Continuing Education Activity
Hypokalemia is a common electrolyte disturbance not given as much attention as hyperkalemia. Hypokalemia has associations with cardiac disease, renal failure, malnutrition, and shock. Additional risk factors, such as hypothermia and increased blood cell production, contribute to this condition's complexity. Clinicians must thus promptly triage, refer, and replete electrolytes to prevent long-term sequelae and mortality. Understanding the nuances of the condition's manifestations and treatment strategies is crucial to enhancing patient outcomes.
This activity for healthcare professionals is designed to enhance learner's proficiency in evaluating and managing hypokalemia. After participation, learners gain insights into hypokalemia's presentations, diagnostic features, and intervention strategies, enabling them to collaborate effectively within an interprofessional team caring for people with this condition.
Objectives:
Identify the signs and symptoms indicative of hypokalemia.
Develop a clinically guided diagnostic plan for a patient with hypokalemia.
Create a personalized management strategy for a patient with hypokalemia.
Develop effective communication protocols within an interprofessional team caring for patients with hypokalemia, specifically when formulating immediate and long-term plans that can improve health outcomes.
Introduction
Hypokalemia Overview
Hypokalemia is one of the most common electrolyte disturbances seen in clinical practice. The condition is more prevalent than hyperkalemia, though most hypokalemia cases are mild. Although variation exists, an acceptable lower limit for normal serum potassium is 3.5 mmol/L.[1] Hypokalemia is classified according to severity. A serum potassium level of 3 to 3.4 mmol/L is classified as mild. A serum potassium level of 2.5 to 3 mmol/L is classified as moderate. Serum potassium levels less than 2.5 mmol/L are classified as severe.
Symptoms of hypokalemia vary depending on the severity and underlying cause but may include muscle weakness, fatigue, cramping, palpitations, and constipation. Severe hypokalemia can lead to life-threatening complications, such as fatal arrhythmias or respiratory muscle paralysis.
Hypokalemia must be addressed promptly to prevent complications. Long-term management may involve monitoring potassium levels, adjusting medications, and addressing any underlying medical conditions contributing to hypokalemia.
Summary of Potassium Regulation
Potassium is a predominantly intracellular electrolyte, the most abundant cation in cell regulation and cellular processes. The fraction of potassium in the extracellular fluid is small. Thus, plasma or serum levels are unreliable indicators of total body potassium stores. The body's potassium level is tightly regulated by the kidneys, digestive tract, endocrine system, and cells actively participating in transcellular shifts, such as the skeletal muscles. Cellular potassium uptake is promoted by alkalemia, insulin, β-adrenergic stimulation, aldosterone, and xanthines, such as caffeine.
Potassium deficiency may arise from various causes, such as poor nutrition, endocrine disorders, diarrhea, medication intake, and prior surgeries. Hypokalemia may thus be a manifestation of a more serious disorder.
Etiology
Hypokalemia can arise from a myriad of causes that may be broadly categorized into the following:
- Decreased potassium intake
- Poor nutrition or specific dietary restrictions
- Eating disorders
- Increased intracellular uptake
- Insulin injection
- β-adrenergic inhalation
- Increased potassium losses
- Excessive sweating
- Loop diuretic intake
- Renal disorders [2][3]
- Diarrhea
- Vomiting
Reduced intake alone rarely results in hypokalemia due to the kidneys' ability to minimize potassium excretion. However, poor intake can contribute to potassium lowering from other causes, such as chronic diuretic therapy.
