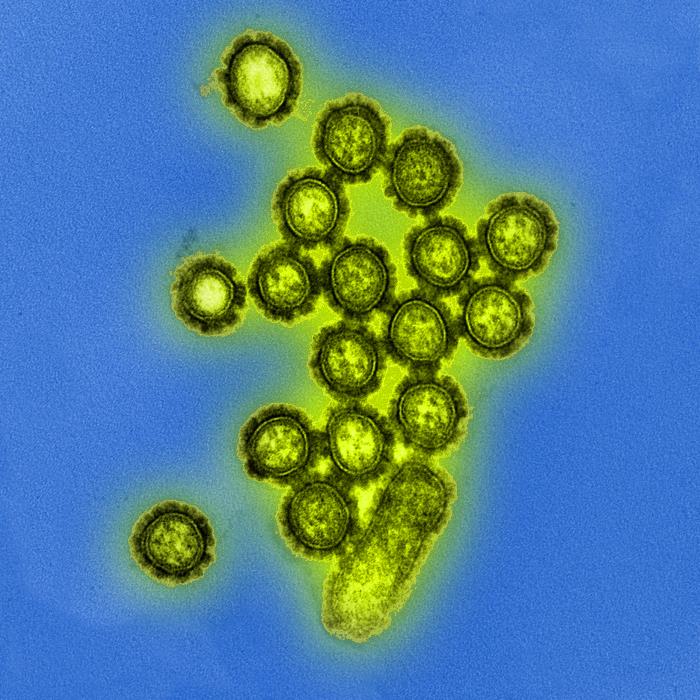
[1]
Hackett S, Hill L, Patel J, Ratnaraja N, Ifeyinwa A, Farooqi M, Nusgen U, Debenham P, Gandhi D, Makwana N, Smit E, Welch S. Clinical characteristics of paediatric H1N1 admissions in Birmingham, UK. Lancet (London, England). 2009 Aug 22:374(9690):605. doi: 10.1016/S0140-6736(09)61511-7. Epub
[PubMed PMID: 19700001]
Level 2 (mid-level) evidence
[2]
Cao B, Li XW, Mao Y, Wang J, Lu HZ, Chen YS, Liang ZA, Liang L, Zhang SJ, Zhang B, Gu L, Lu LH, Wang DY, Wang C, National Influenza A Pandemic (H1N1) 2009 Clinical Investigation Group of China. Clinical features of the initial cases of 2009 pandemic influenza A (H1N1) virus infection in China. The New England journal of medicine. 2009 Dec 24:361(26):2507-17. doi: 10.1056/NEJMoa0906612. Epub 2009 Dec 9
[PubMed PMID: 20007555]
Level 3 (low-level) evidence
[3]
Kshatriya RM, Khara NV, Ganjiwale J, Lote SD, Patel SN, Paliwal RP. Lessons learnt from the Indian H1N1 (swine flu) epidemic: Predictors of outcome based on epidemiological and clinical profile. Journal of family medicine and primary care. 2018 Nov-Dec:7(6):1506-1509. doi: 10.4103/jfmpc.jfmpc_38_18. Epub
[PubMed PMID: 30613550]
Level 2 (mid-level) evidence
[4]
Mena I, Nelson MI, Quezada-Monroy F, Dutta J, Cortes-Fernández R, Lara-Puente JH, Castro-Peralta F, Cunha LF, Trovão NS, Lozano-Dubernard B, Rambaut A, van Bakel H, García-Sastre A. Origins of the 2009 H1N1 influenza pandemic in swine in Mexico. eLife. 2016 Jun 28:5():. pii: e16777. doi: 10.7554/eLife.16777. Epub 2016 Jun 28
[PubMed PMID: 27350259]
[5]
Lewis NS, Russell CA, Langat P, Anderson TK, Berger K, Bielejec F, Burke DF, Dudas G, Fonville JM, Fouchier RA, Kellam P, Koel BF, Lemey P, Nguyen T, Nuansrichy B, Peiris JM, Saito T, Simon G, Skepner E, Takemae N, ESNIP3 consortium, Webby RJ, Van Reeth K, Brookes SM, Larsen L, Watson SJ, Brown IH, Vincent AL. The global antigenic diversity of swine influenza A viruses. eLife. 2016 Apr 15:5():e12217. doi: 10.7554/eLife.12217. Epub 2016 Apr 15
[PubMed PMID: 27113719]
[6]
Mancera Gracia JC, Van den Hoecke S, Saelens X, Van Reeth K. Effect of serial pig passages on the adaptation of an avian H9N2 influenza virus to swine. PloS one. 2017:12(4):e0175267. doi: 10.1371/journal.pone.0175267. Epub 2017 Apr 6
[PubMed PMID: 28384328]
[7]
Johnson NP, Mueller J. Updating the accounts: global mortality of the 1918-1920 "Spanish" influenza pandemic. Bulletin of the history of medicine. 2002 Spring:76(1):105-15
[PubMed PMID: 11875246]
[8]
Doshi P. The elusive definition of pandemic influenza. Bulletin of the World Health Organization. 2011 Jul 1:89(7):532-8. doi: 10.2471/BLT.11.086173. Epub
[PubMed PMID: 21734768]
[9]
Lei H, Li Y, Xiao S, Lin CH, Norris SL, Wei D, Hu Z, Ji S. Routes of transmission of influenza A H1N1, SARS CoV, and norovirus in air cabin: Comparative analyses. Indoor air. 2018 May:28(3):394-403. doi: 10.1111/ina.12445. Epub 2018 Jan 6
[PubMed PMID: 29244221]
Level 2 (mid-level) evidence
[10]
Keenliside J. Pandemic influenza A H1N1 in Swine and other animals. Current topics in microbiology and immunology. 2013:370():259-71. doi: 10.1007/82_2012_301. Epub
[PubMed PMID: 23254339]
Level 3 (low-level) evidence
[11]
Lange E, Kalthoff D, Blohm U, Teifke JP, Breithaupt A, Maresch C, Starick E, Fereidouni S, Hoffmann B, Mettenleiter TC, Beer M, Vahlenkamp TW. Pathogenesis and transmission of the novel swine-origin influenza virus A/H1N1 after experimental infection of pigs. The Journal of general virology. 2009 Sep:90(Pt 9):2119-23. doi: 10.1099/vir.0.014480-0. Epub 2009 Jul 10
[PubMed PMID: 19592456]
[12]
Rassy D, Smith RD. The economic impact of H1N1 on Mexico's tourist and pork sectors. Health economics. 2013 Jul:22(7):824-34. doi: 10.1002/hec.2862. Epub
