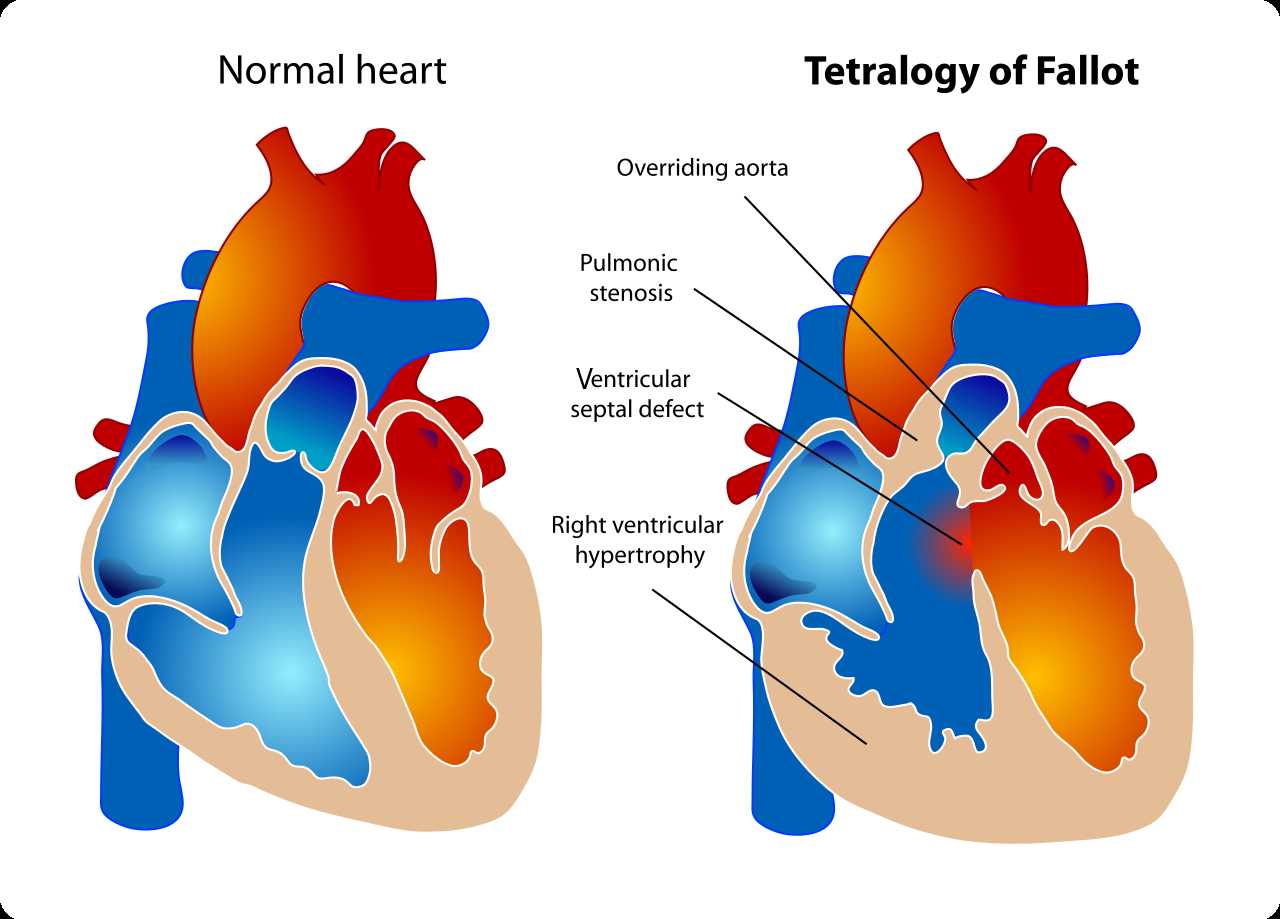
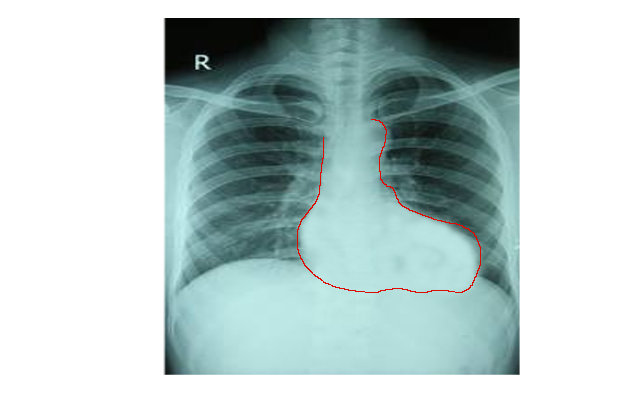
[1]
Barron DJ. Tetralogy of Fallot: controversies in early management. World journal for pediatric & congenital heart surgery. 2013 Apr:4(2):186-91. doi: 10.1177/2150135112471352. Epub
[PubMed PMID: 23799733]
[2]
Bailliard F, Anderson RH. Tetralogy of Fallot. Orphanet journal of rare diseases. 2009 Jan 13:4():2. doi: 10.1186/1750-1172-4-2. Epub 2009 Jan 13
[PubMed PMID: 19144126]
[3]
Karl TR, Stocker C. Tetralogy of Fallot and Its Variants. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies. 2016 Aug:17(8 Suppl 1):S330-6. doi: 10.1097/PCC.0000000000000831. Epub
[PubMed PMID: 27490619]
[4]
Bittel DC, Butler MG, Kibiryeva N, Marshall JA, Chen J, Lofland GK, O'Brien JE Jr. Gene expression in cardiac tissues from infants with idiopathic conotruncal defects. BMC medical genomics. 2011 Jan 5:4():1. doi: 10.1186/1755-8794-4-1. Epub 2011 Jan 5
[PubMed PMID: 21208432]
[5]
Matos-Nieves A, Yasuhara J, Garg V. Another Notch in the Genetic Puzzle of Tetralogy of Fallot. Circulation research. 2019 Feb 15:124(4):462-464. doi: 10.1161/CIRCRESAHA.118.314520. Epub
[PubMed PMID: 30763217]
[6]
Page DJ, Miossec MJ, Williams SG, Monaghan RM, Fotiou E, Cordell HJ, Sutcliffe L, Topf A, Bourgey M, Bourque G, Eveleigh R, Dunwoodie SL, Winlaw DS, Bhattacharya S, Breckpot J, Devriendt K, Gewillig M, Brook JD, Setchfield KJ, Bu'Lock FA, O'Sullivan J, Stuart G, Bezzina CR, Mulder BJM, Postma AV, Bentham JR, Baron M, Bhaskar SS, Black GC, Newman WG, Hentges KE, Lathrop GM, Santibanez-Koref M, Keavney BD. Whole Exome Sequencing Reveals the Major Genetic Contributors to Nonsyndromic Tetralogy of Fallot. Circulation research. 2019 Feb 15:124(4):553-563. doi: 10.1161/CIRCRESAHA.118.313250. Epub
[PubMed PMID: 30582441]
[7]
Chung IM, Rajakumar G. Genetics of Congenital Heart Defects: The NKX2-5 Gene, a Key Player. Genes. 2016 Jan 23:7(2):. doi: 10.3390/genes7020006. Epub 2016 Jan 23
[PubMed PMID: 26805889]
[8]
Villafañe J, Feinstein JA, Jenkins KJ, Vincent RN, Walsh EP, Dubin AM, Geva T, Towbin JA, Cohen MS, Fraser C, Dearani J, Rosenthal D, Kaufman B, Graham TP Jr, Adult Congenital and Pediatric Cardiology Section, American College of Cardiology. Hot topics in tetralogy of Fallot. Journal of the American College of Cardiology. 2013 Dec 10:62(23):2155-66. doi: 10.1016/j.jacc.2013.07.100. Epub 2013 Sep 27
[PubMed PMID: 24076489]
[9]
Pierpont ME, Basson CT, Benson DW Jr, Gelb BD, Giglia TM, Goldmuntz E, McGee G, Sable CA, Srivastava D, Webb CL, American Heart Association Congenital Cardiac Defects Committee, Council on Cardiovascular Disease in the Young. Genetic basis for congenital heart defects: current knowledge: a scientific statement from the American Heart Association Congenital Cardiac Defects Committee, Council on Cardiovascular Disease in the Young: endorsed by the American Academy of Pediatrics. Circulation. 2007 Jun 12:115(23):3015-38