The causes of renal hypokalemia include chloride-responsive metabolic alkalosis, which may result from chloride depletion due to vomiting or nasogastric suction. Chloride-resistant metabolic alkalosis may be due to true mineralocorticoid excesses, such as the following:
- Primary hyperaldosteronism (Conn syndrome) arising from adrenal adenoma, adrenal carcinoma, or bilateral adrenal hyperplasia
- Congenital adrenal hyperplasia from 11-β or 17-α-hydroxylase deficiency
- Renin-secreting tumors
- Ectopic corticotropin stimulation ectopic adrenocorticotropic hormone syndrome
- Cushing disease or Cushing syndrome
- Hereditary glucocorticoid-responsive aldosteronism
- Hypertensive emergency and malignant hypertension
- Renovascular hypertension renal artery stenosis
- Vasculitis
Genetic conditions that can cause renal potassium loss include the following:
- Liddle syndrome
- Syndrome of apparent mineralocorticoid excess
- Gitelman syndrome
- Bartter syndrome [4]
Other conditions that can lead to renal hypokalemia causes include the following:
- Fanconi syndrome
- Renal tubular acidosis (RTA), especially type 1 RTA (manifests with metabolic acidosis)
- Hypomagnesemia, metabolic acidosis due to type I RTA
- Uncontrolled diabetes mellitus
- Ureteral diversion into the colon (ureterosigmoidostomy).[5][6]
Gastrointestinal losses are a common cause of hypokalemia, with severe or chronic diarrhea being the most common extrarenal etiology. Other sources include vomiting and certain conditions such as tumors, infections, intestinal obstruction, bowel diversion, and cancer therapy. Additionally, chronic laxative abuse, clay (bentonite) ingestion, and malabsorption can lead to hypokalemia. Some drugs, such as sodium polystyrene sulfonate, can increase potassium loss in stool and contribute to hypokalemia.[7]
Epidemiology
Hypokalemia is generally associated with cardiac disease, renal failure, malnutrition, and shock. Additionally, psychiatric patients are at risk for hypokalemia due to disordered eating and drug side effects. This condition is prevalent in hospitalized patients, particularly children and febrile and critically ill individuals. In developing countries, an increased risk of mortality has been observed in children when severe hypokalemia is associated with diarrhea and severe malnutrition.
Pathophysiology
Hypokalemia's symptoms are primarily attributed to potassium's role in various physiological processes. Potassium maintains cell membrane potential, nerve impulse conduction, muscle function (including skeletal, smooth, and cardiac muscles), and acid-base balance. Abnormally low potassium levels can lead to disturbances in these physiological processes, resulting in a wide range of symptoms.
Potassium is essential for maintaining normal muscle function. Decreased potassium levels can disrupt neuromuscular transmission and impair muscle contraction. Severe hypokalemia can lead to paralysis, particularly in extreme cases where potassium levels are critically low. This paralysis may affect respiratory muscles, leading to respiratory failure. Severe hypokalemia can also result in muscle cramps, rhabdomyolysis, and myoglobinuria. Rhabdomyolysis results from muscle fibers breaking down and releasing their contents into the bloodstream, leading to kidney damage and potentially life-threatening complications. Potassium deficiency can affect smooth muscle function in the gastrointestinal tract, leading to impaired intestinal motility and constipation.
Prolonged hypokalemia can cause structural and functional kidney changes, including impaired concentrating ability, increased ammonia production, altered sodium reabsorption, and increased bicarbonate absorption. Hypokalemia may also result in glucose intolerance by reducing insulin secretion.
Potassium plays a crucial role in regulating cardiac electrical activity. Hypokalemia can lead to cardiac membrane potential alterations and repolarization delay, predisposing individuals to cardiac arrhythmias such as ventricular ectopy, atrial fibrillation, and potentially life-threatening ventricular tachycardia or fibrillation. In severe hypokalemia cases, profound cardiac electrical activity disturbances can result in cardiac arrest and sudden death if left untreated.
Hypokalemia can affect the heart's response to Digitalis medications, which are used to treat heart failure and other cardiac conditions. Potassium deficiency may increase Digitalis to toxic levels in the bloodstream. Hypokalemia can also reduce insulin receptor sensitivity, leading to insulin resistance and potentially contributing to the development of type 2 diabetes.[8][9][10]
History and Physical
History
Hypokalemia evaluation hinges on comprehensive history-taking and a focused physical examination. Symptoms indicative of potassium deficiency include muscle weakness, cramps, spasms, fatigue, palpitations, constipation, and abdominal discomfort. Clues to the etiology of potassium deficiency may be elicited, such as diuretic or laxative use, poor intake, vomiting, or reports of a hereditary condition.
Clinical symptoms of hypokalemia are inapparent until the serum potassium level is less than 3 mmol/L unless an abrupt change occurs or the patient has a disease process potentiated by hypokalemia. Symptom severity often correlates with the degree and duration of hypokalemia. Symptoms resolve with hypokalemia correction. Significant muscle weakness occurs at serum potassium levels below 2.5 mmol/L but can occur at higher levels if the onset is acute.[11]
Hypokalemia manifests as an ascending paralysis, with the lower extremities developing profound weakness before the trunk and upper extremities. The condition may affect the respiratory muscles, which can lead to respiratory failure and death. Patients may manifest with nausea, vomiting, ileus, and abdominal distension.
