one. 2012:7(1):e30111. doi: 10.1371/journal.pone.0030111. Epub 2012 Jan 11
[PubMed PMID: 22253897]
[81]
Chen GR, Dong L, Ge RS, Hardy MP. [Relationship between phthalates and testicular dysgenesis syndrome]. Zhonghua nan ke xue = National journal of andrology. 2007 Mar:13(3):195-200
[PubMed PMID: 17393778]
[82]
He M, Yang C, Geng R, Zhao X, Hong L, Piao X, Chen T, Quinto M, Li D. Monitoring of phthalates in foodstuffs using gas purge microsyringe extraction coupled with GC-MS. Analytica chimica acta. 2015 Jun 16:879():63-8. doi: 10.1016/j.aca.2015.02.066. Epub 2015 Feb 27
[PubMed PMID: 26002478]
[83]
Cevasco A, Urbatzka R, Bottero S, Massari A, Pedemonte F, Kloas W, Mandich A. Endocrine disrupting chemicals (EDC) with (anti)estrogenic and (anti)androgenic modes of action affecting reproductive biology of Xenopus laevis: II. Effects on gonad histomorphology. Comparative biochemistry and physiology. Toxicology & pharmacology : CBP. 2008 Mar:147(2):241-51
[PubMed PMID: 18032117]
Level 2 (mid-level) evidence
[84]
Mills LJ, Gutjahr-Gobell RE, Zaroogian GE, Horowitz DB, Laws SC. Modulation of aromatase activity as a mode of action for endocrine disrupting chemicals in a marine fish. Aquatic toxicology (Amsterdam, Netherlands). 2014 Feb:147():140-50. doi: 10.1016/j.aquatox.2013.12.023. Epub 2013 Dec 31
[PubMed PMID: 24418745]
[85]
Troisi R, Hyer M, Hatch EE, Titus-Ernstoff L, Palmer JR, Strohsnitter WC, Herbst AL, Adam E, Hoover RN. Medical conditions among adult offspring prenatally exposed to diethylstilbestrol. Epidemiology (Cambridge, Mass.). 2013 May:24(3):430-8. doi: 10.1097/EDE.0b013e318289bdf7. Epub
[PubMed PMID: 23474687]
[86]
Cabaton NJ, Canlet C, Wadia PR, Tremblay-Franco M, Gautier R, Molina J, Sonnenschein C, Cravedi JP, Rubin BS, Soto AM, Zalko D. Effects of low doses of bisphenol A on the metabolome of perinatally exposed CD-1 mice. Environmental health perspectives. 2013 May:121(5):586-93. doi: 10.1289/ehp.1205588. Epub 2013 Feb 21
[PubMed PMID: 23425943]
Level 3 (low-level) evidence
[87]
Virtanen HE, Bjerknes R, Cortes D, Jørgensen N, Rajpert-De Meyts E, Thorsson AV, Thorup J, Main KM. Cryptorchidism: classification, prevalence and long-term consequences. Acta paediatrica (Oslo, Norway : 1992). 2007 May:96(5):611-6
[PubMed PMID: 17462053]
[88]
Berkowitz GS, Lapinski RH, Dolgin SE, Gazella JG, Bodian CA, Holzman IR. Prevalence and natural history of cryptorchidism. Pediatrics. 1993 Jul:92(1):44-9
[PubMed PMID: 8100060]
[89]
Niedzielski JK, Oszukowska E, Słowikowska-Hilczer J. Undescended testis - current trends and guidelines: a review of the literature. Archives of medical science : AMS. 2016 Jun 1:12(3):667-77. doi: 10.5114/aoms.2016.59940. Epub 2016 May 18
[PubMed PMID: 27279862]
[90]
Suzuki Y, Sasagawa I, Itoh K, Ashida J, Ogata T. 5Alpha-reductase type 2 genes in Japanese males do not appear to be associated with cryptorchidism. Fertility and sterility. 2002 Aug:78(2):330-4
[PubMed PMID: 12137870]
[91]
Silva-Ramos M, Oliveira JM, Cabeda JM, Reis A, Soares J, Pimenta A. The CAG repeat within the androgen receptor gene and its relationship to cryptorchidism. International braz j urol : official journal of the Brazilian Society of Urology. 2006 May-Jun:32(3):330-4; discussion 335
[PubMed PMID: 16813680]
[92]