[PubMed PMID: 17519398]
[10]
Pierpont ME, Brueckner M, Chung WK, Garg V, Lacro RV, McGuire AL, Mital S, Priest JR, Pu WT, Roberts A, Ware SM, Gelb BD, Russell MW, American Heart Association Council on Cardiovascular Disease in the Young; Council on Cardiovascular and Stroke Nursing; and Council on Genomic and Precision Medicine. Genetic Basis for Congenital Heart Disease: Revisited: A Scientific Statement From the American Heart Association. Circulation. 2018 Nov 20:138(21):e653-e711. doi: 10.1161/CIR.0000000000000606. Epub
[PubMed PMID: 30571578]
[11]
Wu MH, Lu CW, Chen HC, Kao FY, Huang SK. Adult Congenital Heart Disease in a Nationwide Population 2000-2014: Epidemiological Trends, Arrhythmia, and Standardized Mortality Ratio. Journal of the American Heart Association. 2018 Feb 8:7(4):. doi: 10.1161/JAHA.117.007907. Epub 2018 Feb 8
[PubMed PMID: 29437602]
Level 2 (mid-level) evidence
[12]
Bertranou EG, Blackstone EH, Hazelrig JB, Turner ME, Kirklin JW. Life expectancy without surgery in tetralogy of Fallot. The American journal of cardiology. 1978 Sep:42(3):458-66
[PubMed PMID: 685856]
[13]
Brickner ME, Hillis LD, Lange RA. Congenital heart disease in adults. Second of two parts. The New England journal of medicine. 2000 Feb 3:342(5):334-42
[PubMed PMID: 10655533]
[14]
O'Brien P, Marshall AC. Cardiology patient page. Tetralogy of Fallot. Circulation. 2014 Jul 22:130(4):e26-9. doi: 10.1161/CIRCULATIONAHA.113.005547. Epub
[PubMed PMID: 25047589]
[15]
Diller GP, Kempny A, Liodakis E, Alonso-Gonzalez R, Inuzuka R, Uebing A, Orwat S, Dimopoulos K, Swan L, Li W, Gatzoulis MA, Baumgartner H. Left ventricular longitudinal function predicts life-threatening ventricular arrhythmia and death in adults with repaired tetralogy of fallot. Circulation. 2012 May 22:125(20):2440-6. doi: 10.1161/CIRCULATIONAHA.111.086983. Epub 2012 Apr 11
[PubMed PMID: 22496160]
[16]
Mongeon FP, Gurvitz MZ, Broberg CS, Aboulhosn J, Opotowsky AR, Kay JD, Valente AM, Earing MG, Lui GK, Fernandes SM, Gersony DR, Cook SC, Ting JG, Nickolaus MJ, Landzberg MJ, Khairy P, Alliance for Adult Research in Congenital Cardiology (AARCC). Aortic root dilatation in adults with surgically repaired tetralogy of fallot: a multicenter cross-sectional study. Circulation. 2013 Jan 15:127(2):172-9. doi: 10.1161/CIRCULATIONAHA.112.129585. Epub 2012 Dec 6
[PubMed PMID: 23224208]
Level 2 (mid-level) evidence
[17]
van der Ven JPG, van den Bosch E, Bogers AJCC, Helbing WA. Current outcomes and treatment of tetralogy of Fallot. F1000Research. 2019:8():. pii: F1000 Faculty Rev-1530. doi: 10.12688/f1000research.17174.1. Epub 2019 Aug 29
[PubMed PMID: 31508203]
[18]