Hypokalemic periodic paralysis is a rare inherited or acquired neuromuscular disorder caused by acute transcellular potassium shifts into the cells. The condition is characterized by potentially fatal episodes of muscle weakness that can affect the respiratory muscles.
Physical Examination
Hypokalemia may or may not be associated with hypertension. Thus, blood pressure measurement is essential. A few relevant examples are included in Table 1. (see Table 1. Hypokalemia Differential Diagnosis Based on Blood Pressure Level)
Table 1. Hypokalemia Differential Diagnosis Based on Blood Pressure Level
|
Hypokalemia with hypertension
- Liddle syndrome
- Conn syndrome
- Cushing syndrome
- 11-β hydroxylase deficiency
- Licorice excess
- Carbenoxolone (ulcer medication) intake [12]
|
Hypokalemia without hypertension
- Diuretics
- Diarrhea
- Vomiting
- Bartter syndrome
- Gitelman syndrome
- RTA types 1 and 2 [13]
|
Cardiovascular examination may identify arrhythmias and signs of heart failure. Abdominal examination may detect hypoactive bowel sounds and abdominal tenderness. Muscle assessment shows profound muscle weakness. Consequently, poor chest expansion during inspiration may be noted. Muscle evaluation can differentiate between acute and chronic hypokalemia, as signs of muscle wasting or atrophy are more common in chronic potassium deficiency.
Neurological examination may reveal abnormal reflexes and paresthesias. Deep tendon reflexes may be decreased or increased depending on the etiology or presence of comorbidities. For example, hypokalemic periodic paralysis, malnutrition, and diuretic use often present with hyporeflexia due to impaired motor neuron excitability. Meanwhile, alkalosis and β-adrenergic inhalation manifest with hyperreflexia due to increased neuronal excitability.
Hypokalemia may be accompanied by other physical clues if manifesting as part of a systemic illness. For example, patients with Cushing disease classically present with a buffalo hump, moon facies, and abdominal striae. Malnourished patients may have other signs of vitamin deficiencies, such as emaciation, nonhealing wounds, and dental problems.
Evaluation
The etiology of hypokalemia is evident from the patient’s history. However, diagnostic testing allows for assessing the condition's severity and guides corrective measures, especially if other abnormalities are present. Diagnostic modalities include blood and urine tests, electrocardiography (ECG), and imaging studies. A myriad of tests may be obtained to determine hypokalemia's etiology, but clinical findings should guide the diagnostic workup.
Laboratory Tests
Serum electrolytes can help assess hypokalemia severity and detect other electrolyte deficiencies. For example, alkalosis can present with hypokalemia, hypocalcemia, and hypomagnesemia. Chloride levels may be elevated in renal potassium wasting and depressed in alkalosis from vomiting.
Urinary potassium excretion assessment can help distinguish renal losses from other causes. Potassium excretion is ideally measured via a 24-hour urine collection. Excretion of more than 30 mEq of potassium per day indicates inappropriate renal potassium loss. Alternative measurement methods include a spot urine potassium concentration or urine potassium-to-creatinine ratio. A urine potassium concentration greater than 15 or a ratio greater than 13 mEq/mmol of creatinine, respectively, indicates inappropriate renal potassium loss.
After evaluating for renal potassium wasting, acid-base status may be assessed by obtaining arterial blood gases. The existence of metabolic acidosis or alkalosis with or without renal potassium wasting can further narrow down the differential diagnosis (see Table 2. Hypokalemia Differential Diagnosis Based on Blood pH).