[PubMed PMID: 23744805]
[13]
Nogales A, Martinez-Sobrido L, Chiem K, Topham DJ, DeDiego ML. Functional Evolution of the 2009 Pandemic H1N1 Influenza Virus NS1 and PA in Humans. Journal of virology. 2018 Oct 1:92(19):. doi: 10.1128/JVI.01206-18. Epub 2018 Sep 12
[PubMed PMID: 30021892]
[14]
Tapia R, García V, Mena J, Bucarey S, Medina RA, Neira V. Infection of novel reassortant H1N2 and H3N2 swine influenza A viruses in the guinea pig model. Veterinary research. 2018 Jul 27:49(1):73. doi: 10.1186/s13567-018-0572-4. Epub 2018 Jul 27
[PubMed PMID: 30053826]
[15]
Fouchier RA, Munster V, Wallensten A, Bestebroer TM, Herfst S, Smith D, Rimmelzwaan GF, Olsen B, Osterhaus AD. Characterization of a novel influenza A virus hemagglutinin subtype (H16) obtained from black-headed gulls. Journal of virology. 2005 Mar:79(5):2814-22
[PubMed PMID: 15709000]
[16]
Hirst GK. THE AGGLUTINATION OF RED CELLS BY ALLANTOIC FLUID OF CHICK EMBRYOS INFECTED WITH INFLUENZA VIRUS. Science (New York, N.Y.). 1941 Jul 4:94(2427):22-3
[PubMed PMID: 17777315]
[17]
Zebedee SL, Lamb RA. Influenza A virus M2 protein: monoclonal antibody restriction of virus growth and detection of M2 in virions. Journal of virology. 1988 Aug:62(8):2762-72
[PubMed PMID: 2455818]
[18]
Herrler G, Rott R, Klenk HD, Müller HP, Shukla AK, Schauer R. The receptor-destroying enzyme of influenza C virus is neuraminate-O-acetylesterase. The EMBO journal. 1985 Jun:4(6):1503-6
[PubMed PMID: 2411539]
[19]
Shope RE. SWINE INFLUENZA : III. FILTRATION EXPERIMENTS AND ETIOLOGY. The Journal of experimental medicine. 1931 Jul 31:54(3):373-85
[PubMed PMID: 19869924]
[20]
Kaplan MM. Relationships between animal and human influenza. Bulletin of the World Health Organization. 1969:41(3):485-6
[PubMed PMID: 5309460]
Level 3 (low-level) evidence
[22]
Dawood FS, Iuliano AD, Reed C, Meltzer MI, Shay DK, Cheng PY, Bandaranayake D, Breiman RF, Brooks WA, Buchy P, Feikin DR, Fowler KB, Gordon A, Hien NT, Horby P, Huang QS, Katz MA, Krishnan A, Lal R, Montgomery JM, Mølbak K, Pebody R, Presanis AM, Razuri H, Steens A, Tinoco YO, Wallinga J, Yu H, Vong S, Bresee J, Widdowson MA. Estimated global mortality associated with the first 12 months of 2009 pandemic influenza A H1N1 virus circulation: a modelling study. The Lancet. Infectious diseases. 2012 Sep:12(9):687-95. doi: 10.1016/S1473-3099(12)70121-4. Epub 2012 Jun 26
[PubMed PMID: 22738893]
[23]
Van Kerkhove MD, Hirve S, Koukounari A, Mounts AW, H1N1pdm serology working group. Estimating age-specific cumulative incidence for the 2009 influenza pandemic: a meta-analysis of A(H1N1)pdm09 serological studies from 19 countries. Influenza and other respiratory viruses. 2013 Sep:7(5):872-86. doi: 10.1111/irv.12074. Epub 2013 Jan 21
[PubMed PMID: 23331969]
Level 1 (high-level) evidence
[24]
Nickol ME, Kindrachuk J. A year of terror and a century of reflection: perspectives on the great influenza pandemic of 1918-1919. BMC infectious diseases. 2019 Feb 6:19(1):117. doi: 10.1186/s12879-019-3750-8. Epub 2019 Feb 6
[PubMed PMID: 30727970]
Level 3 (low-level) evidence
[25]
Cohen J. Swine flu pandemic. What's old is new: 1918 virus matches 2009 H1N1 strain. Science (New York, N.Y.). 2010 Mar 26:327(5973):1563-4. doi: 10.1126/science.327.5973.1563. Epub
[PubMed PMID: 20339037]
[26]
Scholtissek C, Rohde W, Von Hoyningen V, Rott R. On the origin of the human influenza virus subtypes H2N2 and H3N2. Virology. 1978 Jun 1:87(1):13-20
[PubMed PMID: 664248]
[27]
Li X, Guo L, Liu C, Cheng Y, Kong M, Yang L, Zhuang Z, Liu J, Zou M, Dong X, Su X, Gu Q. Human infection with a novel reassortant Eurasian-avian lineage swine H1N1 virus in northern China. Emerging microbes & infections. 2019:8(1):1535-1545. doi: 10.1080/22221751.2019.1679611. Epub
[PubMed PMID: 31661383]
[28]