Salazar AM, Newth CC, Khemani RG, Jürg H, Ross PA. Pulmonary function testing in infants with tetralogy of Fallot and absent pulmonary valve syndrome. Annals of pediatric cardiology. 2015 May-Aug:8(2):108-12. doi: 10.4103/0974-2069.154152. Epub
[PubMed PMID: 26085760]
[19]
Wise-Faberowski L, Asija R, McElhinney DB. Tetralogy of Fallot: Everything you wanted to know but were afraid to ask. Paediatric anaesthesia. 2019 May:29(5):475-482. doi: 10.1111/pan.13569. Epub 2019 Apr 15
[PubMed PMID: 30592107]
[20]
Apitz C, Webb GD, Redington AN. Tetralogy of Fallot. Lancet (London, England). 2009 Oct 24:374(9699):1462-71. doi: 10.1016/S0140-6736(09)60657-7. Epub 2009 Aug 14
[PubMed PMID: 19683809]
[21]
Chelliah A, Moon-Grady AJ, Peyvandi S, Chiu JS, Bost JE, Schidlow D, Carroll SJ, Davey B, Divanovic A, Hornberger L, Howley LW, Kavanaugh-McHugh A, Kovalchin JP, Levasseur SM, Lindblade CL, Morris SA, Ngwezi D, Pruetz JD, Puchalski MD, Rychik J, Samai C, Tacy TA, Tworetzky W, Vernon MM, Yeh J, Donofrio MT. Contemporary Outcomes in Tetralogy of Fallot With Absent Pulmonary Valve After Fetal Diagnosis. Journal of the American Heart Association. 2021 Jun 15:10(12):e019713. doi: 10.1161/JAHA.120.019713. Epub 2021 Jun 8
[PubMed PMID: 34098741]
[22]
Recker F, Weber EC, Strizek B, Geipel A, Berg C, Gembruch U. Management and outcome of prenatal absent pulmonary valve syndrome. Archives of gynecology and obstetrics. 2022 Nov:306(5):1449-1454. doi: 10.1007/s00404-022-06397-4. Epub 2022 Jan 18
[PubMed PMID: 35043273]
[23]
Lumens J, Fan CS, Walmsley J, Yim D, Manlhiot C, Dragulescu A, Grosse-Wortmann L, Mertens L, Prinzen FW, Delhaas T, Friedberg MK. Relative Impact of Right Ventricular Electromechanical Dyssynchrony Versus Pulmonary Regurgitation on Right Ventricular Dysfunction and Exercise Intolerance in Patients After Repair of Tetralogy of Fallot. Journal of the American Heart Association. 2019 Jan 22:8(2):e010903. doi: 10.1161/JAHA.118.010903. Epub
[PubMed PMID: 30651018]
[24]
Havers-Borgersen E, Butt JH, Smerup M, Gislason GH, Torp-Pedersen C, Gröning M, Schmidt MR, Søndergaard L, Køber L, Fosbøl EL. Incidence of Infective Endocarditis Among Patients With Tetralogy of Fallot. Journal of the American Heart Association. 2021 Nov 16:10(22):e022445. doi: 10.1161/JAHA.121.022445. Epub 2021 Nov 3
[PubMed PMID: 34730003]
[25]
Sun HY, Proudfoot JA, McCandless RT. Prenatal detection of critical cardiac outflow tract anomalies remains suboptimal despite revised obstetrical imaging guidelines. Congenital heart disease. 2018 Sep:13(5):748-756. doi: 10.1111/chd.12648. Epub 2018 Jul 18
[PubMed PMID: 30022603]
[26]
Mawad W, Mertens LL. Recent Advances and Trends in Pediatric Cardiac Imaging. Current treatment options in cardiovascular medicine. 2018 Feb 21:20(1):9. doi: 10.1007/s11936-018-0599-x. Epub 2018 Feb 21
