Continuing Education Activity
Alopecia totalis (AT) is a chronic condition marked by complete scalp hair loss, which affects a small percentage of patients with alopecia areata (AA). AA is a chronic, autoimmune disorder of nonscarring hair loss mediated by T-lymphocytes against the hair follicular unit. The most common form of disease manifests as smooth, discrete, round patches of hair loss and can be referred to as alopecia areata focalis. AT is a more extensive subtype of AA characterized by complete loss of hair on the scalp. When there is a total loss of scalp and body hair, it is called alopecia universalis.
This activity discusses the evaluation and management of AT, emphasizing its distinctive characteristics and expounding upon the challenges associated with treating it, given its poorer prognosis for hair regrowth and heightened likelihood of treatment failure compared with the focal variant of AA. Learners gain insights into the crucial aspects of early recognition and intervention strategies for AT, optimizing treatment outcomes by addressing the condition at its onset. This course underscores the significance of an interprofessional approach in the care continuum for patients grappling with this autoimmune disorder, accentuating the collaborative role of various healthcare disciplines in enhancing patient care and management. Further, this activity equips healthcare professionals with the necessary knowledge and tools to effectively evaluate, treat, and navigate the complexities of AT, thereby improving patient outcomes and quality of life for individuals affected by this condition.
Objectives:
Identify the underlying autoimmune etiology of alopecia totalis by recognizing the specific T-lymphocyte–mediated mechanism targeting hair follicles.
