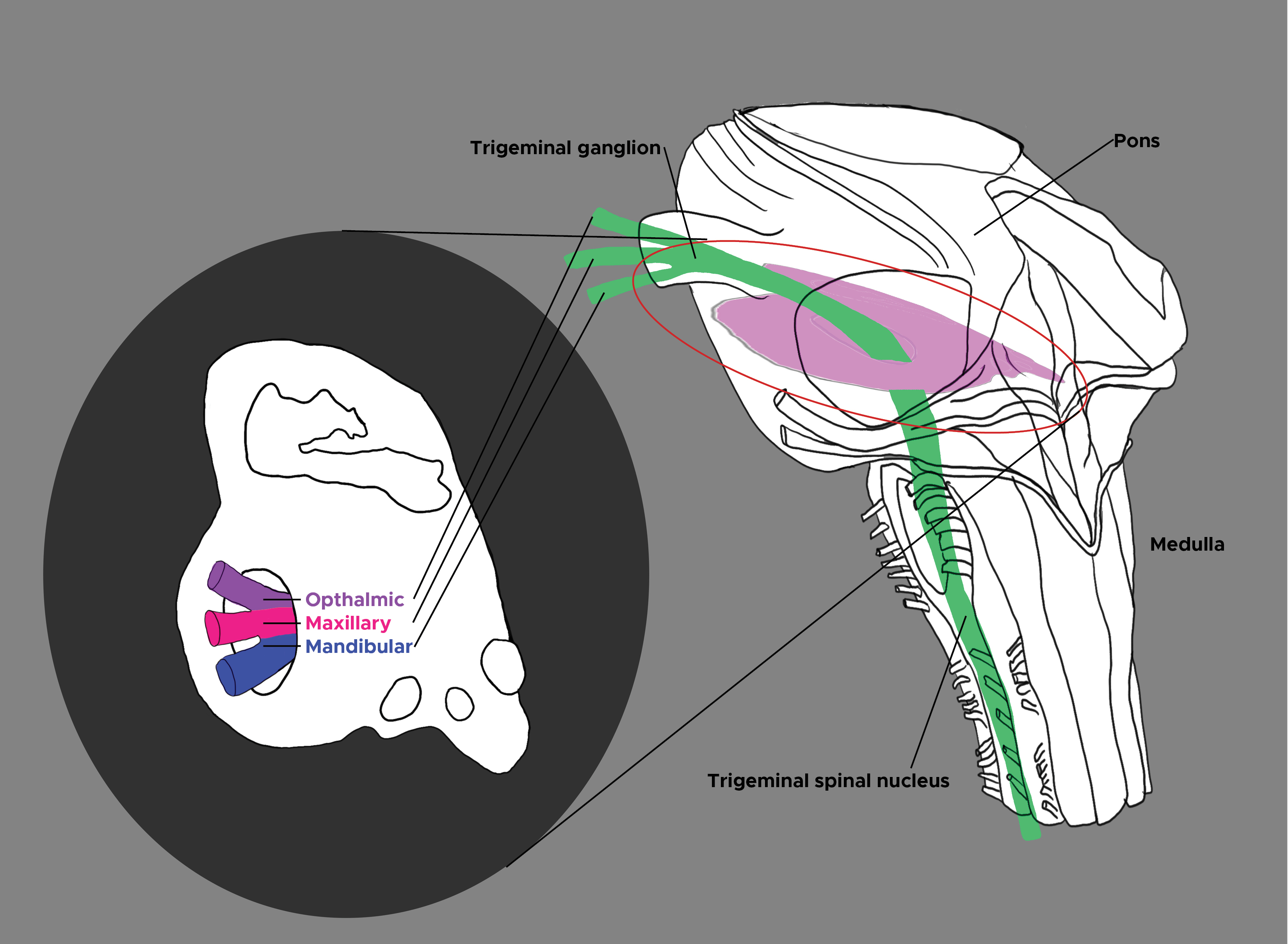
[1]
Henssen DJ, Kurt E, Kozicz T, van Dongen R, Bartels RH, van Cappellen van Walsum AM. New Insights in Trigeminal Anatomy: A Double Orofacial Tract for Nociceptive Input. Frontiers in neuroanatomy. 2016:10():53. doi: 10.3389/fnana.2016.00053. Epub 2016 May 10
[PubMed PMID: 27242449]
[2]
Bartsch T, Goadsby PJ. Increased responses in trigeminocervical nociceptive neurons to cervical input after stimulation of the dura mater. Brain : a journal of neurology. 2003 Aug:126(Pt 8):1801-13
[PubMed PMID: 12821523]
[3]
Marur T, Tuna Y, Demirci S. Facial anatomy. Clinics in dermatology. 2014 Jan-Feb:32(1):14-23. doi: 10.1016/j.clindermatol.2013.05.022. Epub
[PubMed PMID: 24314374]
[4]
Erzurumlu RS,Murakami Y,Rijli FM, Mapping the face in the somatosensory brainstem. Nature reviews. Neuroscience. 2010 Apr
[PubMed PMID: 20179712]
[5]
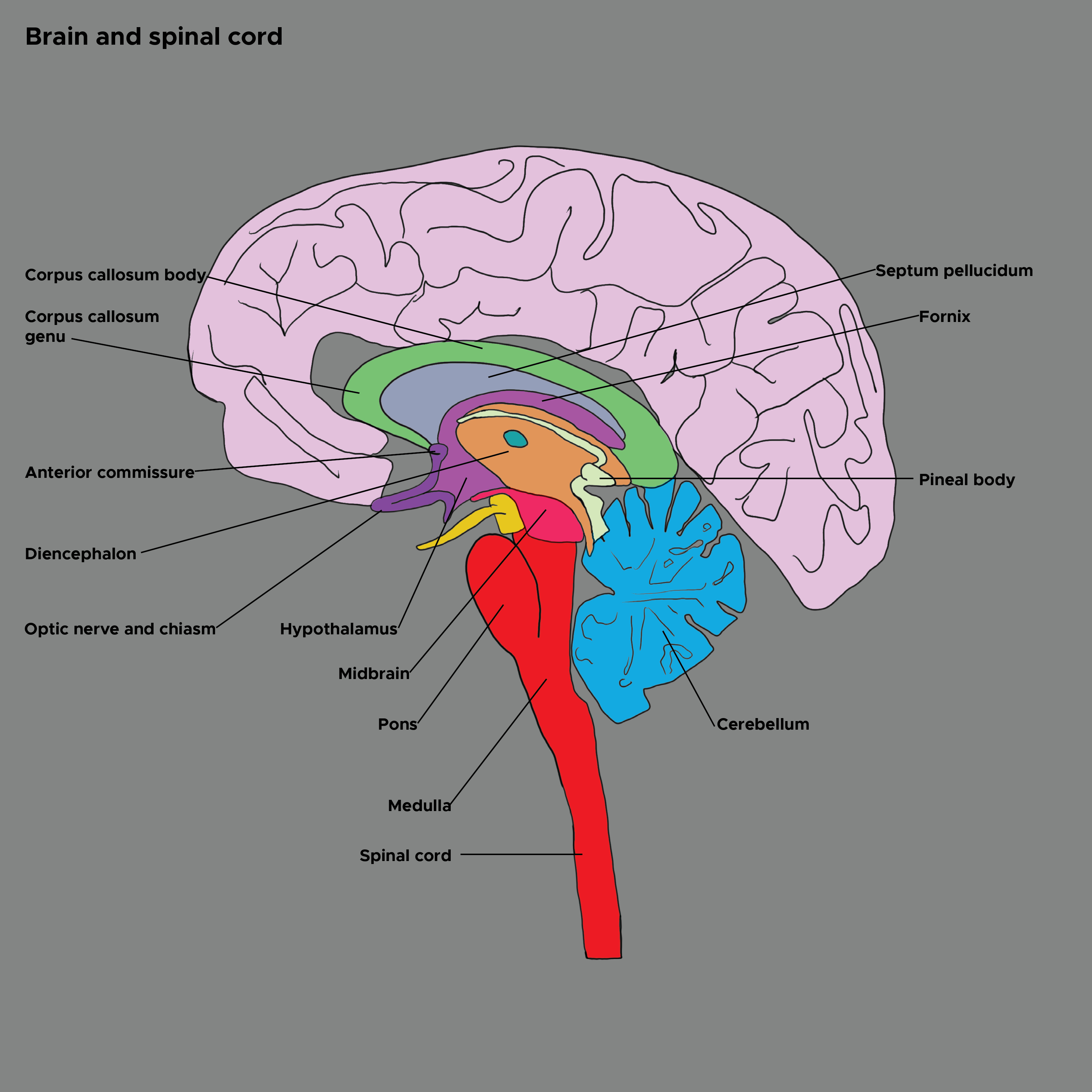
Henssen DJHA, Derks B, van Doorn M, Verhoogt NC, Staats P, Vissers K, Van Cappellen van Walsum AM. Visualizing the trigeminovagal complex in the human medulla by combining ex-vivo ultra-high resolution structural MRI and polarized light imaging microscopy. Scientific reports. 2019 Aug 5:9(1):11305. doi: 10.1038/s41598-019-47855-5. Epub 2019 Aug 5
[PubMed PMID: 31383932]
[6]
Bradnam L, Barry C. The role of the trigeminal sensory nuclear complex in the pathophysiology of craniocervical dystonia. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2013 Nov 20:33(47):18358-67. doi: 10.1523/JNEUROSCI.3544-13.2013. Epub
[PubMed PMID: 24259561]
[7]
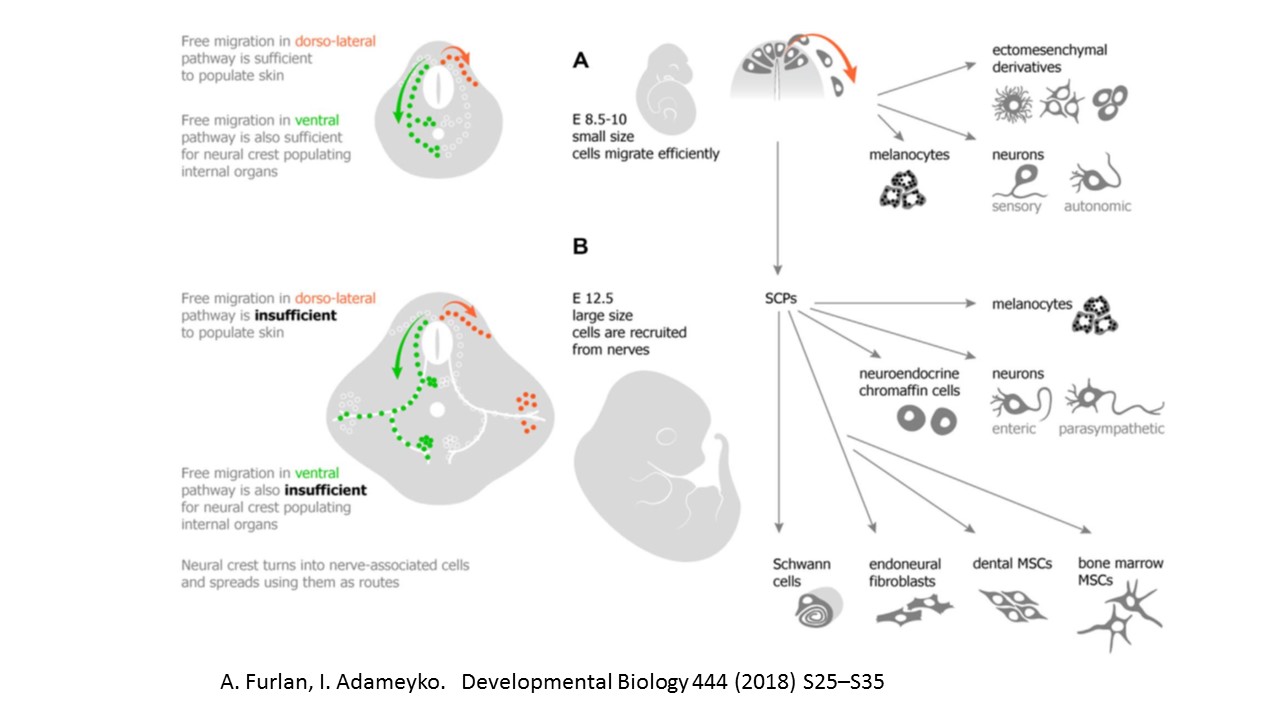