[PubMed PMID: 29468314]
Level 3 (low-level) evidence
[27]
Refaat MM, Ballout J, Mansour M. Ablation of Atrial Fibrillation in Patients with Congenital Heart Disease. Arrhythmia & electrophysiology review. 2017 Dec:6(4):191-194. doi: 10.15420/2017.2017.15.1. Epub
[PubMed PMID: 29326834]
[28]
Al Habib HF, Jacobs JP, Mavroudis C, Tchervenkov CI, O'Brien SM, Mohammadi S, Jacobs ML. Contemporary patterns of management of tetralogy of Fallot: data from the Society of Thoracic Surgeons Database. The Annals of thoracic surgery. 2010 Sep:90(3):813-9; discussion 819-20. doi: 10.1016/j.athoracsur.2010.03.110. Epub
[PubMed PMID: 20732501]
[29]
Sandoval JP, Chaturvedi RR, Benson L, Morgan G, Van Arsdell G, Honjo O, Caldarone C, Lee KJ. Right Ventricular Outflow Tract Stenting in Tetralogy of Fallot Infants With Risk Factors for Early Primary Repair. Circulation. Cardiovascular interventions. 2016 Dec:9(12):. pii: e003979. Epub
[PubMed PMID: 27965298]
[30]
Balzer D. Pulmonary Valve Replacement for Tetralogy of Fallot. Methodist DeBakey cardiovascular journal. 2019 Apr-Jun:15(2):122-132. doi: 10.14797/mdcj-15-2-122. Epub
[PubMed PMID: 31384375]
[31]
Bauser-Heaton H, Borquez A, Han B, Ladd M, Asija R, Downey L, Koth A, Algaze CA, Wise-Faberowski L, Perry SB, Shin A, Peng LF, Hanley FL, McElhinney DB. Programmatic Approach to Management of Tetralogy of Fallot With Major Aortopulmonary Collateral Arteries: A 15-Year Experience With 458 Patients. Circulation. Cardiovascular interventions. 2017 Apr:10(4):. pii: e004952. doi: 10.1161/CIRCINTERVENTIONS.116.004952. Epub
[PubMed PMID: 28356265]
[32]
Jonas RA. Surgical Management of Absent Pulmonary Valve Syndrome. World journal for pediatric & congenital heart surgery. 2016 Sep:7(5):600-4. doi: 10.1177/2150135116651838. Epub
[PubMed PMID: 27587495]
[33]
Valente AM, Gauvreau K, Assenza GE, Babu-Narayan SV, Schreier J, Gatzoulis MA, Groenink M, Inuzuka R, Kilner PJ, Koyak Z, Landzberg MJ, Mulder B, Powell AJ, Wald R, Geva T. Contemporary predictors of death and sustained ventricular tachycardia in patients with repaired tetralogy of Fallot enrolled in the INDICATOR cohort. Heart (British Cardiac Society). 2014 Feb:100(3):247-53. doi: 10.1136/heartjnl-2013-304958. Epub 2013 Oct 31
[PubMed PMID: 24179163]
[34]
Geva T. Tetralogy of Fallot repair: ready for a new paradigm. The Journal of thoracic and cardiovascular surgery. 2012 Jun:143(6):1305-6. doi: 10.1016/j.jtcvs.2012.01.076. Epub 2012 Feb 18
[PubMed PMID: 22342479]
[35]
Yim D, Riesenkampff E, Caro-Dominguez P, Yoo SJ, Seed M, Grosse-Wortmann L. Assessment of Diffuse Ventricular Myocardial Fibrosis Using Native T1 in Children With Repaired Tetralogy of Fallot. Circulation. Cardiovascular imaging. 2017 Mar:10(3):. pii: e005695. doi: 10.1161/CIRCIMAGING.116.005695. Epub
[PubMed PMID: 28292861]