Chastagner A, Enouf V, Peroz D, Hervé S, Lucas P, Quéguiner S, Gorin S, Beven V, Behillil S, Leneveu P, Garin E, Blanchard Y, van der Werf S, Simon G. Bidirectional Human-Swine Transmission of Seasonal Influenza A(H1N1)pdm09 Virus in Pig Herd, France, 2018. Emerging infectious diseases. 2019 Oct:25(10):1940-1943. doi: 10.3201/eid2510.190068. Epub
[PubMed PMID: 31538914]
[29]
Scholtissek C, Bürger H, Kistner O, Shortridge KF. The nucleoprotein as a possible major factor in determining host specificity of influenza H3N2 viruses. Virology. 1985 Dec:147(2):287-94
[PubMed PMID: 2416114]
[30]
Sun H, Xiao Y, Liu J, Wang D, Li F, Wang C, Li C, Zhu J, Song J, Sun H, Jiang Z, Liu L, Zhang X, Wei K, Hou D, Pu J, Sun Y, Tong Q, Bi Y, Chang KC, Liu S, Gao GF, Liu J. Prevalent Eurasian avian-like H1N1 swine influenza virus with 2009 pandemic viral genes facilitating human infection. Proceedings of the National Academy of Sciences of the United States of America. 2020 Jul 21:117(29):17204-17210. doi: 10.1073/pnas.1921186117. Epub 2020 Jun 29
[PubMed PMID: 32601207]
[31]
Jain S, Kamimoto L, Bramley AM, Schmitz AM, Benoit SR, Louie J, Sugerman DE, Druckenmiller JK, Ritger KA, Chugh R, Jasuja S, Deutscher M, Chen S, Walker JD, Duchin JS, Lett S, Soliva S, Wells EV, Swerdlow D, Uyeki TM, Fiore AE, Olsen SJ, Fry AM, Bridges CB, Finelli L, 2009 Pandemic Influenza A (H1N1) Virus Hospitalizations Investigation Team. Hospitalized patients with 2009 H1N1 influenza in the United States, April-June 2009. The New England journal of medicine. 2009 Nov 12:361(20):1935-44. doi: 10.1056/NEJMoa0906695. Epub 2009 Oct 8
[PubMed PMID: 19815859]
[32]
Taubenberger JK, Morens DM. The pathology of influenza virus infections. Annual review of pathology. 2008:3():499-522
[PubMed PMID: 18039138]
[33]
Roberts KL, Shelton H, Stilwell P, Barclay WS. Transmission of a 2009 H1N1 pandemic influenza virus occurs before fever is detected, in the ferret model. PloS one. 2012:7(8):e43303. doi: 10.1371/journal.pone.0043303. Epub 2012 Aug 29
[PubMed PMID: 22952661]
[34]
De Serres G, Rouleau I, Hamelin ME, Quach C, Skowronski D, Flamand L, Boulianne N, Li Y, Carbonneau J, Bourgault A, Couillard M, Charest H, Boivin G. Contagious period for pandemic (H1N1) 2009. Emerging infectious diseases. 2010 May:16(5):783-8. doi: 10.3201/eid1605.091894. Epub
[PubMed PMID: 20409367]
[35]
Centers for Disease Control and Prevention (CDC). Oseltamivir-resistant novel influenza A (H1N1) virus infection in two immunosuppressed patients - Seattle, Washington, 2009. MMWR. Morbidity and mortality weekly report. 2009 Aug 21:58(32):893-6
[PubMed PMID: 19696719]
[36]
Esposito S, Daleno C, Baldanti F, Scala A, Campanini G, Taroni F, Fossali E, Pelucchi C, Principi N. Viral shedding in children infected by pandemic A/H1N1/2009 influenza virus. Virology journal. 2011 Jul 13:8():349. doi: 10.1186/1743-422X-8-349. Epub 2011 Jul 13
[PubMed PMID: 21752272]
[37]
Kumar A, Zarychanski R, Pinto R, Cook DJ, Marshall J, Lacroix J, Stelfox T, Bagshaw S, Choong K, Lamontagne F, Turgeon AF, Lapinsky S, Ahern SP, Smith O, Siddiqui F, Jouvet P, Khwaja K, McIntyre L, Menon K, Hutchison J, Hornstein D, Joffe A, Lauzier F, Singh J, Karachi T, Wiebe K, Olafson K, Ramsey C, Sharma S, Dodek P, Meade M, Hall R, Fowler RA, Canadian Critical Care Trials Group H1N1 Collaborative. Critically ill patients with 2009 influenza A(H1N1) infection in Canada. JAMA. 2009 Nov 4:302(17):1872-9. doi: 10.1001/jama.2009.1496. Epub 2009 Oct 12
[PubMed PMID: 19822627]
[38]
Killip MJ, Fodor E, Randall RE. Influenza virus activation of the interferon system. Virus research. 2015 Nov 2:209():11-22. doi: 10.1016/j.virusres.2015.02.003. Epub 2015 Feb 9
[PubMed PMID: 25678267]
[39]
