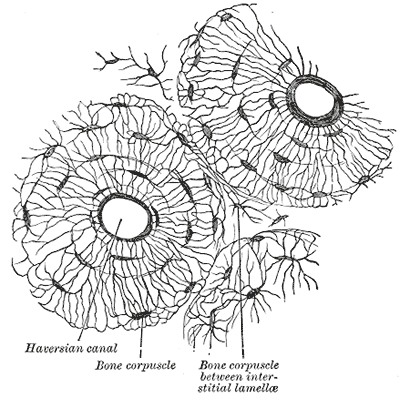
[1]
Hong AR, Kim SW. Effects of Resistance Exercise on Bone Health. Endocrinology and metabolism (Seoul, Korea). 2018 Dec:33(4):435-444. doi: 10.3803/EnM.2018.33.4.435. Epub
[PubMed PMID: 30513557]
[3]
Burrow K , Young W , Carne A , McConnell M , Hammer N , Scholze M , Bekhit AE . Consumption of sheep milk compared to cow milk can affect trabecular bone ultrastructure in a rat model. Food & function. 2019 Jan 22:10(1):163-171. doi: 10.1039/c8fo01598h. Epub
[PubMed PMID: 30516196]
[4]
Xue N,Ding X,Huang R,Jiang R,Huang H,Pan X,Min W,Chen J,Duan JA,Liu P,Wang Y, Bone Tissue Engineering in the Treatment of Bone Defects. Pharmaceuticals (Basel, Switzerland). 2022 Jul 17
[PubMed PMID: 35890177]
[5]
Augusto ACL, Goes PCK, Flores DV, Costa MAF, Takahashi MS, Rodrigues ACO, Padula LC, Gasparetto TD, Nogueira-Barbosa MH, Aihara AY. Imaging Review of Normal and Abnormal Skeletal Maturation. Radiographics : a review publication of the Radiological Society of North America, Inc. 2022 May-Jun:42(3):861-879. doi: 10.1148/rg.210088. Epub 2022 Feb 25
[PubMed PMID: 35213260]
[6]
Marahleh A, Kitaura H, Ohori F, Noguchi T, Mizoguchi I. The osteocyte and its osteoclastogenic potential. Frontiers in endocrinology. 2023:14():1121727. doi: 10.3389/fendo.2023.1121727. Epub 2023 May 24
[PubMed PMID: 37293482]
[7]
Grünewald TA, Johannes A, Wittig NK, Palle J, Rack A, Burghammer M, Birkedal H. Bone mineral properties and 3D orientation of human lamellar bone around cement lines and the Haversian system. IUCrJ. 2023 Mar 1:10(Pt 2):189-198. doi: 10.1107/S2052252523000866. Epub 2023 Mar 1
[PubMed PMID: 36786504]
[8]
Ono T, Hayashi M, Sasaki F, Nakashima T. RANKL biology: bone metabolism, the immune system, and beyond. Inflammation and regeneration. 2020:40():2. doi: 10.1186/s41232-019-0111-3. Epub 2020 Feb 7
[PubMed PMID: 32047573]
[9]
Da W, Tao L, Zhu Y. The Role of Osteoclast Energy Metabolism in the Occurrence and Development of Osteoporosis. Frontiers in endocrinology. 2021:12():675385. doi: 10.3389/fendo.2021.675385. Epub 2021 May 12
[PubMed PMID: 34054735]
[10]
Gavali S, Gupta MK, Daswani B, Wani MR, Sirdeshmukh R, Khatkhatay MI. Estrogen enhances human osteoblast survival and function via promotion of autophagy. Biochimica et biophysica acta. Molecular cell research. 2019 Sep:1866(9):1498-1507. doi: 10.1016/j.bbamcr.2019.06.014. Epub 2019 Jun 28
[PubMed PMID: 31255720]
[12]
Chen J, Hendriks M, Chatzis A, Ramasamy SK, Kusumbe AP. Bone Vasculature and Bone Marrow Vascular Niches in Health and Disease. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research. 2020 Nov:35(11):2103-2120. doi: 10.1002/jbmr.4171. Epub 2020 Sep 23
[PubMed PMID: 32845550]
[13]
Brazill JM, Beeve AT, Craft CS, Ivanusic JJ, Scheller EL. Nerves in Bone: Evolving Concepts in Pain and Anabolism. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research. 2019 Aug:34(8):1393-1406. doi: 10.1002/jbmr.3822. Epub 2019 Jul 26
[PubMed PMID: 31247122]
[14]
Liu S, Liu S, Li S, Liang B, Han X, Liang Y, Wei X. Nerves within bone and their application in tissue engineering of bone regeneration. Frontiers in neurology. 2022:13():1085560. doi: 10.3389/fneur.2022.1085560. Epub 2023 Feb 2
[PubMed PMID: 36818724]
[17]
Condino S, Turini G, Parchi PD, Viglialoro RM, Piolanti N, Gesi M, Ferrari M, Ferrari V. How to Build a Patient-Specific Hybrid Simulator for Orthopaedic Open Surgery: Benefits and Limits of Mixed-Reality Using the Microsoft HoloLens. Journal of healthcare engineering. 2018:2018():5435097. doi: 10.1155/2018/5435097. Epub 2018 Nov 1
[PubMed PMID: 30515284]
[18]