[36]
Tretter JT, Redington AN. Risk Factors and Biomarkers of Poor Outcomes: Time to Throw Out Right Ventricular Volumes in Repaired Tetralogy of Fallot? Lessons From the INDICATOR Cohort. Circulation. 2018 Nov 6:138(19):2116-2118. doi: 10.1161/CIRCULATIONAHA.118.037000. Epub
[PubMed PMID: 30474414]
[37]
Chen CA, Dusenbery SM, Valente AM, Powell AJ, Geva T. Myocardial ECV Fraction Assessed by CMR Is Associated With Type of Hemodynamic Load and Arrhythmia in Repaired Tetralogy of Fallot. JACC. Cardiovascular imaging. 2016 Jan:9(1):1-10. doi: 10.1016/j.jcmg.2015.09.011. Epub 2015 Dec 9
[PubMed PMID: 26684969]
[38]
Hui W, Slorach C, Dragulescu A, Mertens L, Bijnens B, Friedberg MK. Mechanisms of right ventricular electromechanical dyssynchrony and mechanical inefficiency in children after repair of tetralogy of fallot. Circulation. Cardiovascular imaging. 2014 Jul:7(4):610-8. doi: 10.1161/CIRCIMAGING.113.001483. Epub 2014 May 1
[PubMed PMID: 24785673]
[39]
Andrade AC, Jerosch-Herold M, Wegner P, Gabbert DD, Voges I, Pham M, Shah R, Hedderich J, Kramer HH, Rickers C. Determinants of Left Ventricular Dysfunction and Remodeling in Patients With Corrected Tetralogy of Fallot. Journal of the American Heart Association. 2019 Sep 3:8(17):e009618. doi: 10.1161/JAHA.118.009618. Epub 2019 Aug 31
[PubMed PMID: 31474177]
[40]
Moon TJ, Choueiter N, Geva T, Valente AM, Gauvreau K, Harrild DM. Relation of biventricular strain and dyssynchrony in repaired tetralogy of fallot measured by cardiac magnetic resonance to death and sustained ventricular tachycardia. The American journal of cardiology. 2015 Mar 1:115(5):676-80. doi: 10.1016/j.amjcard.2014.12.024. Epub 2014 Dec 18
[PubMed PMID: 25727084]
[41]
Ghonim S, Gatzoulis MA, Ernst S, Li W, Moon JC, Smith GC, Heng EL, Keegan J, Ho SY, McCarthy KP, Shore DF, Uebing A, Kempny A, Alpendurada F, Diller GP, Dimopoulos K, Pennell DJ, Babu-Narayan SV. Predicting Survival in Repaired Tetralogy of Fallot: A Lesion-Specific and Personalized Approach. JACC. Cardiovascular imaging. 2022 Feb:15(2):257-268. doi: 10.1016/j.jcmg.2021.07.026. Epub 2021 Oct 13
[PubMed PMID: 34656466]
[42]
Krieger EV, Zeppenfeld K, DeWitt ES, Duarte VE, Egbe AC, Haeffele C, Lin KY, Robinson MR, Sillman C, Upadhyay S, American Heart Association Adults With Congenital Heart Disease Committee of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young and Council on Clinical Cardiology. Arrhythmias in Repaired Tetralogy of Fallot: A Scientific Statement From the American Heart Association. Circulation. Arrhythmia and electrophysiology. 2022 Nov:15(11):e000084. doi: 10.1161/HAE.0000000000000084. Epub 2022 Oct 20
[PubMed PMID: 36263773]
[43]
Dennis M, Moore B, Kotchetkova I, Pressley L, Cordina R, Celermajer DS. Adults with repaired tetralogy: low mortality but high morbidity up to middle age. Open heart. 2017:4(1):e000564. doi: 10.1136/openhrt-2016-000564. Epub 2017 Mar 1