ANZIC Influenza Investigators, Webb SA, Pettilä V, Seppelt I, Bellomo R, Bailey M, Cooper DJ, Cretikos M, Davies AR, Finfer S, Harrigan PW, Hart GK, Howe B, Iredell JR, McArthur C, Mitchell I, Morrison S, Nichol AD, Paterson DL, Peake S, Richards B, Stephens D, Turner A, Yung M. Critical care services and 2009 H1N1 influenza in Australia and New Zealand. The New England journal of medicine. 2009 Nov 12:361(20):1925-34. doi: 10.1056/NEJMoa0908481. Epub 2009 Oct 8
[PubMed PMID: 19815860]
[40]
Taubenberger JK, Layne SP. Diagnosis of influenza virus: coming to grips with the molecular era. Molecular diagnosis : a journal devoted to the understanding of human disease through the clinical application of molecular biology. 2001 Dec:6(4):291-305
[PubMed PMID: 11774194]
Level 3 (low-level) evidence
[41]
Nin N, Sánchez-Rodríguez C, Ver LS, Cardinal P, Ferruelo A, Soto L, Deicas A, Campos N, Rocha O, Ceraso DH, El-Assar M, Ortín J, Fernández-Segoviano P, Esteban A, Lorente JA. Lung histopathological findings in fatal pandemic influenza A (H1N1). Medicina intensiva. 2012 Jan-Feb:36(1):24-31. doi: 10.1016/j.medin.2011.10.005. Epub 2011 Dec 10
[PubMed PMID: 22154847]
[42]
Morris DE, Cleary DW, Clarke SC. Secondary Bacterial Infections Associated with Influenza Pandemics. Frontiers in microbiology. 2017:8():1041. doi: 10.3389/fmicb.2017.01041. Epub 2017 Jun 23
[PubMed PMID: 28690590]
[43]
Shiley KT, Nadolski G, Mickus T, Fishman NO, Lautenbach E. Differences in the epidemiological characteristics and clinical outcomes of pandemic (H1N1) 2009 influenza, compared with seasonal influenza. Infection control and hospital epidemiology. 2010 Jul:31(7):676-82. doi: 10.1086/653204. Epub
[PubMed PMID: 20500086]
Level 2 (mid-level) evidence
[44]
Carcione D, Giele C, Dowse GK, Mak DB, Goggin L, Kwan K, Williams S, Smith D, Effler P. Comparison of pandemic (H1N1) 2009 and seasonal influenza, Western Australia, 2009. Emerging infectious diseases. 2010 Sep:16(9):1388-95. doi: 10.3201/eid1609.100076. Epub
[PubMed PMID: 20735922]
[45]
Lee N, Chan PK, Lui GC, Wong BC, Sin WW, Choi KW, Wong RY, Lee EL, Yeung AC, Ngai KL, Chan MC, Lai RW, Yu AW, Hui DS. Complications and outcomes of pandemic 2009 Influenza A (H1N1) virus infection in hospitalized adults: how do they differ from those in seasonal influenza? The Journal of infectious diseases. 2011 Jun 15:203(12):1739-47. doi: 10.1093/infdis/jir187. Epub
[PubMed PMID: 21606532]
[46]
Lemaitre M, Carrat F. Comparative age distribution of influenza morbidity and mortality during seasonal influenza epidemics and the 2009 H1N1 pandemic. BMC infectious diseases. 2010 Jun 9:10():162. doi: 10.1186/1471-2334-10-162. Epub 2010 Jun 9
[PubMed PMID: 20534113]
Level 2 (mid-level) evidence
[47]
Yu H, Feng Z, Uyeki TM, Liao Q, Zhou L, Feng L, Ye M, Xiang N, Huai Y, Yuan Y, Jiang H, Zheng Y, Gargiullo P, Peng Z, Feng Y, Zheng J, Xu C, Zhang Y, Shu Y, Gao Z, Yang W, Wang Y. Risk factors for severe illness with 2009 pandemic influenza A (H1N1) virus infection in China. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2011 Feb 15:52(4):457-65. doi: 10.1093/cid/ciq144. Epub 2011 Jan 10
[PubMed PMID: 21220768]
[48]
Jernigan DB, Lindstrom SL, Johnson JR, Miller JD, Hoelscher M, Humes R, Shively R, Brammer L, Burke SA, Villanueva JM, Balish A, Uyeki T, Mustaquim D, Bishop A, Handsfield JH, Astles R, Xu X, Klimov AI, Cox NJ, Shaw MW. Detecting 2009 pandemic influenza A (H1N1) virus infection: availability of diagnostic testing led to rapid pandemic response. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2011 Jan 1:52 Suppl 1():S36-43. doi: 10.1093/cid/ciq020. Epub
[PubMed PMID: 21342897]
[49]
Wood JM, Gaines-Das RE, Taylor J, Chakraverty P. Comparison of influenza serological techniques by international collaborative study. Vaccine. 1994 Feb:12(2):167-74