Zimmermann S, Steding G, Emmen JM, Brinkmann AO, Nayernia K, Holstein AF, Engel W, Adham IM. Targeted disruption of the Insl3 gene causes bilateral cryptorchidism. Molecular endocrinology (Baltimore, Md.). 1999 May:13(5):681-91
[PubMed PMID: 10319319]
[94]
Hutson JM, Hasthorpe S. Testicular descent and cryptorchidism: the state of the art in 2004. Journal of pediatric surgery. 2005 Feb:40(2):297-302
[PubMed PMID: 15750918]
[95]
Hutson JM, Li R, Southwell BR, Petersen BL, Thorup J, Cortes D. Germ cell development in the postnatal testis: the key to prevent malignancy in cryptorchidism? Frontiers in endocrinology. 2012:3():176. doi: 10.3389/fendo.2012.00176. Epub 2013 Jan 3
[PubMed PMID: 23316184]
[96]
Ivell R, Hartung S. The molecular basis of cryptorchidism. Molecular human reproduction. 2003 Apr:9(4):175-81
[PubMed PMID: 12651898]
[97]
Wood HM, Elder JS. Cryptorchidism and testicular cancer: separating fact from fiction. The Journal of urology. 2009 Feb:181(2):452-61. doi: 10.1016/j.juro.2008.10.074. Epub 2008 Dec 13
[PubMed PMID: 19084853]
[98]
Brauner R, Neve M, Allali S, Trivin C, Lottmann H, Bashamboo A, McElreavey K. Clinical, biological and genetic analysis of anorchia in 26 boys. PloS one. 2011:6(8):e23292. doi: 10.1371/journal.pone.0023292. Epub 2011 Aug 10
[PubMed PMID: 21853106]
[99]
Abeyaratne MR, Aherne WA, Scott JE. The vanishing testis. Lancet (London, England). 1969 Oct 18:2(7625):822-4
[PubMed PMID: 4186283]
[101]
Vinci G, Anjot MN, Trivin C, Lottmann H, Brauner R, McElreavey K. An analysis of the genetic factors involved in testicular descent in a cohort of 14 male patients with anorchia. The Journal of clinical endocrinology and metabolism. 2004 Dec:89(12):6282-5
[PubMed PMID: 15579790]
[102]
Philibert P, Zenaty D, Lin L, Soskin S, Audran F, Léger J, Achermann JC, Sultan C. Mutational analysis of steroidogenic factor 1 (NR5a1) in 24 boys with bilateral anorchia: a French collaborative study. Human reproduction (Oxford, England). 2007 Dec:22(12):3255-61
[PubMed PMID: 17940071]
[103]
Fouatih K, Belin F, Lambert AS, Bouligand J, Bouvattier C. Pubertal growth spurt in patients with bilateral anorchia after testosterone replacement therapy. Archives de pediatrie : organe officiel de la Societe francaise de pediatrie. 2019 Sep:26(6):320-323. doi: 10.1016/j.arcped.2019.06.008. Epub 2019 Jul 25
[PubMed PMID: 31353150]
[104]
Smith NM, Byard RW, Bourne AJ. Testicular regression syndrome--a pathological study of 77 cases. Histopathology. 1991 Sep:19(3):269-72
[PubMed PMID: 1916702]
Level 3 (low-level) evidence
[105]
Szmer J, Chrzan R. Decision-making challenges in children with congenital and acquired monorchism: a critical literature review. Folia medica Cracoviensia. 2019:59(1):127-136
[PubMed PMID: 31180081]
[106]
Pirgon Ö, Dündar BN. Vanishing testes: a literature review. Journal of clinical research in pediatric endocrinology. 2012 Sep:4(3):116-20. doi: 10.4274/Jcrpe.728. Epub
[PubMed PMID: 22985611]
[107]
Rozanski TA, Wojno KJ, Bloom DA. The remnant orchiectomy. The Journal of urology. 1996 Feb:155(2):712-3; discussion 714
[PubMed PMID: 8558712]
[108]
Velázquez de Cuéllar Paracchi M, Leal Orozco A, Ruíz Serrano C, Aguado Roncero P, Pérez Tejerizo G, Soriano Guillén L. [Micropenis and bilateral cryptorchidism secondary to vanishing testes syndrome]. Anales de pediatria (Barcelona, Spain : 2003). 2009 Feb:70(2):199-200. doi: 10.1016/j.anpedi.2008.09.004. Epub 2008 Dec 5
