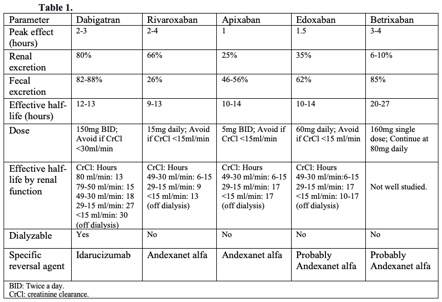
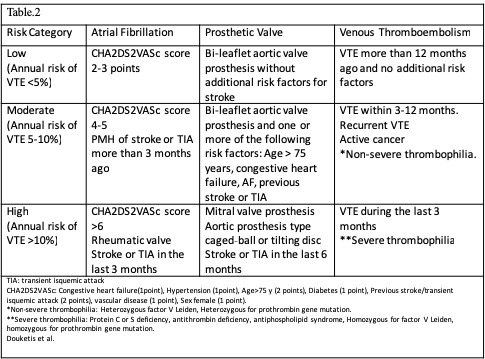
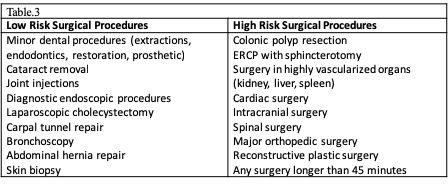
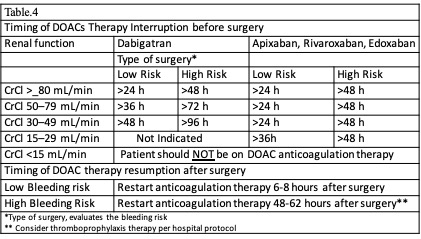
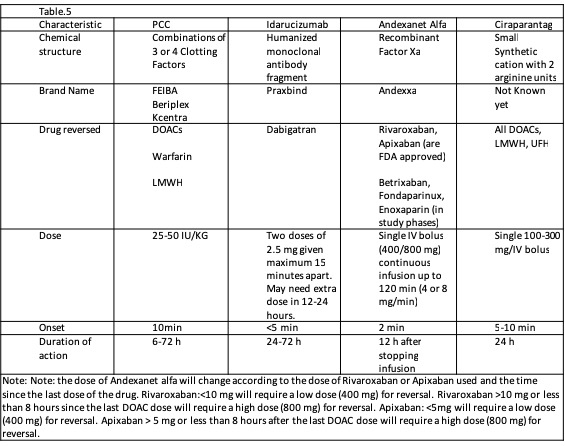
[1]
Zulkifly H, Lip GYH, Lane DA. Epidemiology of atrial fibrillation. International journal of clinical practice. 2018 Mar:72(3):e13070. doi: 10.1111/ijcp.13070. Epub 2018 Mar 1
[PubMed PMID: 29493854]
[2]
Doherty JU, Gluckman TJ, Hucker WJ, Januzzi JL Jr, Ortel TL, Saxonhouse SJ, Spinler SA. 2017 ACC Expert Consensus Decision Pathway for Periprocedural Management of Anticoagulation in Patients With Nonvalvular Atrial Fibrillation: A Report of the American College of Cardiology Clinical Expert Consensus Document Task Force. Journal of the American College of Cardiology. 2017 Feb 21:69(7):871-898. doi: 10.1016/j.jacc.2016.11.024. Epub 2017 Jan 9
[PubMed PMID: 28081965]
Level 3 (low-level) evidence
[3]
Kalra K, Franzese CJ, Gesheff MG, Lev EI, Pandya S, Bliden KP, Tantry US, Gurbel PA. Pharmacology of antiplatelet agents. Current atherosclerosis reports. 2013 Dec:15(12):371. doi: 10.1007/s11883-013-0371-3. Epub
[PubMed PMID: 24142550]
[4]
Narouze S, Benzon HT, Provenzano D, Buvanendran A, De Andres J, Deer T, Rauck R, Huntoon MA. Interventional Spine and Pain Procedures in Patients on Antiplatelet and Anticoagulant Medications (Second Edition): Guidelines From the American Society of Regional Anesthesia and Pain Medicine, the European Society of Regional Anaesthesia and Pain Therapy, the American Academy of Pain Medicine, the International Neuromodulation Society, the North American Neuromodulation Society, and the World Institute of Pain. Regional anesthesia and pain medicine. 2018 Apr:43(3):225-262. doi: 10.1097/AAP.0000000000000700. Epub
[PubMed PMID: 29278603]
[5]
Hall R, Mazer CD. Antiplatelet drugs: a review of their pharmacology and management in the perioperative period. Anesthesia and analgesia. 2011 Feb:112(2):292-318. doi: 10.1213/ANE.0b013e318203f38d. Epub 2011 Jan 6
[PubMed PMID: 21212258]
[6]
Ferri N, Corsini A, Bellosta S. Pharmacology of the new P2Y12 receptor inhibitors: insights on pharmacokinetic and pharmacodynamic properties. Drugs. 2013 Oct:73(15):1681-709. doi: 10.1007/s40265-013-0126-z. Epub
[PubMed PMID: 24114622]
[7]
Capodanno D, Milluzzo RP, Angiolillo DJ. Intravenous antiplatelet therapies (glycoprotein IIb/IIIa receptor inhibitors and cangrelor) in percutaneous coronary intervention: from pharmacology to indications for clinical use. Therapeutic advances in cardiovascular disease. 2019 Jan-Dec:13():1753944719893274. doi: 10.1177/1753944719893274. Epub
[PubMed PMID: 31823688]
Level 3 (low-level) evidence
[8]
Detsi A, Kontogiorgis C, Hadjipavlou-Litina D. Coumarin derivatives: an updated patent review (2015-2016). Expert opinion on therapeutic patents. 2017 Nov:27(11):1201-1226. doi: 10.1080/13543776.2017.1360284. Epub 2017 Aug 14
[PubMed PMID: 28756713]
Level 3 (low-level) evidence
[9]