Table 2. Hypokalemia Differential Diagnosis Based on Blood pH
|
Major causes of metabolic alkalosis associated with hypokalemia
- Conn syndrome: often with hypertension
- Vomiting: may present with poor capillary refill, hypotension, and sunken eyeballs if severely dehydrated
- Diuretic use: may be related to chronic use or overdose
- Hereditary renal channelopathies
- Liddle syndrome: presents with high blood pressure, low renin, and low aldosterone level
- Gitelman or Bartter syndrome: may be asymptomatic, usually normotensive [14][15][16]
|
|
Major causes of metabolic nonanion-gap acidosis associated with hypokalemia
- RTA
- Fanconi syndrome (proximal RTA type 2): may present with weak bones, glucosuria, aminoaciduria, and phosphaturia
- Autoimmune disease (distal RTA type 1): present with a history of osteoporosis and nephrolithiasis (calcium phosphate)
- Diarrhea: serum chloride may be normal; signs of dehydration may be present
- Diabetic ketoacidosis: may present with generalized weakness, altered sensorium, and tachypnea
|
Electrocardiogram
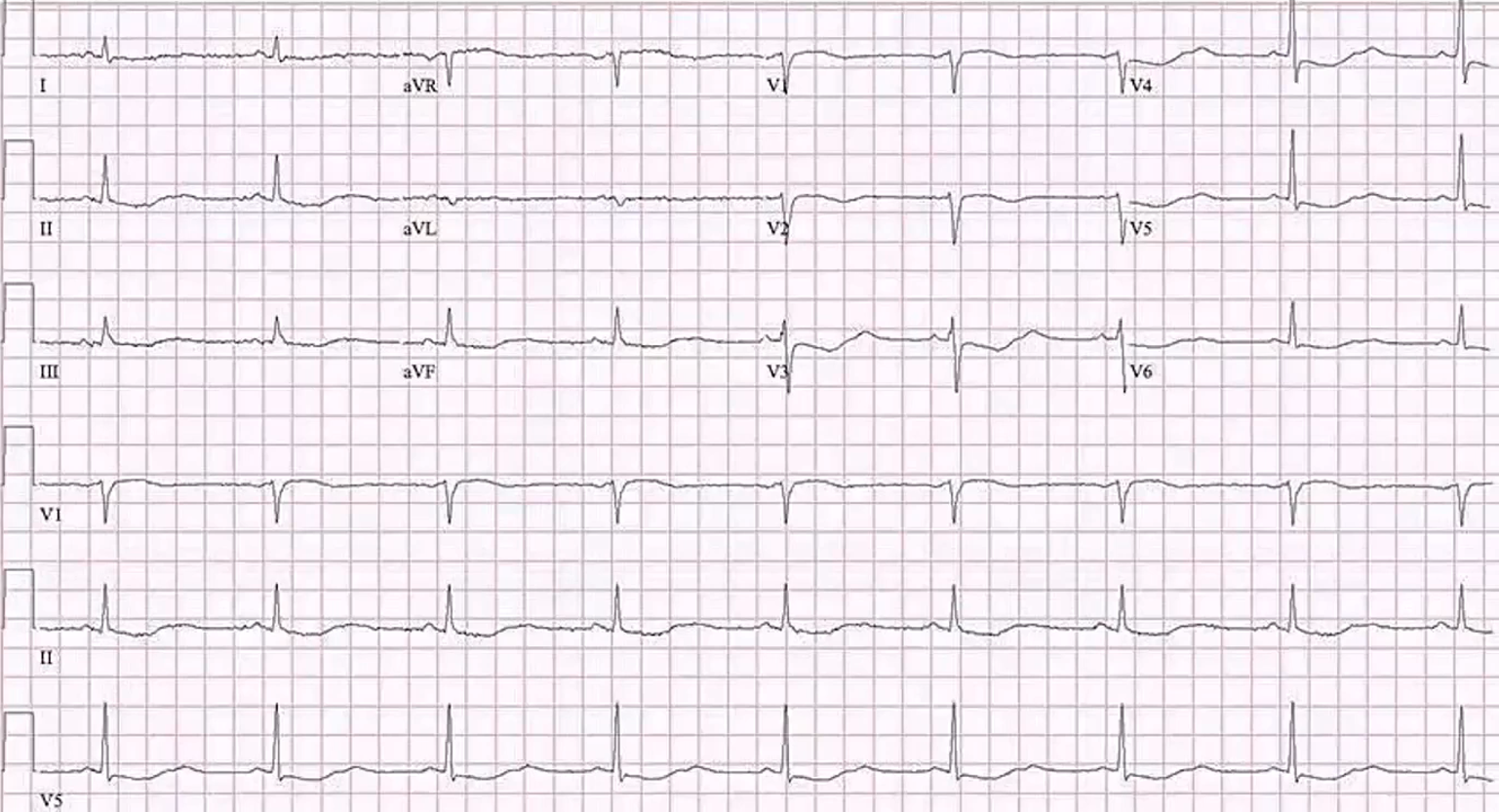
Hypokalemia can cause a variety of arrhythmias, such as premature atrial complex, premature ventricular beats, sinus bradycardia, paroxysmal atrial or junctional tachycardia, atrioventricular block, ventricular tachycardia or fibrillation.[17] The most common ECG abnormalities associated with potassium deficiency include decreased T wave amplitude, depressed ST segment, appearance of a U wave, and prolonged QT interval. T-wave flattening initially manifests, followed by ST depression and U wave emergence. PR and QT intervals may also be prolonged. The U wave is often seen in the lateral precordial leads of V4 to V6 but may be difficult to distinguish from the T wave (see Image. ECG in a Patient With Hypokalemia). However, not all patients present with these classic changes.