[PubMed PMID: 19217585]
[109]
Grinspon RP, Habib C, Bedecarrás P, Gottlieb S, Rey RA. Compensatory function of the remaining testis is dissociated in boys and adolescents with monorchidism. European journal of endocrinology. 2016 Mar:174(3):399-407. doi: 10.1530/EJE-15-0938. Epub 2015 Dec 15
[PubMed PMID: 26671976]
[110]
Uğuz S, Gürağaç A, Demirer Z, Yilmaz S, Aydur E. Bilateral polyorchidism with ipsilateral two undescended testes: a rare congenital anomaly. Andrologia. 2017 May:49(4):. doi: 10.1111/and.12643. Epub 2016 Jul 4
[PubMed PMID: 27373456]
[111]
Yeniyol CO, Nergiz N, Tuna A. Abdominal polyorchidism: a case report and review of the literature. International urology and nephrology. 2004:36(3):407-8
[PubMed PMID: 15783116]
Level 3 (low-level) evidence
[112]
Balasar M, Sönmez MG, Oltulu P, Kandemir A, Kılıç M, Göğer YE, Pişkin MM. Polyorchidism; unilateral, one atrophic undescended double testicles. Urology annals. 2017 Apr-Jun:9(2):208-210. doi: 10.4103/0974-7796.204190. Epub
[PubMed PMID: 28479781]
[113]
Jakhere SG, Saifi SA, Ranwaka AA. Supernumerary testis: Imaging appearance of a rare entity. Indian journal of urology : IJU : journal of the Urological Society of India. 2014 Apr:30(2):233-4. doi: 10.4103/0970-1591.126918. Epub
[PubMed PMID: 24744529]
[115]
Favorito LA, Cavalcante AG, Costa WS. Anatomic aspects of epididymis and tunica vaginalis in patients with testicular torsion. International braz j urol : official journal of the Brazilian Society of Urology. 2004 Sep-Oct:30(5):420-4
[PubMed PMID: 15610580]
[116]
Castilla EE, Sod R, Anzorena O, Texido J. Neonatal testicular torsion in two brothers. Journal of medical genetics. 1975 Mar:12(1):112-3
[PubMed PMID: 1121017]
[117]
Collins K, Broecker BH. Familial torsion of the spermatic cord. The Journal of urology. 1989 Jan:141(1):128-9
[PubMed PMID: 2908936]
[119]
Martin AD, Rushton HG. The prevalence of bell clapper anomaly in the solitary testis in cases of prior perinatal torsion. The Journal of urology. 2014 May:191(5 Suppl):1573-7. doi: 10.1016/j.juro.2013.09.013. Epub 2014 Mar 26
[PubMed PMID: 24679875]
Level 3 (low-level) evidence
[120]
Monteilh C, Calixte R, Burjonrappa S. Controversies in the management of neonatal testicular torsion: A meta-analysis. Journal of pediatric surgery. 2019 Apr:54(4):815-819. doi: 10.1016/j.jpedsurg.2018.07.006. Epub 2018 Aug 8
[PubMed PMID: 30098810]
Level 1 (high-level) evidence
[121]
Riaz-Ul-Haq M, Mahdi DE, Elhassan EU. Neonatal testicular torsion; a review article. Iranian journal of pediatrics. 2012 Sep:22(3):281-9
[PubMed PMID: 23400637]
[122]
Da Silva Rios S, Monteiro IC, Braz Dos Santos LG, Caldas NG, Chen AC, Chen JR, Silva HS. A case of swyer syndrome associated with advanced gonadal dysgerminoma involving long survival. Case reports in oncology. 2015 Jan-Apr:8(1):179-84. doi: 10.1159/000381451. Epub 2015 Mar 31
[PubMed PMID: 25960730]
Level 3 (low-level) evidence
[123]
Lipay MV, Bianco B, Verreschi IT. [Gonadal dysgenesis and tumors: genetic and clinical features]. Arquivos brasileiros de endocrinologia e metabologia. 2005 Feb:49(1):60-70
[PubMed PMID: 16544035]
[124]
King TF, Conway GS. Swyer syndrome. Current opinion in endocrinology, diabetes, and obesity. 2014 Dec:21(6):504-10. doi: 10.1097/MED.0000000000000113. Epub