[PubMed PMID: 8147099]
[50]
Vasoo S, Stevens J, Singh K. Rapid antigen tests for diagnosis of pandemic (Swine) influenza A/H1N1. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2009 Oct 1:49(7):1090-3. doi: 10.1086/644743. Epub
[PubMed PMID: 19725784]
[51]
Salvesen HA, Whitelaw CBA. Current and prospective control strategies of influenza A virus in swine. Porcine health management. 2021 Feb 28:7(1):23. doi: 10.1186/s40813-021-00196-0. Epub 2021 Feb 28
[PubMed PMID: 33648602]
[52]
Mengual-Chuliá B, Alonso-Cordero A, Cano L, Mosquera MDM, de Molina P, Vendrell R, Reyes-Prieto M, Jané M, Torner N, Martínez AI, Vila J, Díez-Domingo J, Marcos MÁ, López-Labrador FX. Whole-Genome Analysis Surveillance of Influenza A Virus Resistance to Polymerase Complex Inhibitors in Eastern Spain from 2016 to 2019. Antimicrobial agents and chemotherapy. 2021 May 18:65(6):. doi: 10.1128/AAC.02718-20. Epub 2021 May 18
[PubMed PMID: 33782005]
[53]
Jeong EK, Bae JE, Kim IS. Inactivation of influenza A virus H1N1 by disinfection process. American journal of infection control. 2010 Jun:38(5):354-60. doi: 10.1016/j.ajic.2010.03.003. Epub 2010 Apr 28
[PubMed PMID: 20430477]
[54]
Yamaya M, Nishimura H, Lusamba Kalonji N, Deng X, Momma H, Shimotai Y, Nagatomi R. Effects of high temperature on pandemic and seasonal human influenza viral replication and infection-induced damage in primary human tracheal epithelial cell cultures. Heliyon. 2019 Feb:5(2):e01149. doi: 10.1016/j.heliyon.2019.e01149. Epub 2019 Feb 5
[PubMed PMID: 30839917]
[55]
Lee JH, Gramer MR, Joo HS. Efficacy of swine influenza A virus vaccines against an H3N2 virus variant. Canadian journal of veterinary research = Revue canadienne de recherche veterinaire. 2007 Jul:71(3):207-12
[PubMed PMID: 17695596]
[56]
Fablet C, Simon G, Dorenlor V, Eono F, Eveno E, Gorin S, Quéguiner S, Madec F, Rose N. Different herd level factors associated with H1N1 or H1N2 influenza virus infections in fattening pigs. Preventive veterinary medicine. 2013 Nov 1:112(3-4):257-65. doi: 10.1016/j.prevetmed.2013.07.006. Epub 2013 Aug 19
[PubMed PMID: 23968780]
[57]
Ma W, Kahn RE, Richt JA. The pig as a mixing vessel for influenza viruses: Human and veterinary implications. Journal of molecular and genetic medicine : an international journal of biomedical research. 2008 Nov 27:3(1):158-66
[PubMed PMID: 19565018]
[58]
Borkenhagen LK, Wang GL, Simmons RA, Bi ZQ, Lu B, Wang XJ, Wang CX, Chen SH, Song SX, Li M, Zhao T, Wu MN, Park LP, Cao WC, Ma MJ, Gray GC. High Risk of Influenza Virus Infection Among Swine Workers: Examining a Dynamic Cohort in China. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2020 Jul 27:71(3):622-629. doi: 10.1093/cid/ciz865. Epub
[PubMed PMID: 31504322]
[59]
Zhang L, Peng Z, Ou J, Zeng G, Fontaine RE, Liu M, Cui F, Hong R, Zhou H, Huai Y, Chuang SK, Leung YH, Feng Y, Luo Y, Shen T, Zhu BP, Widdowson MA, Yu H. Protection by face masks against influenza A(H1N1)pdm09 virus on trans-Pacific passenger aircraft, 2009. Emerging infectious diseases. 2013:19(9):1403-10. doi: 10.3201/eid1909.121765. Epub
[PubMed PMID: 23968983]
[60]
Zhang N, Li Y. Transmission of Influenza A in a Student Office Based on Realistic Person-to-Person Contact and Surface Touch Behaviour. International journal of environmental research and public health. 2018 Aug 9:15(8):. doi: 10.3390/ijerph15081699. Epub 2018 Aug 9
[PubMed PMID: 30096894]
[61]
Rewar S, Mirdha D, Rewar P. Treatment and Prevention of Pandemic H1N1 Influenza. Annals of global health. 2015 Sep-Oct:81(5):645-53. doi: 10.1016/j.aogh.2015.08.014. Epub
[PubMed PMID: 27036721]
[62]
Borse RH, Shrestha SS, Fiore AE, Atkins CY, Singleton JA, Furlow C, Meltzer MI. Effects of vaccine program against pandemic influenza A(H1N1) virus, United States, 2009-2010. Emerging infectious diseases. 2013 Mar:19(3):439-48. doi: 10.3201/eid1903.120394. Epub