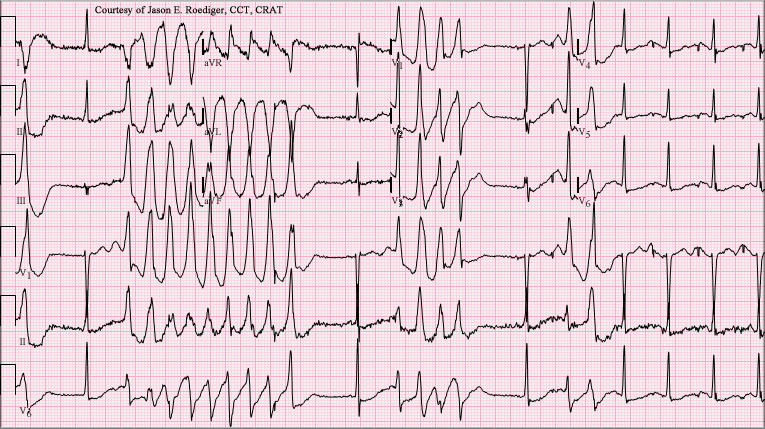
Besides the above abnormalities, severe hypokalemia can cause torsades de pointes and ventricular tachycardia (see Image. Ventricular Tachycardia With 1:1 Ventriculoatrial Conduction). The risk of arrhythmias is highest in older patients, those with heart disease, and those prescribed Digoxin or antiarrhythmics. Anesthesia carries a heightened risk of dysrhythmias and impaired cardiac contractility in individuals with hypokalemia, with the risk being more pronounced in acute than chronic hypokalemia.
Although ECG abnormalities are more likely associated with moderate to severe hypokalemia, a high degree of individual variability can occur with mild serum potassium level decreases. Concomitant factors such as magnesium depletion and digitalis therapy can precipitate cardiac dysrhythmias in patients with potassium deficiency.
Hypomagnesemia frequently coexists with and can exacerbate hypokalemia, particularly in conditions such as chronic diarrhea, alcohol use disorder, genetic disorders, diuretic use, and chemotherapy.[18] Both electrolyte deficiencies promote the development of cardiac dysrhythmias. The combination of hypokalemia and hypomagnesemia is associated with an increased risk of torsades de pointe, particularly in individuals on QT-prolonging medications (see Image. ECG in a Patient With Torsades de Pointes). Low serum magnesium levels can also increase urine potassium loss, exacerbating hypokalemia and hindering replacement efforts.
Imaging Studies
Imaging studies are not typically the first-line diagnostic modalities used for evaluating hypokalemia's etiology. However, these tests may help detect structural abnormalities causing hypokalemia or evaluate concomitant conditions. For example, renal ultrasound may be performed to assess for kidney abnormalities, such as renal cysts, tumors, or obstructive uropathy. Computed tomography and magnetic resonance imaging can help look for adrenal gland or central nervous system lesions, giving rise to aberrant hormonal causes of hypokalemia. A chest x-ray may reveal lung pathology contributing to alkalosis and potassium shifts, such as chronic obstructive pulmonary disease. Osteoporosis or osteomalacia may be assessed by a bone density scan in patients presenting with bone weakness and nephrolithiasis.
The choice of imaging modality should be based on clinical findings and the need for further evaluation of specific organ systems. The imaging findings should be interpreted in conjunction with clinical history and other diagnostic tests to guide management.
Treatment / Management
The therapeutic goals for hypokalemia are to prevent or treat life-threatening complications, correct the potassium deficit, and address the underlying cause. Therapeutic urgency depends on the severity of hypokalemia, the existence of comorbid conditions, and the rate of decline of serum potassium levels. Potassium replacement is indicated in most hypokalemia cases, especially when renal or gastrointestinal losses are significant. Concomitant hypomagnesemia should also be corrected if present. Potassium replacement alone may be ineffective if hypokalemia occurs concurrently with hypomagnesemia.