Ansell J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008 Jun:133(6 Suppl):160S-198S. doi: 10.1378/chest.08-0670. Epub
[PubMed PMID: 18574265]
Level 1 (high-level) evidence
[10]
Gómez-Outes A, Suárez-Gea ML, Lecumberri R, Terleira-Fernández AI, Vargas-Castrillón E. Direct-acting oral anticoagulants: pharmacology, indications, management, and future perspectives. European journal of haematology. 2015 Nov:95(5):389-404. doi: 10.1111/ejh.12610. Epub 2015 Jul 16
[PubMed PMID: 26095540]
Level 3 (low-level) evidence
[11]
Alquwaizani M, Buckley L, Adams C, Fanikos J. Anticoagulants: A Review of the Pharmacology, Dosing, and Complications. Current emergency and hospital medicine reports. 2013 Jun:1(2):83-97
[PubMed PMID: 23687625]
[12]
Douketis JD, Spyropoulos AC, Spencer FA, Mayr M, Jaffer AK, Eckman MH, Dunn AS, Kunz R. Perioperative management of antithrombotic therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012 Feb:141(2 Suppl):e326S-e350S. doi: 10.1378/chest.11-2298. Epub
[PubMed PMID: 22315266]
Level 1 (high-level) evidence
[13]
Milling TJ Jr, Ziebell CM. A review of oral anticoagulants, old and new, in major bleeding and the need for urgent surgery. Trends in cardiovascular medicine. 2020 Feb:30(2):86-90. doi: 10.1016/j.tcm.2019.03.004. Epub 2019 Mar 26
[PubMed PMID: 30952383]
[14]
January CT, Wann LS, Calkins H, Chen LY, Cigarroa JE, Cleveland JC Jr, Ellinor PT, Ezekowitz MD, Field ME, Furie KL, Heidenreich PA, Murray KT, Shea JB, Tracy CM, Yancy CW. 2019 AHA/ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Journal of the American College of Cardiology. 2019 Jul 9:74(1):104-132. doi: 10.1016/j.jacc.2019.01.011. Epub 2019 Jan 28
[PubMed PMID: 30703431]
Level 1 (high-level) evidence
[15]
Mehdi Z, Birns J, Partridge J, Bhalla A, Dhesi J. Perioperative management of adult patients with a history of stroke or transient ischaemic attack undergoing elective non-cardiac surgery. Clinical medicine (London, England). 2016 Dec:16(6):535-540
[PubMed PMID: 27927817]
[16]
Omran H, Bauersachs R, Rübenacker S, Goss F, Hammerstingl C. The HAS-BLED score predicts bleedings during bridging of chronic oral anticoagulation. Results from the national multicentre BNK Online bRiDging REgistRy (BORDER). Thrombosis and haemostasis. 2012 Jul:108(1):65-73. doi: 10.1160/TH11-12-0827. Epub 2012 Apr 26
[PubMed PMID: 22534746]
[17]
Vivas D, Roldán I, Ferrandis R, Marín F, Roldán V, Tello-Montoliu A, Ruiz-Nodar JM, Gómez-Doblas JJ, Martín A, Llau JV, Ramos-Gallo MJ, Muñoz R, Arcelus JI, Leyva F, Alberca F, Oliva R, Gómez AM, Montero C, Arikan F, Ley L, Santos-Bueso E, Figuero E, Bujaldón A, Urbano J, Otero R, Hermida JF, Egocheaga I, Llisterri JL, Lobos JM, Serrano A, Madridano O, Ferreiro JL, Expert reviewers. Perioperative and Periprocedural Management of Antithrombotic Therapy: Consensus Document of SEC, SEDAR, SEACV, SECTCV, AEC, SECPRE, SEPD, SEGO, SEHH, SETH, SEMERGEN, SEMFYC, SEMG, SEMICYUC, SEMI, SEMES, SEPAR, SENEC, SEO, SEPA, SERVEI, SECOT and AEU. Revista espanola de cardiologia (English ed.). 2018 Jul:71(7):553-564. doi: 10.1016/j.rec.2018.01.029. Epub 2018 Jun 8