[PubMed PMID: 28698799]
[44]
Bokma JP, Geva T, Sleeper LA, Lee JH, Lu M, Sompolinsky T, Babu-Narayan SV, Wald RM, Mulder BJM, Valente AM. Improved Outcomes After Pulmonary Valve Replacement in Repaired Tetralogy of Fallot. Journal of the American College of Cardiology. 2023 May 30:81(21):2075-2085. doi: 10.1016/j.jacc.2023.02.052. Epub
[PubMed PMID: 37225360]
[45]
Giglia TM, Massicotte MP, Tweddell JS, Barst RJ, Bauman M, Erickson CC, Feltes TF, Foster E, Hinoki K, Ichord RN, Kreutzer J, McCrindle BW, Newburger JW, Tabbutt S, Todd JL, Webb CL, American Heart Association Congenital Heart Defects Committee of the Council on Cardiovascular Disease in the Young, Council on Cardiovascular and Stroke Nursing, Council on Epidemiology and Prevention, and Stroke Council. Prevention and treatment of thrombosis in pediatric and congenital heart disease: a scientific statement from the American Heart Association. Circulation. 2013 Dec 17:128(24):2622-703. doi: 10.1161/01.cir.0000436140.77832.7a. Epub 2013 Nov 13
[PubMed PMID: 24226806]
[46]
Mouws EMJP, de Groot NMS, van de Woestijne PC, de Jong PL, Helbing WA, van Beynum IM, Bogers AJJC. Tetralogy of Fallot in the Current Era. Seminars in thoracic and cardiovascular surgery. 2019 Autumn:31(3):496-504. doi: 10.1053/j.semtcvs.2018.10.015. Epub 2018 Nov 2
[PubMed PMID: 30395964]
[47]
He D, Sznycer-Taub N, Cheng Y, McCarter R, Jonas RA, Hanumanthaiah S, Moak JP. Magnesium Lowers the Incidence of Postoperative Junctional Ectopic Tachycardia in Congenital Heart Surgical Patients: Is There a Relationship to Surgical Procedure Complexity? Pediatric cardiology. 2015 Aug:36(6):1179-85. doi: 10.1007/s00246-015-1141-5. Epub 2015 Mar 12
[PubMed PMID: 25762470]
[48]
Sahu MK, Das A, Siddharth B, Talwar S, Singh SP, Abraham A, Choudhury A. Arrhythmias in Children in Early Postoperative Period After Cardiac Surgery. World journal for pediatric & congenital heart surgery. 2018 Jan:9(1):38-46. doi: 10.1177/2150135117737687. Epub
[PubMed PMID: 29310559]
[49]
Hoffman TM, Bush DM, Wernovsky G, Cohen MI, Wieand TS, Gaynor JW, Spray TL, Rhodes LA. Postoperative junctional ectopic tachycardia in children: incidence, risk factors, and treatment. The Annals of thoracic surgery. 2002 Nov:74(5):1607-11
[PubMed PMID: 12440616]
[50]
Gatzoulis MA, Till JA, Redington AN. Depolarization-repolarization inhomogeneity after repair of tetralogy of Fallot. The substrate for malignant ventricular tachycardia? Circulation. 1997 Jan 21:95(2):401-4
[PubMed PMID: 9008456]
[51]