Mild-to-moderate hypokalemia may be asymptomatic. Thus, repletion is often not urgent in such cases. Mild-to-moderate hypokalemia is typically treated with oral potassium supplements. Providing 60 to 80 mmol/day over days to weeks is usually sufficient. Oral supplementation can irritate the gastrointestinal mucosa and cause bleeding or ulceration. However, oral potassium replacement is associated with a lower risk of rebound hyperkalemia. Potassium chloride is the preferred formulation for replacement therapy in most cases. Increasing dietary potassium is usually inadequate to treat hypokalemia because most of the potassium in foods is coupled with phosphate. Most cases of hypokalemia involve chloride depletion and respond best to potassium chloride replacement. Intravenous (IV) repletion is administered if oral therapy is not tolerated.
Replacement therapy must be provided rapidly when severe hypokalemia or clinical symptoms are present. Potassium chloride of 40 mmol given every 3 to 4 hours for 3 doses is preferred. Rapid correction is via oral intake, IV administration, or both. IV administration is preferred in the presence of cardiac dysrhythmias, digitalis toxicity, and recent or ongoing cardiac ischemia. Pain and phlebitis usually occur with peripheral IV infusions when infusion rates exceed 10 mmol/h. A risk of rebound hyperkalemia exists when rates exceed 20 mmol/h. Generally, 20 mmol/h of potassium chloride will increase serum potassium levels by an average of 0.25 mmol/h. Potassium should not be repleted using dextrose-containing solutions because dextrose stimulates insulin secretion, exacerbating hypokalemia.[19] IV replacement should be done cautiously, as rapid potassium infusion can cause cardiac arrest.
Serum potassium levels should be checked every 2 to 4 hours. Potassium replacement can occur more slowly once the serum potassium level is persistently above 3 mmol/L or clinical symptoms have resolved. Regardless of severity, careful serum potassium level monitoring is required, as hyperkalemia commonly develops in hospitalized patients.
The potassium deficit varies directly with the severity of hypokalemia. Every 0.3 mmol/L incremental decrease accounts for a reduction of approximately 100 mmol in total body potassium stores. For example, a patient with a serum potassium concentration of 2.0 mEq/L may have a 400 to 800 mEq potassium deficit.[20] Accurate quantification is difficult, especially when transcellular shifts cause hypokalemia. Therefore, careful monitoring is required to prevent hyperkalemia from excessive supplementation.
In cases of renal or gastrointestinal losses, potassium replacement is aimed at promptly elevating serum potassium levels to a safe range, followed by gradual replenishment of the remaining deficit over days to weeks. A potassium-sparing diuretic should also be considered when hypokalemia involves iatrogenic renal potassium wasting, as potassium replacement therapy alone may not suffice.
Establishing the presence of an acid-base disorder is crucial, as management varies for different causes of redistributive hypokalemia, where potassium shifts transcellularly with pH changes. Potassium repletion should be considered when paralysis or cardiac dysrhythmias are present. Rebound hyperkalemia is a potential complication of potassium therapy when cellular redistribution is the cause of hypokalemia.[21] As the underlying redistribution resolves, potassium may shift from cells to extracellular fluid, potentially leading to hyperkalemia during repletion. Patients with hypokalemic periodic paralysis face a significant risk of rebound hyperkalemia with potassium repletion. Monitoring serum potassium levels is essential during replacement therapy due to the heightened risk of hyperkalemia, irrespective of the cause.
Differential Diagnosis
The differential diagnosis of hypokalemia is vast. Notable ones include the following:
- Endocrine disorders: hyperthyroidism, thyrotoxicosis, Conn syndrome
- Iatrogenic cause: chronic corticosteroid intake, insulin therapy
- Congenital conditions: Bartter syndrome, hypokalemic periodic paralysis
- Renal disorders: RTA, osmotic diuresis
- Gastrointestinal losses: laxative abuse, vomiting
- Poor intake: eating disorders, restrictive diets
- Skin losses: excessive sweating
- Redistribution: alkalosis from chronic obstructive pulmonary disease, nonanion-gap acidosis from diabetic ketoacidosis
- Concomitant electrolyte imbalances: hypocalcemia, hypomagnesemia
A thorough clinical evaluation and diagnostic workup can differentiate these conditions.
Staging
The stages of hypokalemia are as follows:
- Mild hypokalemia: serum potassium level between 3.0 and 3.5 mEq/L
- Moderate hypokalemia: serum potassium level between 2.5 and 3.0 mEq/L
- Severe hypokalemia: serum potassium level below 2.5 mEq/L
Symptoms and complications vary depending on the condition's severity.