[PubMed PMID: 29887180]
Level 3 (low-level) evidence
[18]
Hornor MA, Duane TM, Ehlers AP, Jensen EH, Brown PS Jr, Pohl D, da Costa PM, Ko CY, Laronga C. American College of Surgeons' Guidelines for the Perioperative Management of Antithrombotic Medication. Journal of the American College of Surgeons. 2018 Nov:227(5):521-536.e1. doi: 10.1016/j.jamcollsurg.2018.08.183. Epub 2018 Aug 24
[PubMed PMID: 30145286]
[19]
Steffel J, Verhamme P, Potpara TS, Albaladejo P, Antz M, Desteghe L, Haeusler KG, Oldgren J, Reinecke H, Roldan-Schilling V, Rowell N, Sinnaeve P, Collins R, Camm AJ, Heidbüchel H, ESC Scientific Document Group. The 2018 European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation. European heart journal. 2018 Apr 21:39(16):1330-1393. doi: 10.1093/eurheartj/ehy136. Epub
[PubMed PMID: 29562325]
[20]
Fabbro M 2nd, Dunn S, Rodriguez-Blanco YF, Jain P. A Narrative Review for Perioperative Physicians of the 2017 ACC Expert Consensus Decision Pathway on Management of Bleeding in Patients on Oral Anticoagulants. Journal of cardiothoracic and vascular anesthesia. 2019 Feb:33(2):290-301. doi: 10.1053/j.jvca.2018.07.023. Epub 2018 Jul 20
[PubMed PMID: 30146466]
Level 3 (low-level) evidence
[21]
Childers CP, Maggard-Gibbons M, Ulloa JG, MacQueen IT, Miake-Lye IM, Shanman R, Mak S, Beroes JM, Shekelle PG. Perioperative management of antiplatelet therapy in patients undergoing non-cardiac surgery following coronary stent placement: a systematic review. Systematic reviews. 2018 Jan 10:7(1):4. doi: 10.1186/s13643-017-0635-z. Epub 2018 Jan 10
[PubMed PMID: 29321066]
Level 1 (high-level) evidence
[22]
Clark NP, Douketis JD, Hasselblad V, Schulman S, Kindzelski AL, Ortel TL, BRIDGE Investigators. Predictors of perioperative major bleeding in patients who interrupt warfarin for an elective surgery or procedure: Analysis of the BRIDGE trial. American heart journal. 2018 Jan:195():108-114. doi: 10.1016/j.ahj.2017.09.015. Epub 2017 Sep 21
[PubMed PMID: 29224638]
[23]
Precht C, Demirel Y, Assaf AT, Pinnschmidt HO, Knipfer C, Hanken H, Friedrich RE, Wikner J. Perioperative Management in Patients With Undergoing Direct Oral Anticoagulant Therapy in Oral Surgery - A Multicentric Questionnaire Survey. In vivo (Athens, Greece). 2019 May-Jun:33(3):855-862. doi: 10.21873/invivo.11550. Epub
[PubMed PMID: 31028208]
Level 3 (low-level) evidence
[24]
Chahine J, Khoudary MN, Nasr S. Anticoagulation Use prior to Common Dental Procedures: A Systematic Review. Cardiology research and practice. 2019:2019():9308631. doi: 10.1155/2019/9308631. Epub 2019 Jun 2
[PubMed PMID: 31275643]
Level 1 (high-level) evidence
[25]
Shahi V, Brinjikji W, Murad MH, Asirvatham SJ, Kallmes DF. Safety of Uninterrupted Warfarin Therapy in Patients Undergoing Cardiovascular Endovascular Procedures: A Systematic Review and Meta-Analysis. Radiology. 2016 Feb:278(2):383-94. doi: 10.1148/radiol.2015142531. Epub 2015 Jul 23