[PubMed PMID: 25314337]
Level 3 (low-level) evidence
[125]
Nieschlag E. Klinefelter syndrome: the commonest form of hypogonadism, but often overlooked or untreated. Deutsches Arzteblatt international. 2013 May:110(20):347-53. doi: 10.3238/arztebl.2013.0347. Epub 2013 May 17
[PubMed PMID: 23825486]
[126]
Kumar P, Kumar N, Thakur DS, Patidar A. Male hypogonadism: Symptoms and treatment. Journal of advanced pharmaceutical technology & research. 2010 Jul:1(3):297-301. doi: 10.4103/0110-5558.72420. Epub
[PubMed PMID: 22247861]
[127]
Tüttelmann F, Gromoll J. Novel genetic aspects of Klinefelter's syndrome. Molecular human reproduction. 2010 Jun:16(6):386-95. doi: 10.1093/molehr/gaq019. Epub 2010 Mar 12
[PubMed PMID: 20228051]
[128]
Maiburg M, Repping S, Giltay J. The genetic origin of Klinefelter syndrome and its effect on spermatogenesis. Fertility and sterility. 2012 Aug:98(2):253-60. doi: 10.1016/j.fertnstert.2012.06.019. Epub 2012 Jun 29
[PubMed PMID: 22749222]
[129]
Batista RL, Costa EMF, Rodrigues AS, Gomes NL, Faria JA Jr, Nishi MY, Arnhold IJP, Domenice S, Mendonca BB. Androgen insensitivity syndrome: a review. Archives of endocrinology and metabolism. 2018 Mar-Apr:62(2):227-235. doi: 10.20945/2359-3997000000031. Epub
[PubMed PMID: 29768628]
[130]
Mendonca BB, Costa EM, Belgorosky A, Rivarola MA, Domenice S. 46,XY DSD due to impaired androgen production. Best practice & research. Clinical endocrinology & metabolism. 2010 Apr:24(2):243-62. doi: 10.1016/j.beem.2009.11.003. Epub
[PubMed PMID: 20541150]
[131]
Melo KF, Mendonça BB, Billerbeck AE, Costa EM, Latronico AC, Arnhold IJ. [Androgen insensitivity syndrome: clinical, hormonal and molecular analysis of 33 cases]. Arquivos brasileiros de endocrinologia e metabologia. 2005 Feb:49(1):87-97
[PubMed PMID: 16544039]
Level 3 (low-level) evidence
[132]
Hughes IA, Deeb A. Androgen resistance. Best practice & research. Clinical endocrinology & metabolism. 2006 Dec:20(4):577-98
[PubMed PMID: 17161333]
[133]
Tan MH, Li J, Xu HE, Melcher K, Yong EL. Androgen receptor: structure, role in prostate cancer and drug discovery. Acta pharmacologica Sinica. 2015 Jan:36(1):3-23. doi: 10.1038/aps.2014.18. Epub 2014 Jun 9
[PubMed PMID: 24909511]
[134]
Mongan NP, Tadokoro-Cuccaro R, Bunch T, Hughes IA. Androgen insensitivity syndrome. Best practice & research. Clinical endocrinology & metabolism. 2015 Aug:29(4):569-80. doi: 10.1016/j.beem.2015.04.005. Epub 2015 Apr 26
[PubMed PMID: 26303084]
[135]
Gulía C, Baldassarra S, Zangari A, Briganti V, Gigli S, Gaffi M, Signore F, Vallone C, Nucciotti R, Costantini FM, Pizzuti A, Bernardo S, Porrello A, Piergentili R. Androgen insensitivity syndrome. European review for medical and pharmacological sciences. 2018 Jun:22(12):3873-3887. doi: 10.26355/eurrev_201806_15272. Epub
[PubMed PMID: 29949163]
[136]
Cheon CK. Practical approach to steroid 5alpha-reductase type 2 deficiency. European journal of pediatrics. 2011 Jan:170(1):1-8. doi: 10.1007/s00431-010-1189-4. Epub 2010 Mar 28
[PubMed PMID: 20349245]
[137]
Mendonca BB, Domenice S, Arnhold IJ, Costa EM. 46,XY disorders of sex development (DSD). Clinical endocrinology. 2009 Feb:70(2):173-87. doi: 10.1111/j.1365-2265.2008.03392.x. Epub
[PubMed PMID: 18811725]
[138]