[PubMed PMID: 23622679]
[63]
Centers for Disease Control and Prevention (CDC). Interim results: state-specific influenza A (H1N1) 2009 monovalent vaccination coverage - United States, October 2009-January 2010. MMWR. Morbidity and mortality weekly report. 2010 Apr 2:59(12):363-8
[PubMed PMID: 20360670]
[64]
Sukhal S, Sethi J, Ganesh M, Villablanca PA, Malhotra AK, Ramakrishna H. Extracorporeal membrane oxygenation in severe influenza infection with respiratory failure: A systematic review and meta-analysis. Annals of cardiac anaesthesia. 2017 Jan-Mar:20(1):14-21. doi: 10.4103/0971-9784.197820. Epub
[PubMed PMID: 28074789]
Level 1 (high-level) evidence
[65]
Treanor JJ, Hayden FG, Vrooman PS, Barbarash R, Bettis R, Riff D, Singh S, Kinnersley N, Ward P, Mills RG. Efficacy and safety of the oral neuraminidase inhibitor oseltamivir in treating acute influenza: a randomized controlled trial. US Oral Neuraminidase Study Group. JAMA. 2000 Feb 23:283(8):1016-24
[PubMed PMID: 10697061]
Level 1 (high-level) evidence
[67]
Kohno S, Kida H, Mizuguchi M, Shimada J, S-021812 Clinical Study Group. Efficacy and safety of intravenous peramivir for treatment of seasonal influenza virus infection. Antimicrobial agents and chemotherapy. 2010 Nov:54(11):4568-74. doi: 10.1128/AAC.00474-10. Epub 2010 Aug 16
[PubMed PMID: 20713668]
[68]
Sharma Y, Horwood C, Hakendorf P, Thompson C. Effectiveness of Oseltamivir in reducing 30-day readmissions and mortality among patients with severe seasonal influenza in Australian hospitalized patients. International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases. 2021 Mar:104():232-238. doi: 10.1016/j.ijid.2021.01.011. Epub 2021 Jan 9
[PubMed PMID: 33434667]
[69]
Al-Husban N, Obeidat N, Al-Kuran O, Al Oweidat K, Bakri F. H1N1 Infection in Pregnancy; A Retrospective Study of Feto-Maternal Outcome and Impact of the Timing of Antiviral Therapy. Mediterranean journal of hematology and infectious diseases. 2019:11(1):e2019020. doi: 10.4084/MJHID.2019.020. Epub 2019 Mar 1
[PubMed PMID: 30858958]
Level 2 (mid-level) evidence
[70]
Nordstrom BL, Oh K, Sacks ST, L'Italien GJ. Skin reactions in patients with influenza treated with oseltamivir: a retrospective cohort study. Antiviral therapy. 2004 Apr:9(2):187-95
[PubMed PMID: 15134180]
Level 2 (mid-level) evidence
[71]
Smith EV, Pynn MC, Blackford S, Leopold DJ. Stevens-Johnson syndrome secondary to oseltamivir (Tamiflu). The British journal of general practice : the journal of the Royal College of General Practitioners. 2010 Feb:60(571):133-4. doi: 10.3399/bjgp10X483292. Epub
[PubMed PMID: 20132714]
[72]
Fang S, Qi L, Zhou N, Li C. Case report on alimentary tract hemorrhage and liver injury after therapy with oseltamivir: A case report. Medicine. 2018 Sep:97(38):e12497. doi: 10.1097/MD.0000000000012497. Epub
[PubMed PMID: 30235756]
Level 2 (mid-level) evidence
[73]
Lee D, Jo H, Jang Y, Bae S, Agura T, Kang D, Kang M, Kim Y, Cho NH, Kim Y, Kang JS. Alloferon and Zanamivir Show Effective Antiviral Activity against Influenza A Virus (H1N1) Infection In Vitro and In Vivo. International journal of molecular sciences. 2022 Dec 30:24(1):. doi: 10.3390/ijms24010678. Epub 2022 Dec 30
[PubMed PMID: 36614125]
[75]
Eiland LS, Eiland EH. Zanamivir for the prevention of influenza in adults and children age 5 years and older. Therapeutics and clinical risk management. 2007 Jun:3(3):461-5
[PubMed PMID: 18488077]
[76]
Freund B, Gravenstein S, Elliott M, Miller I. Zanamivir: a review of clinical safety. Drug safety. 1999 Oct:21(4):267-81
[PubMed PMID: 10514019]
[77]