[PubMed PMID: 26203535]
Level 1 (high-level) evidence
[26]
Hess CN, Norgren L, Ansel GM, Capell WH, Fletcher JP, Fowkes FGR, Gottsäter A, Hitos K, Jaff MR, Nordanstig J, Hiatt WR. A Structured Review of Antithrombotic Therapy in Peripheral Artery Disease With a Focus on Revascularization: A TASC (InterSociety Consensus for the Management of Peripheral Artery Disease) Initiative. Circulation. 2017 Jun 20:135(25):2534-2555. doi: 10.1161/CIRCULATIONAHA.117.024469. Epub
[PubMed PMID: 28630267]
Level 3 (low-level) evidence
[27]
Cappato R, Marchlinski FE, Hohnloser SH, Naccarelli GV, Xiang J, Wilber DJ, Ma CS, Hess S, Wells DS, Juang G, Vijgen J, Hügl BJ, Balasubramaniam R, De Chillou C, Davies DW, Fields LE, Natale A, VENTURE-AF Investigators. Uninterrupted rivaroxaban vs. uninterrupted vitamin K antagonists for catheter ablation in non-valvular atrial fibrillation. European heart journal. 2015 Jul 21:36(28):1805-11. doi: 10.1093/eurheartj/ehv177. Epub 2015 May 14
[PubMed PMID: 25975659]
[28]
Calkins H, Willems S, Gerstenfeld EP, Verma A, Schilling R, Hohnloser SH, Okumura K, Serota H, Nordaby M, Guiver K, Biss B, Brouwer MA, Grimaldi M, RE-CIRCUIT Investigators. Uninterrupted Dabigatran versus Warfarin for Ablation in Atrial Fibrillation. The New England journal of medicine. 2017 Apr 27:376(17):1627-1636. doi: 10.1056/NEJMoa1701005. Epub 2017 Mar 19
[PubMed PMID: 28317415]
[29]
Birnie DH, Healey JS, Wells GA, Verma A, Tang AS, Krahn AD, Simpson CS, Ayala-Paredes F, Coutu B, Leiria TL, Essebag V, BRUISE CONTROL Investigators. Pacemaker or defibrillator surgery without interruption of anticoagulation. The New England journal of medicine. 2013 May 30:368(22):2084-93. doi: 10.1056/NEJMoa1302946. Epub 2013 May 9
[PubMed PMID: 23659733]
[30]
Birnie DH, Healey JS, Wells GA, Ayala-Paredes F, Coutu B, Sumner GL, Becker G, Verma A, Philippon F, Kalfon E, Eikelboom J, Sandhu RK, Nery PB, Lellouche N, Connolly SJ, Sapp J, Essebag V. Continued vs. interrupted direct oral anticoagulants at the time of device surgery, in patients with moderate to high risk of arterial thrombo-embolic events (BRUISE CONTROL-2). European heart journal. 2018 Nov 21:39(44):3973-3979. doi: 10.1093/eurheartj/ehy413. Epub
[PubMed PMID: 30462279]
[31]
Tafur A, Douketis J. Perioperative management of anticoagulant and antiplatelet therapy. Heart (British Cardiac Society). 2018 Sep:104(17):1461-1467. doi: 10.1136/heartjnl-2016-310581. Epub 2017 Dec 7
[PubMed PMID: 29217632]
[32]
Siegal D, Yudin J, Kaatz S, Douketis JD, Lim W, Spyropoulos AC. Periprocedural heparin bridging in patients receiving vitamin K antagonists: systematic review and meta-analysis of bleeding and thromboembolic rates. Circulation. 2012 Sep 25:126(13):1630-9
[PubMed PMID: 22912386]
Level 1 (high-level) evidence
[33]