Speiser PW. Congenital Adrenal Hyperplasia. F1000Research. 2015:4(F1000 Faculty Rev):601. doi: 10.12688/f1000research.6543.1. Epub 2015 Aug 20
[PubMed PMID: 26339484]
[139]
Speiser PW, White PC. Congenital adrenal hyperplasia. The New England journal of medicine. 2003 Aug 21:349(8):776-88
[PubMed PMID: 12930931]
[140]
Witchel SF. Congenital Adrenal Hyperplasia. Journal of pediatric and adolescent gynecology. 2017 Oct:30(5):520-534. doi: 10.1016/j.jpag.2017.04.001. Epub 2017 Apr 24
[PubMed PMID: 28450075]
[141]
Simard J, Moisan AM, Morel Y. Congenital adrenal hyperplasia due to 3beta-hydroxysteroid dehydrogenase/Delta(5)-Delta(4) isomerase deficiency. Seminars in reproductive medicine. 2002 Aug:20(3):255-76
[PubMed PMID: 12428206]
[142]
Krone N, Dhir V, Ivison HE, Arlt W. Congenital adrenal hyperplasia and P450 oxidoreductase deficiency. Clinical endocrinology. 2007 Feb:66(2):162-72
[PubMed PMID: 17223983]
[143]
New MI. Male pseudohermaphroditism due to 17 alpha-hydroxylase deficiency. The Journal of clinical investigation. 1970 Oct:49(10):1930-41
[PubMed PMID: 5456802]
[144]
Hauffa BP, Miller WL, Grumbach MM, Conte FA, Kaplan SL. Congenital adrenal hyperplasia due to deficient cholesterol side-chain cleavage activity (20, 22-desmolase) in a patient treated for 18 years. Clinical endocrinology. 1985 Nov:23(5):481-93
[PubMed PMID: 3841304]
[145]
Bose HS, Sugawara T, Strauss JF 3rd, Miller WL, International Congenital Lipoid Adrenal Hyperplasia Consortium. The pathophysiology and genetics of congenital lipoid adrenal hyperplasia. The New England journal of medicine. 1996 Dec 19:335(25):1870-8
[PubMed PMID: 8948562]
[146]
Metherell LA, Naville D, Halaby G, Begeot M, Huebner A, Nürnberg G, Nürnberg P, Green J, Tomlinson JW, Krone NP, Lin L, Racine M, Berney DM, Achermann JC, Arlt W, Clark AJ. Nonclassic lipoid congenital adrenal hyperplasia masquerading as familial glucocorticoid deficiency. The Journal of clinical endocrinology and metabolism. 2009 Oct:94(10):3865-71. doi: 10.1210/jc.2009-0467. Epub 2009 Sep 22
[PubMed PMID: 19773404]
[147]
Primatesta P, Goldacre MJ. Inguinal hernia repair: incidence of elective and emergency surgery, readmission and mortality. International journal of epidemiology. 1996 Aug:25(4):835-9
[PubMed PMID: 8921464]
[148]
Ruhl CE, Everhart JE. Risk factors for inguinal hernia among adults in the US population. American journal of epidemiology. 2007 May 15:165(10):1154-61
[PubMed PMID: 17374852]
[149]
Tanyel FC, Müftüoglu S, Dagdeviren A, Kaymaz FF, Büyükpamukçu N. Myofibroblasts defined by electron microscopy suggest the dedifferentiation of smooth muscle within the sac walls associated with congenital inguinal hernia. BJU international. 2001 Feb:87(3):251-5
[PubMed PMID: 11167652]
[150]
Rodprasert W, Virtanen HE, Mäkelä JA, Toppari J. Hypogonadism and Cryptorchidism. Frontiers in endocrinology. 2019:10():906. doi: 10.3389/fendo.2019.00906. Epub 2020 Jan 15
[PubMed PMID: 32010061]
[151]
Foresta C, Zuccarello D, Garolla A, Ferlin A. Role of hormones, genes, and environment in human cryptorchidism. Endocrine reviews. 2008 Aug:29(5):560-80. doi: 10.1210/er.2007-0042. Epub 2008 Apr 24
[PubMed PMID: 18436703]
[152]
Ramareddy RS, Alladi A, Siddappa OS. Ectopic testis in children: experience with seven cases. Journal of pediatric surgery. 2013 Mar:48(3):538-41. doi: 10.1016/j.jpedsurg.2012.10.005. Epub