Quevedo González FJ, Lipman JD, Lo D, De Martino I, Sculco PK, Sculco TP, Catani F, Wright TM. Mechanical performance of cementless total knee replacements: It is not all about the maximum loads. Journal of orthopaedic research : official publication of the Orthopaedic Research Society. 2019 Feb:37(2):350-357. doi: 10.1002/jor.24194. Epub 2019 Jan 3
[PubMed PMID: 30499604]
[19]
Meeta M, Harinarayan CV, Marwah R, Sahay R, Kalra S, Babhulkar S. Clinical Practice Guidelines on Postmenopausal Osteoporosis: *An Executive Summary and Recommendations - Update 2019-2020. Journal of mid-life health. 2020 Apr-Jun:11(2):96-112. doi: 10.4103/jmh.JMH_143_20. Epub 2020 Aug 10
[PubMed PMID: 33281419]
Level 1 (high-level) evidence
[20]
Jeys LM, Thorne CJ, Parry M, Gaston CL, Sumathi VP, Grimer JR. A Novel System for the Surgical Staging of Primary High-grade Osteosarcoma: The Birmingham Classification. Clinical orthopaedics and related research. 2017 Mar:475(3):842-850. doi: 10.1007/s11999-016-4851-y. Epub
[PubMed PMID: 27138473]
[21]
Rojas GA, Hubbard AK, Diessner BJ, Ribeiro KB, Spector LG. International trends in incidence of osteosarcoma (1988-2012). International journal of cancer. 2021 Sep 1:149(5):1044-1053. doi: 10.1002/ijc.33673. Epub 2021 May 17
[PubMed PMID: 33963769]
[22]
Al-Chalabi MMM, Jamil I, Wan Sulaiman WA. Unusual Location of Bone Tumor Easily Misdiagnosed: Distal Radius Osteosarcoma Treated as Osteomyelitis. Cureus. 2021 Nov:13(11):e19905. doi: 10.7759/cureus.19905. Epub 2021 Nov 25
[PubMed PMID: 34976513]
[23]
Cersosimo F, Lonardi S, Bernardini G, Telfer B, Mandelli GE, Santucci A, Vermi W, Giurisato E. Tumor-Associated Macrophages in Osteosarcoma: From Mechanisms to Therapy. International journal of molecular sciences. 2020 Jul 23:21(15):. doi: 10.3390/ijms21155207. Epub 2020 Jul 23
[PubMed PMID: 32717819]
[24]
Pu Y, Ji Q. Tumor-Associated Macrophages Regulate PD-1/PD-L1 Immunosuppression. Frontiers in immunology. 2022:13():874589. doi: 10.3389/fimmu.2022.874589. Epub 2022 May 3
[PubMed PMID: 35592338]
[25]
Mao X, Xu J, Wang W, Liang C, Hua J, Liu J, Zhang B, Meng Q, Yu X, Shi S. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Molecular cancer. 2021 Oct 11:20(1):131. doi: 10.1186/s12943-021-01428-1. Epub 2021 Oct 11
[PubMed PMID: 34635121]
Level 3 (low-level) evidence
[26]
Barry KC, Hsu J, Broz ML, Cueto FJ, Binnewies M, Combes AJ, Nelson AE, Loo K, Kumar R, Rosenblum MD, Alvarado MD, Wolf DM, Bogunovic D, Bhardwaj N, Daud AI, Ha PK, Ryan WR, Pollack JL, Samad B, Asthana S, Chan V, Krummel MF. A natural killer-dendritic cell axis defines checkpoint therapy-responsive tumor microenvironments. Nature medicine. 2018 Aug:24(8):1178-1191. doi: 10.1038/s41591-018-0085-8. Epub 2018 Jun 25
[PubMed PMID: 29942093]
[27]
Miwa S, Shirai T, Yamamoto N, Hayashi K, Takeuchi A, Igarashi K, Tsuchiya H. Current and Emerging Targets in Immunotherapy for Osteosarcoma. Journal of oncology. 2019:2019():7035045. doi: 10.1155/2019/7035045. Epub 2019 Jan 1
[PubMed PMID: 30693030]
[28]
Papalexis N, Savarese LG, Peta G, Errani C, Tuzzato G, Spinnato P, Ponti F, Miceli M, Facchini G. The New Ice Age of Musculoskeletal Intervention: Role of Percutaneous Cryoablation in Bone and Soft Tissue Tumors. Current oncology (Toronto, Ont.). 2023 Jul 17:30(7):6744-6770. doi: 10.3390/curroncol30070495. Epub 2023 Jul 17
[PubMed PMID: 37504355]
[32]
Siismets EM, Hatch NE. Cranial Neural Crest Cells and Their Role in the Pathogenesis of Craniofacial Anomalies and Coronal Craniosynostosis. Journal of developmental biology. 2020 Sep 9:8(3):. doi: 10.3390/jdb8030018. Epub 2020 Sep 9
[PubMed PMID: 32916911]
[33]
Losco M, Ceglia MJ, Lazzarini F, De Biase P, Buzzi R. A rare case of avulsion fracture of the iliac crest apophysis in a young female athlete. Trauma case reports. 2019 Dec:24():100257. doi: 10.1016/j.tcr.2019.100257. Epub 2019 Nov 7
[PubMed PMID: 31737774]
Level 3 (low-level) evidence