Essebag V, Proietti R, Birnie DH, Wang J, Douketis J, Coutu B, Parkash R, Lip GYH, Hohnloser SH, Moriarty A, Oldgren J, Connolly SJ, Ezekowitz M, Healey JS. Short-term dabigatran interruption before cardiac rhythm device implantation: multi-centre experience from the RE-LY trial. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2017 Oct 1:19(10):1630-1636. doi: 10.1093/europace/euw409. Epub
[PubMed PMID: 28339794]
[34]
Wight JM, Columb MO. Perioperative bridging anticoagulation for atrial fibrillation-the first randomised controlled trial. Perioperative medicine (London, England). 2016:5():14. doi: 10.1186/s13741-016-0040-5. Epub 2016 Jun 7
[PubMed PMID: 27280017]
Level 1 (high-level) evidence
[35]
Douketis JD. Perioperative management of patients who are receiving warfarin therapy: an evidence-based and practical approach. Blood. 2011 May 12:117(19):5044-9. doi: 10.1182/blood-2011-02-329979. Epub 2011 Mar 17
[PubMed PMID: 21415266]
[36]
Skelley JW, Thomason AR, Nolen JC, Candidate P. Betrixaban (Bevyxxa): A Direct-Acting Oral Anticoagulant Factor Xa Inhibitor. P & T : a peer-reviewed journal for formulary management. 2018 Feb:43(2):85-120
[PubMed PMID: 29386864]
[37]
Douketis JD, Spyropoulos AC, Duncan J, Carrier M, Le Gal G, Tafur AJ, Vanassche T, Verhamme P, Shivakumar S, Gross PL, Lee AYY, Yeo E, Solymoss S, Kassis J, Le Templier G, Kowalski S, Blostein M, Shah V, MacKay E, Wu C, Clark NP, Bates SM, Spencer FA, Arnaoutoglou E, Coppens M, Arnold DM, Caprini JA, Li N, Moffat KA, Syed S, Schulman S. Perioperative Management of Patients With Atrial Fibrillation Receiving a Direct Oral Anticoagulant. JAMA internal medicine. 2019 Nov 1:179(11):1469-1478. doi: 10.1001/jamainternmed.2019.2431. Epub
[PubMed PMID: 31380891]
[38]
Yee J, Kaide CG. Emergency Reversal of Anticoagulation. The western journal of emergency medicine. 2019 Aug 6:20(5):770-783. doi: 10.5811/westjem.2018.5.38235. Epub 2019 Aug 6
[PubMed PMID: 31539334]
[39]
Sarode R, Milling TJ Jr, Refaai MA, Mangione A, Schneider A, Durn BL, Goldstein JN. Efficacy and safety of a 4-factor prothrombin complex concentrate in patients on vitamin K antagonists presenting with major bleeding: a randomized, plasma-controlled, phase IIIb study. Circulation. 2013 Sep 10:128(11):1234-43. doi: 10.1161/CIRCULATIONAHA.113.002283. Epub 2013 Aug 9
[PubMed PMID: 23935011]
Level 1 (high-level) evidence
[40]
Siddiqui F, Tafur A, Ramacciotti LS, Jeske W, Hoppensteadt D, Ramacciotti E, Iqbal O, Fareed J. Reversal of Factor Xa Inhibitors by Andexanet Alfa May Increase Thrombogenesis Compared to Pretreatment Values. Clinical and applied thrombosis/hemostasis : official journal of the International Academy of Clinical and Applied Thrombosis/Hemostasis. 2019 Jan-Dec:25():1076029619863493. doi: 10.1177/1076029619863493. Epub
[PubMed PMID: 31298056]
[41]
Kustos SA, Fasinu PS. Direct-Acting Oral Anticoagulants and Their Reversal Agents-An Update. Medicines (Basel, Switzerland). 2019 Oct 15:6(4):. doi: 10.3390/medicines6040103. Epub 2019 Oct 15
[PubMed PMID: 31618893]