Cuypers JA, Menting ME, Konings EE, Opić P, Utens EM, Helbing WA, Witsenburg M, van den Bosch AE, Ouhlous M, van Domburg RT, Rizopoulos D, Meijboom FJ, Boersma E, Bogers AJ, Roos-Hesselink JW. Unnatural history of tetralogy of Fallot: prospective follow-up of 40 years after surgical correction. Circulation. 2014 Nov 25:130(22):1944-53. doi: 10.1161/CIRCULATIONAHA.114.009454. Epub 2014 Oct 23
[PubMed PMID: 25341442]
[52]
Cabalka AK, Asnes JD, Balzer DT, Cheatham JP, Gillespie MJ, Jones TK, Justino H, Kim DW, Lung TH, Turner DR, McElhinney DB. Transcatheter pulmonary valve replacement using the melody valve for treatment of dysfunctional surgical bioprostheses: A multicenter study. The Journal of thoracic and cardiovascular surgery. 2018 Apr:155(4):1712-1724.e1. doi: 10.1016/j.jtcvs.2017.10.143. Epub 2017 Dec 6
[PubMed PMID: 29395214]
Level 2 (mid-level) evidence
[53]
Niwa K. Metabolic syndrome and coronary artery disease in adults with congenital heart disease. Cardiovascular diagnosis and therapy. 2021 Apr:11(2):563-576. doi: 10.21037/cdt-20-781. Epub
[PubMed PMID: 33968634]
[54]
Marino BS, Lipkin PH, Newburger JW, Peacock G, Gerdes M, Gaynor JW, Mussatto KA, Uzark K, Goldberg CS, Johnson WH Jr, Li J, Smith SE, Bellinger DC, Mahle WT, American Heart Association Congenital Heart Defects Committee, Council on Cardiovascular Disease in the Young, Council on Cardiovascular Nursing, and Stroke Council. Neurodevelopmental outcomes in children with congenital heart disease: evaluation and management: a scientific statement from the American Heart Association. Circulation. 2012 Aug 28:126(9):1143-72
[PubMed PMID: 22851541]
[55]
Balci A, Drenthen W, Mulder BJ, Roos-Hesselink JW, Voors AA, Vliegen HW, Moons P, Sollie KM, van Dijk AP, van Veldhuisen DJ, Pieper PG. Pregnancy in women with corrected tetralogy of Fallot: occurrence and predictors of adverse events. American heart journal. 2011 Feb:161(2):307-13. doi: 10.1016/j.ahj.2010.10.027. Epub 2011 Jan 15
[PubMed PMID: 21315213]
[56]
Wang K, Xin J, Wang X, Yu H, Liu X. Pregnancy outcomes among 31 patients with tetralogy of Fallot, a retrospective study. BMC pregnancy and childbirth. 2019 Dec 10:19(1):486. doi: 10.1186/s12884-019-2630-y. Epub 2019 Dec 10
[PubMed PMID: 31823779]
Level 2 (mid-level) evidence
[57]
Phillips S, Pirics M. Congenital Heart Disease and Reproductive Risk: An Overview for Obstetricians, Cardiologists, and Primary Care Providers. Methodist DeBakey cardiovascular journal. 2017 Oct-Dec:13(4):238-242. doi: 10.14797/mdcj-13-4-238. Epub
[PubMed PMID: 29744016]
Level 3 (low-level) evidence
[58]
Townsley MM, Windsor J, Briston D, Alegria J, Ramakrishna H. Tetralogy of Fallot: Perioperative Management and Analysis of Outcomes. Journal of cardiothoracic and vascular anesthesia. 2019 Feb:33(2):556-565. doi: 10.1053/j.jvca.2018.03.035. Epub 2018 Mar 24
[PubMed PMID: 29706570]
[59]