[PubMed PMID: 23480908]
Level 3 (low-level) evidence
[153]
Ferro F, Lais A, Matarazzo E, Capozza N, Caione P. [Retractile testis and gliding testis. Two distinct clinical entities]. Minerva urologica e nefrologica = The Italian journal of urology and nephrology. 1996 Sep:48(3):145-9
[PubMed PMID: 8966651]
[154]
. Cryptorchidism: a prospective study of 7500 consecutive male births, 1984-8. John Radcliffe Hospital Cryptorchidism Study Group. Archives of disease in childhood. 1992 Jul:67(7):892-9
[PubMed PMID: 1355643]
[155]
Barteczko KJ, Jacob MI. The testicular descent in human. Origin, development and fate of the gubernaculum Hunteri, processus vaginalis peritonei, and gonadal ligaments. Advances in anatomy, embryology, and cell biology. 2000:156():III-X, 1-98
[PubMed PMID: 11008363]
Level 3 (low-level) evidence
[158]
Aynsley-Green A, Zachmann M, Illig R, Rampini S, Prader A. Congenital bilateral anorchia in childhood: a clinical, endocrine and therapeutic evaluation of twenty-one cases. Clinical endocrinology. 1976 Jul:5(4):381-91
[PubMed PMID: 786507]
Level 3 (low-level) evidence
[159]
Zenaty D, Dijoud F, Morel Y, Cabrol S, Mouriquand P, Nicolino M, Bouvatier C, Pinto G, Lecointre C, Pienkowski C, Soskin S, Bost M, Bertrand AM, El-Ghoneimi A, Nihoul-Fekete C, Léger J. Bilateral anorchia in infancy: occurence of micropenis and the effect of testosterone treatment. The Journal of pediatrics. 2006 Nov:149(5):687-91
[PubMed PMID: 17095345]
[160]
De Marchi M, Campagnoli C, Ghiringhello B, Ponzio G, Carbonara A. Gonadal agenesis in a phenotypically normal female with positive H-Y antigen. Human genetics. 1981:56(3):417-9
[PubMed PMID: 7239525]
[161]
Shadpour P, Kashi AH, Arvin A. Scrotal testis size in unilateral non-palpable cryptorchidism, what it can and cannot tell: Study of a Middle Eastern population. Journal of pediatric urology. 2017 Jun:13(3):268.e1-268.e6. doi: 10.1016/j.jpurol.2016.12.013. Epub 2017 Jan 21
[PubMed PMID: 28254240]
[162]
Hodhod A, Capolicchio JP, Jednak R, El-Sherbiny M. Testicular hypertrophy as a predictor for contralateral monorchism: Retrospective review of prospectively recorded data. Journal of pediatric urology. 2016 Feb:12(1):34.e1-5. doi: 10.1016/j.jpurol.2015.06.010. Epub 2015 Jul 30
[PubMed PMID: 26279100]
Level 2 (mid-level) evidence
[163]
Kheirandish P, Chinegwundoh F. An unusual case of triorchidism. JRSM short reports. 2010 Nov 25:1(6):55. doi: 10.1258/shorts.2010.010067. Epub 2010 Nov 25
[PubMed PMID: 21234118]
Level 3 (low-level) evidence
[164]
Arlen AM, Holzman SA, Weiss AD, Garola RE, Cerwinka WH. Functional supernumerary testis in a child with testicular torsion and review of polyorchidism. Pediatric surgery international. 2014 May:30(5):565-8. doi: 10.1007/s00383-014-3485-y. Epub 2014 Feb 21
[PubMed PMID: 24557155]
[165]
Abduljabbar AH. A Case Report: Triorchidism; is a Rare Mistaken Cause for Extra Testicular Neoplasm. Urology case reports. 2015 May:3(3):89-91. doi: 10.1016/j.eucr.2015.02.002. Epub 2015 Mar 14
[PubMed PMID: 26793513]
Level 3 (low-level) evidence
[166]
Kharrazi SM, Rahmani MR, Sakipour M, Khoob S. Polyorchidism: a case report and review of literature. Urology journal. 2006 Summer:3(3):180-3
[PubMed PMID: 17559037]
Level 3 (low-level) evidence
[167]