Prognosis
Mild hypokalemia can be easily treated with oral potassium supplements and dietary changes. In contrast, severe hypokalemia can lead to life-threatening complications such as cardiac arrhythmias, respiratory failure, and renal dysfunction if left untreated or undiagnosed. The presence of medical conditions, such as hypertension, diabetes, and chronic kidney disease, also influences the prognosis. Patients with these comorbidities are at a higher risk of developing hypokalemia-related complications.
Complications
Hypokalemia by itself can cause the following complications:
- Cardiac arrhythmias
- Cardiac failure
- Cardiac ischemia
- Left ventricular hypertrophy
- Muscular weakness
- Intestinal paralysis
- Respiratory compromise [22][23]
Other complications may arise if potassium deficiency is only part of another systemic disorder, such as Cushing or thyroid disease.
Consultations
Internists, hospitalists, and advanced clinicians usually manage hypokalemia. However, depending on the case, further consultations may be needed.
- Nephrologists assess and manage the underlying kidney disease causing potassium wasting and provide guidance on the appropriate treatment.
- Endocrinologists rule out or manage underlying endocrinological disorders.
- Dietitians provide dietary recommendations and ensure patients consume appropriate amounts of potassium through their diet or supplements.
An interprofessional approach to hypokalemia management should be initiated early to ensure timely and appropriate management and improve outcomes.
Deterrence and Patient Education
Preventive measures for hypokalemia depend on the underlying cause and individual risk factors. Strategies that can help prevent this condition include the following:
- Increasing oral potassium intake
- Staying hydrated
- Limiting alcohol and caffeine
- Managing medications
- Monitoring electrolyte levels
- Treating underlying conditions
Individuals with congenital conditions causing hypokalemia may be offered genetic counseling and family planning. Patients should also be aware of the signs and symptoms of potassium deficiency and instructed to contact their clinician immediately if they experience any manifestations of hypokalemia.
Pearls and Other Issues
The key points to remember in evaluating and managing hypokalemia are as follows:
- The serum potassium level determines hypokalemia severity. Mild hypokalemia is between 3.0 and 3.5 mEq/L, moderate hypokalemia is between 2.5 and 3.0 mEq/L, and severe hypokalemia is less than 2.5 mEq/L.
- Hypokalemia has a wide array of causes, both congenital and acquired.
- Hypokalemia's primary symptoms involve excitable tissues in the muscles, heart, intestines, and nervous system.
- Hypokalemia can be associated with other electrolyte imbalances, such as hypomagnesemia and hypocalcemia, which can exacerbate its symptoms and complications.
- The clinical presentation must guide the diagnostic workup. Serum and urine electrolytes and arterial blood gases are good initial tests, as they can help narrow down the differentials early on.
- Management is by careful potassium replacement. Mild-to-moderate hypokalemia may improve with oral supplementation and diet modification. Severe hypokalemia may require simultaneous oral and IV supplementation. However, care must be taken during potassium correction as rapid infusion may cause cardiac arrest.
- Patients may need long-term follow-up, depending on the etiology.
Preventive measures depend on the cause of potassium deficiency. Patients must be counseled about the signs and symptoms of hypokalemia and the importance of seeking prompt treatment.
Enhancing Healthcare Team Outcomes
An interprofessional team approach to managing hypokalemia ensures comprehensive patient care and better outcomes. Primary care physicians, internists, and pediatricians are central in diagnosing and managing hypokalemia, conducting initial evaluations, and coordinating care with specialists as needed. Nephrologists may be involved in caring for individuals with renal hypokalemia etiologies, providing expertise in kidney function assessment and management. Endocrinologists may be consulted for cases involving hormonal imbalances affecting potassium regulation.
Nurses are crucial in monitoring patients' vital signs, electrolyte levels, and symptoms, administering medications, and educating patients about dietary modifications and preventive measures. Pharmacists play an essential role in medication management, reviewing drug regimens for adverse effects contributing to hypokalemia and providing recommendations for alternative medications. Dietitians provide nutritional guidance, helping patients incorporate potassium-rich foods into their diet to prevent hypokalemia recurrence. By collaborating across disciplines, the interprofessional team ensures comprehensive and patient-centered care for individuals with hypokalemia, addressing immediate treatment needs and long-term management strategies.[24][25]