Introduction
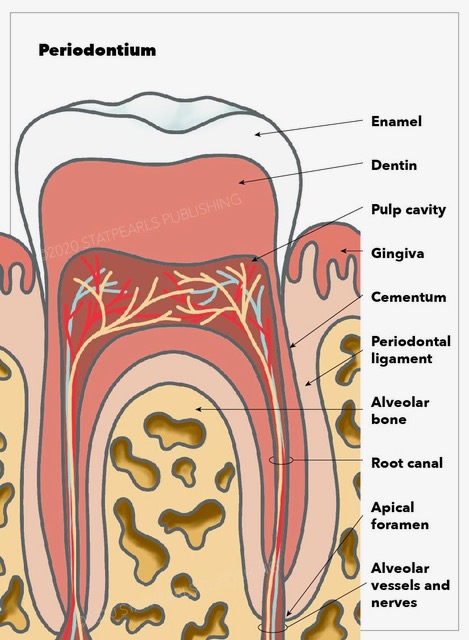
The periodontium is a connective tissue consisting of four components: cementum, the periodontal ligament (PDL), alveolar bone, and gingival tissue.[1] This article will detail the histological and embryological structure and origin of this tissue, as well as the function, clinical significance, and pathophysiology of some of the periodontal diseases.
Structure
Embryologically, PDL, cementum, and alveolar bone development are accompanied by the formation of the root of the tooth and its eruption.[2] However, the gingival component of the periodontium is derived from the ectoderm of the pharyngeal arches. It contains keratinized stratified squamous epithelium on its masticatory surface and non-keratinized on its crevicular and junctional surface.[1] PDL, cementum, and alveolar bone, as mentioned, are derived from neuro-mesenchymal stem cells of the dental sac.[3]
To understand the gingival epithelium better, it can be studied as 3 different sections, the oral epithelium, sulcular epithelium, and junctional epithelium, which is the closest to the tooth surface and attaches to it with hemidesmosomes. Histologically cementum is comprised of 2 parts cellular and acellular. The gingival epithelium is composed of stratified squamous epithelium.
Function
The periodontium functions include supporting the tooth, protecting it against oral microflora, and making the attachment of the tooth to the bone possible.[1] To understand the function of the periodontium even further, we can look at its components function individually.
The periodontal ligament (PDL) is a connective tissue that consists mostly of type I collagen bands and fibroblasts. PDL connects the cementum of the tooth to the gingivae in the alveolar socket using collagen bands. On the other hand, fibroblasts have a role in forming and repairing the alveolar bone and cementum.[4] Besides attaching the tooth roots to the bone and gingival tissue, PDL is also involved in proprioception by relaying information to the somatosensory cortex. This is important for avoiding occlusal forces during mastication and speech.[5]
Cementum is the mineralized tissue covering the dentin surfaces and is known to be the attachment site for PDL.
Tissue Preparation
Various processing techniques have been suggested and studied in the literature, exploring different fixing and demineralizing solutions of tooth and periodontium. In light microscopy (LM) analysis done on histological processing of teeth and periodontium, it was demonstrated that 10% buffered neutral formalin was used as a fixative solution and 10% pH 7.3 EDTA, as a demineralizing solution, has shown satisfactory results. To obtain morphometric and morphological satisfaction, paraffin-embedded sections of the sample are obtained, each with a thickness of 6 μm. These slides were then stained with hematoxylin and eosin (H&E). This routine protocol is also beneficial since it allows the possibility of serial sectioning for more specific techniques. For instance, techniques such as histochemical and immunohistochemical analyses that are more fitting for evaluating cellular components and extracellular matrix of the periodontium can be done using this routine protocol.[6] The biologically oriented preparation technique is a reliable alternative to conventional horizontal finish lines.[7]
In a cross-sectional study conducted in Turkey by Alkan et al., the effects of antibiotic therapy in oral histology were studied. The results showed that in healthy participants in oral histology trials, a minimum of 3 months should pass after the final dose of amoxicillin to avoid the negative effect of gingival tissue smear samples.[8]
Pathophysiology
Understanding the structure and histology of the periodontium is clinically significant to the discussion of wound healing, drugs and environmental factors affecting it, periodontal diseases, and overall health risk factors. Lamina propria of gingiva regenerates more readily due to fiber differentiation after wounding, compared to the alveolar bone, which relies on osteocyte, bone marrow cells, endosteum cells, and osteogenic cells of periosteum, which occupy different compartments.[1]
Histopathology of periodontal diseases is described in 4 stages:[9]
- Initially, the gingival crevicular fluid (GCF) amount is increased due to vascular changes in response to the initial insult. At this benign stage, polymorphonuclear neutrophils are attracted to the lesion site, and T lymphocytes are responsible for fibroblasts in the area.
- The early lesion is characterized by redness of the lesion. At this stage, PMNs clear and breakdown the collagen fibers, which leads to an increase in the previously made space for infiltrates.
- At this stage, the established lesion is dominated by B cells and leukocyte aggregation. This will initiate the lesion side transformation by changing both junctional and sulcular epithelium into an extremely vulnerable epithelium called the pocket epithelium. This is apparent as bleeding upon gentle gingival manipulation.
- An advanced lesion characterized as loss of gingival fibers and alveolar bone is caused by migration of biofilm into the pocket and creating an environment for anaerobic bacterias proliferation.