Bergholz R, Wenke K. Polyorchidism: a meta-analysis. The Journal of urology. 2009 Nov:182(5):2422-7. doi: 10.1016/j.juro.2009.07.063. Epub 2009 Sep 17
[PubMed PMID: 19765760]
Level 1 (high-level) evidence
[168]
Zhao LC, Lautz TB, Meeks JJ, Maizels M. Pediatric testicular torsion epidemiology using a national database: incidence, risk of orchiectomy and possible measures toward improving the quality of care. The Journal of urology. 2011 Nov:186(5):2009-13. doi: 10.1016/j.juro.2011.07.024. Epub 2011 Sep 23
[PubMed PMID: 21944120]
Level 2 (mid-level) evidence
[169]
Huang WY, Chen YF, Chang HC, Yang TK, Hsieh JT, Huang KH. The incidence rate and characteristics in patients with testicular torsion: a nationwide, population-based study. Acta paediatrica (Oslo, Norway : 1992). 2013 Aug:102(8):e363-7. doi: 10.1111/apa.12275. Epub 2013 May 13
[PubMed PMID: 23611668]
[170]
Lee SM, Huh JS, Baek M, Yoo KH, Min GE, Lee HL, Lee DG. A nationwide epidemiological study of testicular torsion in Korea. Journal of Korean medical science. 2014 Dec:29(12):1684-7. doi: 10.3346/jkms.2014.29.12.1684. Epub 2014 Nov 21
[PubMed PMID: 25469070]
Level 2 (mid-level) evidence
[172]
Yiee JH, Chang L, Kaplan A, Kwan L, Chung PJ, Litwin MS. Patterns of care in testicular torsion: influence of hospital transfer on testicular outcomes. Journal of pediatric urology. 2013 Dec:9(6 Pt A):713-20. doi: 10.1016/j.jpurol.2013.06.003. Epub 2013 Jul 27
[PubMed PMID: 23896260]
[173]
Pogorelić Z, Mrklić I, Jurić I, Biočić M, Furlan D. Testicular torsion in the inguinal canal in children. Journal of pediatric urology. 2013 Dec:9(6 Pt A):793-7. doi: 10.1016/j.jpurol.2012.10.013. Epub 2012 Oct 31
[PubMed PMID: 23123082]
[174]
Yazbeck S, Patriquin HB. Accuracy of Doppler sonography in the evaluation of acute conditions of the scrotum in children. Journal of pediatric surgery. 1994 Sep:29(9):1270-2
[PubMed PMID: 7807366]
[175]
Wikström AM, Hoei-Hansen CE, Dunkel L, Rajpert-De Meyts E. Immunoexpression of androgen receptor and nine markers of maturation in the testes of adolescent boys with Klinefelter syndrome: evidence for degeneration of germ cells at the onset of meiosis. The Journal of clinical endocrinology and metabolism. 2007 Feb:92(2):714-9
[PubMed PMID: 17148558]
[176]
Avendaño A, Paradisi I, Cammarata-Scalisi F, Callea M. 5-α-Reductase type 2 deficiency: is there a genotype-phenotype correlation? A review. Hormones (Athens, Greece). 2018 Jun:17(2):197-204. doi: 10.1007/s42000-018-0013-9. Epub 2018 Apr 20
[PubMed PMID: 29858846]
[177]
Byne W. Developmental endocrine influences on gender identity: implications for management of disorders of sex development. The Mount Sinai journal of medicine, New York. 2006 Nov:73(7):950-9
[PubMed PMID: 17195880]
[178]
Thilén A, Larsson A. Congenital adrenal hyperplasia in Sweden 1969-1986. Prevalence, symptoms and age at diagnosis. Acta paediatrica Scandinavica. 1990 Feb:79(2):168-75
[PubMed PMID: 2321478]
[179]
Bois E, Mornet E, Chompret A, Feingold J, Hochez J, Goulet V. [Congenital adrenal hyperplasia (21-OH) in France. Population genetics]. Archives francaises de pediatrie. 1985 Mar:42(3):175-9
[PubMed PMID: 3873926]
[180]
Yanase T, Simpson ER, Waterman MR. 17 alpha-hydroxylase/17,20-lyase deficiency: from clinical investigation to molecular definition. Endocrine reviews. 1991 Feb:12(1):91-108
[PubMed PMID: 2026124]