Méndez-Maldonado K, Vega-López GA, Aybar MJ, Velasco I. Neurogenesis From Neural Crest Cells: Molecular Mechanisms in the Formation of Cranial Nerves and Ganglia. Frontiers in cell and developmental biology. 2020:8():635. doi: 10.3389/fcell.2020.00635. Epub 2020 Aug 7
[PubMed PMID: 32850790]
[8]
Wang CZ,Shi M,Yang LL,Yang RQ,Luo ZG,Jacquin MF,Chen ZF,Ding YQ, Development of the mesencephalic trigeminal nucleus requires a paired homeodomain transcription factor, Drg11. Molecular and cellular neurosciences. 2007 Jun
[PubMed PMID: 17482477]
[9]
Novy J. Spinal cord syndromes. Frontiers of neurology and neuroscience. 2012:30():195-8. doi: 10.1159/000333682. Epub 2012 Feb 14
[PubMed PMID: 22377894]
[10]
Saylam C, Ucerler H, Orhan M, Cagli S, Zileli M. The relationship of the posterior inferior cerebellar artery to cranial nerves VII-XII. Clinical anatomy (New York, N.Y.). 2007 Nov:20(8):886-91
[PubMed PMID: 17907205]
[11]
Miao HL, Zhang DY, Wang T, Jiao XT, Jiao LQ. Clinical Importance of the Posterior Inferior Cerebellar Artery: A Review of the Literature. International journal of medical sciences. 2020:17(18):3005-3019. doi: 10.7150/ijms.49137. Epub 2020 Oct 18
[PubMed PMID: 33173421]
[12]
Cloud GC,Markus HS, Diagnosis and management of vertebral artery stenosis. QJM : monthly journal of the Association of Physicians. 2003 Jan
[PubMed PMID: 12509646]
[13]
Ruedrich ED, Chikkannaiah M, Kumar G. Wallenberg's lateral medullary syndrome in an adolescent. The American journal of emergency medicine. 2016 Nov:34(11):2254.e1-2254.e2. doi: 10.1016/j.ajem.2016.05.022. Epub 2016 May 12