Morray BH, McElhinney DB, Boudjemline Y, Gewillig M, Kim DW, Grant EK, Bocks ML, Martin MH, Armstrong AK, Berman D, Danon S, Hoyer M, Delaney JW, Justino H, Qureshi AM, Meadows JJ, Jones TK. Multicenter Experience Evaluating Transcatheter Pulmonary Valve Replacement in Bovine Jugular Vein (Contegra) Right Ventricle to Pulmonary Artery Conduits. Circulation. Cardiovascular interventions. 2017 Jun:10(6):. pii: e004914. doi: 10.1161/CIRCINTERVENTIONS.116.004914. Epub
[PubMed PMID: 28600328]
[60]
Van Hare GF, Ackerman MJ, Evangelista JA, Kovacs RJ, Myerburg RJ, Shafer KM, Warnes CA, Washington RL, American Heart Association Electrocardiography and Arrhythmias Committee of Council on Clinical Cardiology, Council on Cardiovascular Disease in Young, Council on Cardiovascular and Stroke Nursing, Council on Functional Genomics and Translational Biology, and American College of Cardiology. Eligibility and Disqualification Recommendations for Competitive Athletes With Cardiovascular Abnormalities: Task Force 4: Congenital Heart Disease: A Scientific Statement From the American Heart Association and American College of Cardiology. Circulation. 2015 Dec 1:132(22):e281-91. doi: 10.1161/CIR.0000000000000240. Epub 2015 Nov 2
[PubMed PMID: 26621645]
[61]
Quattrone A, Lie OH, Nestaas E, de Lange C, Try K, Lindberg HL, Skulstad H, Erikssen G, Edvardsen T, Haugaa K, Estensen ME. Impact of pregnancy and risk factors for ventricular arrhythmias in women with tetralogy of Fallot. Open heart. 2021 Jan:8(1):. doi: 10.1136/openhrt-2020-001400. Epub
[PubMed PMID: 33414183]
[62]
Pedersen LM, Pedersen TA, Ravn HB, Hjortdal VE. Outcomes of pregnancy in women with tetralogy of Fallot. Cardiology in the young. 2008 Aug:18(4):423-9. doi: 10.1017/S1047951108002345. Epub 2008 Jun 18
[PubMed PMID: 18559134]
[63]
Veldtman GR, Connolly HM, Grogan M, Ammash NM, Warnes CA. Outcomes of pregnancy in women with tetralogy of Fallot. Journal of the American College of Cardiology. 2004 Jul 7:44(1):174-80
[PubMed PMID: 15234429]
[64]
Diller GP, Baumgartner H. Endocarditis in adults with congenital heart disease: new answers-new questions. European heart journal. 2017 Jul 7:38(26):2057-2059. doi: 10.1093/eurheartj/ehx044. Epub
[PubMed PMID: 28329113]
[65]
Canobbio MM, Warnes CA, Aboulhosn J, Connolly HM, Khanna A, Koos BJ, Mital S, Rose C, Silversides C, Stout K, American Heart Association Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology; Council on Cardiovascular Disease in the Young; Council on Functional Genomics and Translational Biology; and Council on Quality of Care and Outcomes Research. Management of Pregnancy in Patients With Complex Congenital Heart Disease: A Scientific Statement for Healthcare Professionals From the American Heart Association. Circulation. 2017 Feb 21:135(8):e50-e87. doi: 10.1161/CIR.0000000000000458. Epub 2017 Jan 12
[PubMed PMID: 28082385]
Level 2 (mid-level) evidence