Monto AS, Robinson DP, Herlocher ML, Hinson JM Jr, Elliott MJ, Crisp A. Zanamivir in the prevention of influenza among healthy adults: a randomized controlled trial. JAMA. 1999 Jul 7:282(1):31-5
[PubMed PMID: 10404908]
Level 1 (high-level) evidence
[78]
Takemoto Y, Asai T, Ikezoe I, Yano T, Ichikawa M, Miyagawa S, Matsumoto J. Clinical effects of oseltamivir, zanamivir, laninamivir and peramivir on seasonal influenza infection in outpatients in Japan during the winter of 2012-2013. Chemotherapy. 2013:59(5):373-8. doi: 10.1159/000362436. Epub 2014 May 10
[PubMed PMID: 24821568]
[79]
Nakamura S, Miyazaki T, Izumikawa K, Kakeya H, Saisho Y, Yanagihara K, Miyazaki Y, Mukae H, Kohno S. Efficacy and Safety of Intravenous Peramivir Compared With Oseltamivir in High-Risk Patients Infected With Influenza A and B Viruses: A Multicenter Randomized Controlled Study. Open forum infectious diseases. 2017 Summer:4(3):ofx129. doi: 10.1093/ofid/ofx129. Epub 2017 Jun 19
[PubMed PMID: 28761899]
Level 1 (high-level) evidence
[80]
Komeda T, Ishii S, Itoh Y, Ariyasu Y, Sanekata M, Yoshikawa T, Shimada J. Post-marketing safety and effectiveness evaluation of the intravenous anti-influenza neuraminidase inhibitor peramivir. II: a pediatric drug use investigation. Journal of infection and chemotherapy : official journal of the Japan Society of Chemotherapy. 2015 Mar:21(3):194-201. doi: 10.1016/j.jiac.2014.11.009. Epub 2014 Nov 26
[PubMed PMID: 25523716]
[81]
Thorlund K, Awad T, Boivin G, Thabane L. Systematic review of influenza resistance to the neuraminidase inhibitors. BMC infectious diseases. 2011 May 19:11():134. doi: 10.1186/1471-2334-11-134. Epub 2011 May 19
[PubMed PMID: 21592407]
Level 1 (high-level) evidence
[82]
Somerville LK, Basile K, Dwyer DE, Kok J. The impact of influenza virus infection in pregnancy. Future microbiology. 2018 Feb:13():263-274. doi: 10.2217/fmb-2017-0096. Epub 2018 Jan 11
[PubMed PMID: 29320882]
[83]
Littauer EQ, Esser ES, Antao OQ, Vassilieva EV, Compans RW, Skountzou I. H1N1 influenza virus infection results in adverse pregnancy outcomes by disrupting tissue-specific hormonal regulation. PLoS pathogens. 2017 Nov:13(11):e1006757. doi: 10.1371/journal.ppat.1006757. Epub 2017 Nov 27
[PubMed PMID: 29176767]
[84]
Creanga AA, Johnson TF, Graitcer SB, Hartman LK, Al-Samarrai T, Schwarz AG, Chu SY, Sackoff JE, Jamieson DJ, Fine AD, Shapiro-Mendoza CK, Jones LE, Uyeki TM, Balter S, Bish CL, Finelli L, Honein MA. Severity of 2009 pandemic influenza A (H1N1) virus infection in pregnant women. Obstetrics and gynecology. 2010 Apr:115(4):717-726. doi: 10.1097/AOG.0b013e3181d57947. Epub
[PubMed PMID: 20308830]
[85]
Dunstan HJ, Mill AC, Stephens S, Yates LM, Thomas SH. Pregnancy outcome following maternal use of zanamivir or oseltamivir during the 2009 influenza A/H1N1 pandemic: a national prospective surveillance study. BJOG : an international journal of obstetrics and gynaecology. 2014 Jun:121(7):901-6. doi: 10.1111/1471-0528.12640. Epub 2014 Mar 7
[PubMed PMID: 24602087]
[86]
Van Kerkhove MD, Vandemaele KA, Shinde V, Jaramillo-Gutierrez G, Koukounari A, Donnelly CA, Carlino LO, Owen R, Paterson B, Pelletier L, Vachon J, Gonzalez C, Hongjie Y, Zijian F, Chuang SK, Au A, Buda S, Krause G, Haas W, Bonmarin I, Taniguichi K, Nakajima K, Shobayashi T, Takayama Y, Sunagawa T, Heraud JM, Orelle A, Palacios E, van der Sande MA, Wielders CC, Hunt D, Cutter J, Lee VJ, Thomas J, Santa-Olalla P, Sierra-Moros MJ, Hanshaoworakul W, Ungchusak K, Pebody R, Jain S, Mounts AW, WHO Working Group for Risk Factors for Severe H1N1pdm Infection. Risk factors for severe outcomes following 2009 influenza A (H1N1) infection: a global pooled analysis. PLoS medicine. 2011 Jul:8(7):e1001053. doi: 10.1371/journal.pmed.1001053. Epub 2011 Jul 5
[PubMed PMID: 21750667]
[87]