[PubMed PMID: 27246963]
[14]
Day GS, Swartz RH, Chenkin J, Shamji AI, Frost DW. Lateral medullary syndrome: a diagnostic approach illustrated through case presentation and literature review. CJEM. 2014 Mar:16(2):164-70
[PubMed PMID: 24626124]
Level 3 (low-level) evidence
[15]
Cruccu G. Trigeminal Neuralgia. Continuum (Minneapolis, Minn.). 2017 Apr:23(2, Selected Topics in Outpatient Neurology):396-420. doi: 10.1212/CON.0000000000000451. Epub
[PubMed PMID: 28375911]
[16]
Peker S, Sirin A. Primary trigeminal neuralgia and the role of pars oralis of the spinal trigeminal nucleus. Medical hypotheses. 2017 Mar:100():15-18. doi: 10.1016/j.mehy.2017.01.008. Epub 2017 Jan 16
[PubMed PMID: 28236840]
[17]
Lee KH. Facial pain: trigeminal neuralgia. Annals of the Academy of Medicine, Singapore. 1993 Mar:22(2):193-6
[PubMed PMID: 8363331]
[18]
Merrill RL, Goodman D. Chronic Orofacial Pain and Behavioral Medicine. Oral and maxillofacial surgery clinics of North America. 2016 Aug:28(3):247-60. doi: 10.1016/j.coms.2016.03.007. Epub
[PubMed PMID: 27475505]
[19]
Okada S, Katagiri A, Saito H, Lee J, Ohara K, Iinuma T, Bereiter DA, Iwata K. Differential activation of ascending noxious pathways associated with trigeminal nerve injury. Pain. 2019 Jun:160(6):1342-1360. doi: 10.1097/j.pain.0000000000001521. Epub
[PubMed PMID: 30747907]
[20]
Wilcox SL, Gustin SM, Macey PM, Peck CC, Murray GM, Henderson LA. Anatomical changes at the level of the primary synapse in neuropathic pain: evidence from the spinal trigeminal nucleus. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2015 Feb 11:35(6):2508-15. doi: 10.1523/JNEUROSCI.3756-14.2015. Epub