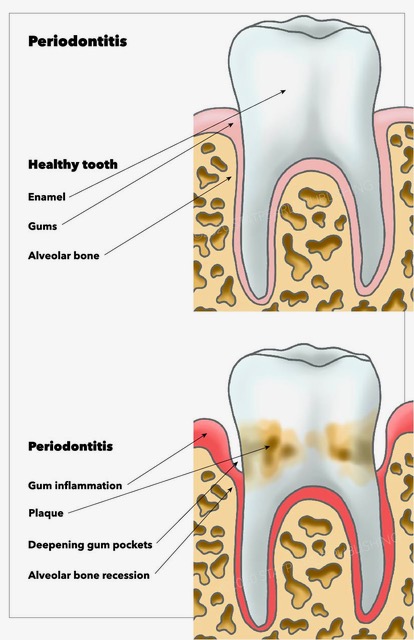
Gingivitis is an initial response to combat the first insult to the periodontium. As the name suggests is the inflammation of the gingiva, which is a component of the periodontium resulting from the accumulation of microbial plaque near the gingival sulcus. Gingivitis has a delayed appearance in children with a predominance of T lymphocyte infiltrates.[10] Most cases are transient, non-progressive, and reversible. However, it can progress to Periodontitis.
Periodontitis is the inflammation of the periodontium as a whole which extends to the jawbone and is irreversible. Pathophysiology of the periodontitis is explained by replacement and remodeling of previously normal alveolar bone, especially the spongy bone, that can happen in healthy individuals due to chronic inflammation. White blood cells are active during inflammation, and osteoclasts lineage is related to WBC. As a result, osteoblast is inhibited without reducing the rate of osteoclasts. It is observed on a radiograph as reduced opacity of the alveolar crest.[11]
Hyper-cementosis is thought to be due to growth factors disturbances in conditions such as acromegaly, gigantism, or Paget’s disease. Cementoblasts are located within the PDL and can undergo remodeling and repair. Excessive cementoblast deposition can cause hyper-cementosis. This can further cause concrescence which is the adherence of two teeth together by the roots (hence a radiograph is needed before tooth extraction). This is a rare finding that has been seen in patients with systemic lupus erythematosus (SLE).[12]
Cementicle is a condition in which masses of acellular cementum appear in abnormal location due to disruption of oppositional growth of cementum. This condition happens mostly in the elderly and appears as psammoma body-like calcifications. The process is thought to develop due to the encounter of cementoblasts with thrombus lodged in a nearby capillary.[13][14]
Mesial drift: Also known as physiological drift, is referred to as the migration of teeth in a mesial direction caused by asymmetrical remodeling of alveolar bone tissue. Two major concepts are believed to cause this drift: First, unbalanced bone remodeling of 2 adjacent teeth. And second, remodeling due to occlusal forces leads to an imbalance of acellular cementum. This asymmetrical remodeling causes tooth movement.[15]
Peri-implantitis is due to the failure to create the junction between the implant and the oral mucosa, and that leaves the underlying connective tissue exposed to the permanent oral microbiome and creates chronic inflammation of gingival and bone tissue.[11] Knowledge of histology helped to demonstrate that coating the peri-implant pocket with hyaluronic acid (HA) gel reduces inflammation in peri-implantitis by improving the connection between the core and recipients’ connective tissue through reducing inflammation.[16]
Widening of PDL space: with occlusal/orthodontic trauma, the fibroblast in PDL responds by increasing its activity and lead to the widening of PDL (meaning the neighboring tissue is lost). This can be seen on a radiograph and can be accompanied by bone loss or hyper-cementosis. Certain medications such as bisphosphonate are used to manage rheumatoid arthritis osteoporosis, osteogenesis imperfecta, multiple myeloma.[4] can cause PDL widening. In contrast, conditions in which bone health such as radiation-induced bone defect are affected and reduce the force and also cause widening of the PDL space. Infection and inflammation, and malignancy also can cause PDL widening.
Clinical Significance
The periodontium is also significant because of its effect on systemic health and environmental risk factors affecting it.
Some medications reported as affecting the periodontium include anti-convulsants (diphenylhydantoin), immunosuppressants, corticosteroids, NSAIDs, and CCBs.[17] These drugs could directly affect the periodontium by increasing matrix production or decreasing its degradation and, as a result, cause gingival hyperplasia.[18]
Smoking and bacterial biofilms within periodontal pockets are known to be the common causes of loss of attachment between cementum, and alveolar bone promotes tooth loss.[19] Periodontal diseases have also been studied as a risk for dental implants over time.[20] This study has shown that periodontal status and smoking are important contributors to long-term implant failure.
Epidemiological studies have shown that periodontal diseases are associated with an increased risk of preterm birth and low birth weight (LBW). This can happen by direct vertical transfer of maternal bacteria that cause inflammation (Gram-negative bacteria contained in the dental plaque biofilm, such as Porphyromonas gingivalis and Fusobacterium nucleatum.[11] It is also believed the release of inflammatory cytokines, specifically IL-lα, IL-1β, IL-6, and IL-8 and systemic inflammation, is a contribution to preterm birth and LBW. This emphasizes the importance of oral hygiene measurements and preventions during pregnancy.[21]