Nguyen-Van-Tam JS, Openshaw PJ, Hashim A, Gadd EM, Lim WS, Semple MG, Read RC, Taylor BL, Brett SJ, McMenamin J, Enstone JE, Armstrong C, Nicholson KG, Influenza Clinical Information Network (FLU-CIN). Risk factors for hospitalisation and poor outcome with pandemic A/H1N1 influenza: United Kingdom first wave (May-September 2009). Thorax. 2010 Jul:65(7):645-51. doi: 10.1136/thx.2010.135210. Epub
[PubMed PMID: 20627925]
[88]
Sertogullarindan B, Ozbay B, Gunini H, Sunnetcioglu A, Arisoy A, Bilgin HM, Mermit Cilingir B, Duran M, Yildiz H, Ekin S, Baran A. Clinical and prognostic features of patients with pandemic 2009 influenza A (H1N1) virus in the intensive care unit. African health sciences. 2011 Jun:11(2):163-70
[PubMed PMID: 21857845]
[89]
Delgado-Rodríguez M, Castilla J, Godoy P, Martín V, Soldevila N, Alonso J, Astray J, Baricot M, Cantón R, Castro A, Gónzález-Candelas F, Mayoral JM, Quintana JM, Pumarola T, Tamames S, Sáez M, Domínguez A, CIBERESP Cases and Controls in Pandemic Influenza Working Group. Prognosis of hospitalized patients with 2009 H1N1 influenza in Spain: influence of neuraminidase inhibitors. The Journal of antimicrobial chemotherapy. 2012 Jul:67(7):1739-45. doi: 10.1093/jac/dks098. Epub 2012 Mar 30
[PubMed PMID: 22467633]
Level 3 (low-level) evidence
[90]
Bellei NC, Cabeça TK, Carraro E, Goto JM, Cuba GT, Hidalgo SR, Burattini MN. Pandemic H1N1 illness prognosis: evidence from clinical and epidemiological data from the first pandemic wave in São Paulo, Brazil. Clinics (Sao Paulo, Brazil). 2013 Jun:68(6):840-5. doi: 10.6061/clinics/2013(06)19. Epub
[PubMed PMID: 23778481]
Level 2 (mid-level) evidence
[91]
Viasus D, Oteo Revuelta JA, Martínez-Montauti J, Carratalà J. Influenza A(H1N1)pdm09-related pneumonia and other complications. Enfermedades infecciosas y microbiologia clinica. 2012 Oct:30 Suppl 4():43-8. doi: 10.1016/S0213-005X(12)70104-0. Epub
[PubMed PMID: 23116792]
[92]
Davis LE. Neurologic and muscular complications of the 2009 influenza A (H1N1) pandemic. Current neurology and neuroscience reports. 2010 Nov:10(6):476-83. doi: 10.1007/s11910-010-0135-1. Epub
[PubMed PMID: 20697982]
[93]
Luyt CE, Combes A, Becquemin MH, Beigelman-Aubry C, Hatem S, Brun AL, Zraik N, Carrat F, Grenier PA, Richard JM, Mercat A, Brochard L, Brun-Buisson C, Chastre J, REVA Study Group. Long-term outcomes of pandemic 2009 influenza A(H1N1)-associated severe ARDS. Chest. 2012 Sep:142(3):583-592. doi: 10.1378/chest.11-2196. Epub
[PubMed PMID: 22948576]
[94]
Del Rio C, Hernandez-Avila M. Lessons from previous influenza pandemics and from the Mexican response to the current influenza pandemic. Archives of medical research. 2009 Nov:40(8):677-80. doi: 10.1016/j.arcmed.2009.12.005. Epub
[PubMed PMID: 20304256]
[95]
Bridges CB, Kuehnert MJ, Hall CB. Transmission of influenza: implications for control in health care settings. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2003 Oct 15:37(8):1094-101
[PubMed PMID: 14523774]
[96]
Fell DB, Sprague AE, Liu N, Yasseen AS 3rd, Wen SW, Smith G, Walker MC, Better Outcomes Registry & Network (BORN) Ontario. H1N1 influenza vaccination during pregnancy and fetal and neonatal outcomes. American journal of public health. 2012 Jun:102(6):e33-40. doi: 10.2105/AJPH.2011.300606. Epub 2012 Apr 19
[PubMed PMID: 22515877]
[97]
Jackson C, Vynnycky E, Hawker J, Olowokure B, Mangtani P. School closures and influenza: systematic review of epidemiological studies. BMJ open. 2013:3(2):. doi: 10.1136/bmjopen-2012-002149. Epub 2013 Feb 26
[PubMed PMID: 23447463]
Level 1 (high-level) evidence
[98]
Cauchemez S, Ferguson NM, Wachtel C, Tegnell A, Saour G, Duncan B, Nicoll A. Closure of schools during an influenza pandemic. The Lancet. Infectious diseases. 2009 Aug:9(8):473-81. doi: 10.1016/S1473-3099(09)70176-8. Epub
[PubMed PMID: 19628172]