[PubMed PMID: 25673845]
[21]
Albanese A, Bhatia K, Bressman SB, Delong MR, Fahn S, Fung VS, Hallett M, Jankovic J, Jinnah HA, Klein C, Lang AE, Mink JW, Teller JK. Phenomenology and classification of dystonia: a consensus update. Movement disorders : official journal of the Movement Disorder Society. 2013 Jun 15:28(7):863-73. doi: 10.1002/mds.25475. Epub 2013 May 6
[PubMed PMID: 23649720]
Level 3 (low-level) evidence
[22]
Lohmann K, Klein C. Genetics of dystonia: what's known? What's new? What's next? Movement disorders : official journal of the Movement Disorder Society. 2013 Jun 15:28(7):899-905. doi: 10.1002/mds.25536. Epub
[PubMed PMID: 23893446]
[23]
Serrao M, Rossi P, Parisi L, Perrotta A, Bartolo M, Cardinali P, Amabile G, Pierelli F. Trigemino-cervical-spinal reflexes in humans. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2003 Sep:114(9):1697-703
[PubMed PMID: 12948799]
[24]
Blood AJ, Kuster JK, Woodman SC, Kirlic N, Makhlouf ML, Multhaupt-Buell TJ, Makris N, Parent M, Sudarsky LR, Sjalander G, Breiter H, Breiter HC, Sharma N. Evidence for altered basal ganglia-brainstem connections in cervical dystonia. PloS one. 2012:7(2):e31654. doi: 10.1371/journal.pone.0031654. Epub 2012 Feb 22
[PubMed PMID: 22384048]
[25]
Murata Y, Li MZ, Masuko S. Developmental expression of oxytocin receptors in the neonatal medulla oblongata and pons. Neuroscience letters. 2011 Sep 20:502(3):157-61. doi: 10.1016/j.neulet.2011.07.032. Epub 2011 Jul 29
[PubMed PMID: 21820489]
Level 3 (low-level) evidence