Identify distinct physical examination findings of alopecia totalis, including complete hair loss on the scalp without scarring, distinguishing it from other forms of alopecia.
Differentiate between various treatment options for alopecia totalis based on their mechanisms of action, efficacy, and potential side effects, tailoring choices to individual patient profiles.
Implement effective collaboration and communication among interprofessional team members to improve outcomes and treatment efficacy for patients with alopecia totalis.
Introduction
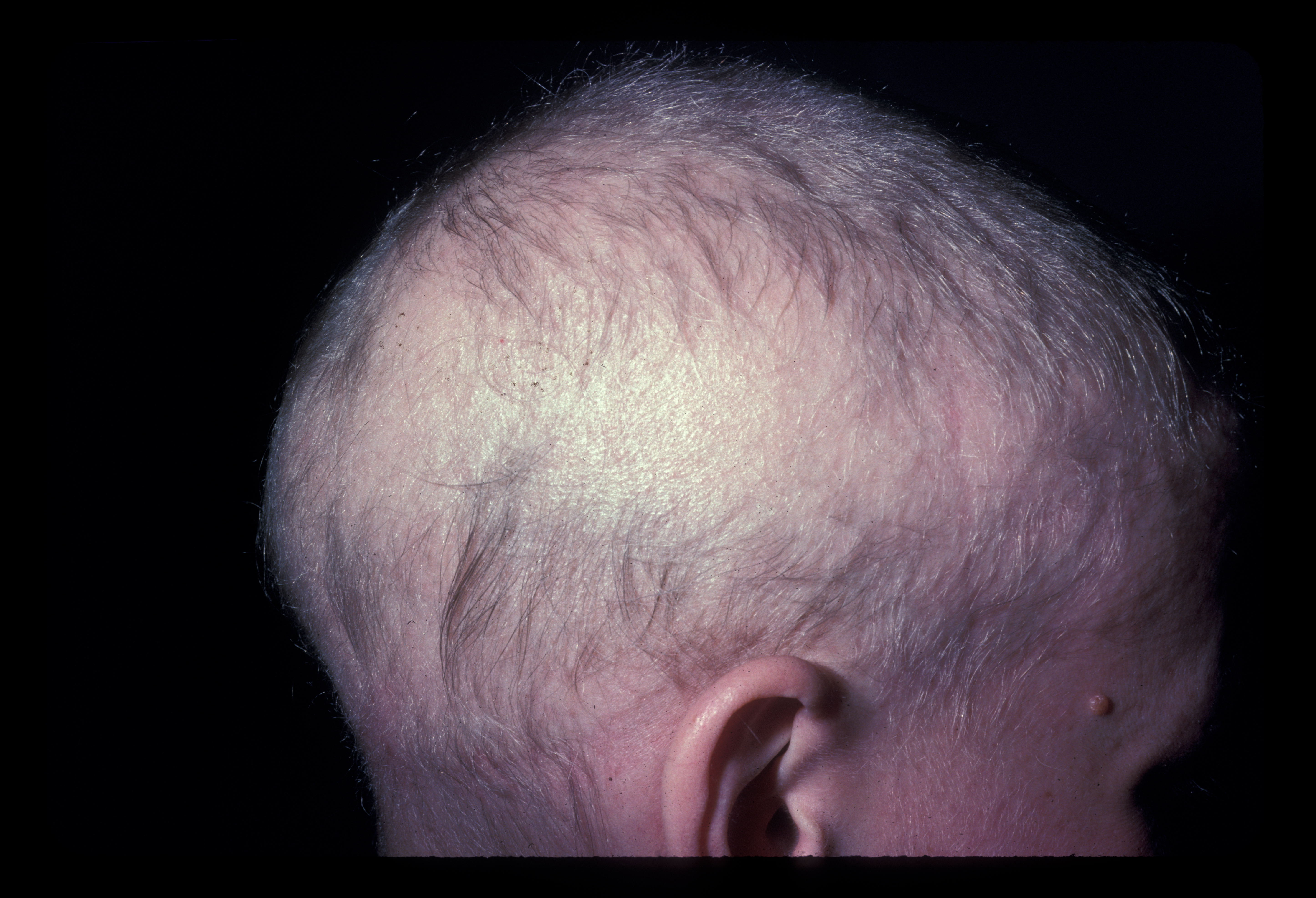
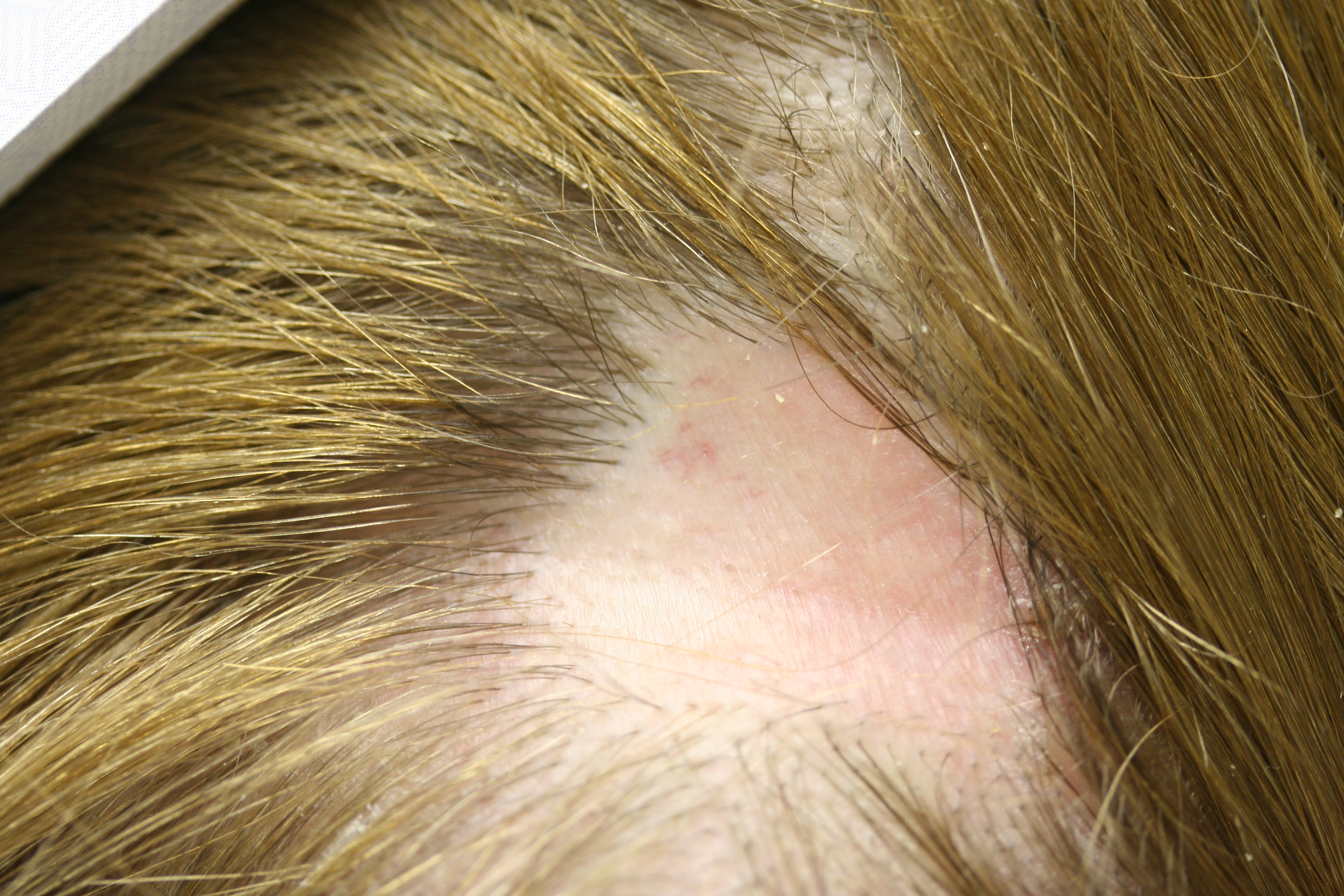
Alopecia areata (AA) is a chronic, autoimmune disorder of nonscarring hair loss mediated by T-lymphocytes against the hair follicular unit (see Image. Alopecia Areata, Localized Hair Loss).[1] The most common form of disease manifests as smooth, discrete, round patches of hair loss and can be referred to as alopecia areata focalis (AF). Alopecia totalis (AT) is a more extensive subtype of AA characterized by complete loss of hair on the scalp.[2] When there is a total loss of scalp and body hair, it is called alopecia universalis (AU). Compared to focal AA, patients with AT have a poorer prognosis for hair regrowth and are more likely to have treatment failure (see Image. Alopecia Totalis With Regrowth).[3]
Etiology
Genetic and environmental factors contribute to the development of AT. Twin and familial studies support a genetic predisposition to the disease. Studies of identical twins report a concordance rate of 55%. A positive family history of AA is found in about 20% of patients with AT.[4]
Genetic predisposition has been supported by genome-wide associated studies identifying certain human leukocyte antigen (HLA) genes associated with the development of AA. HLA-DRB1 polymorphisms have increased susceptibility for AA, specifically HLA-DRB1*04 and HLA-DRB1*16.[5] HLA-DQB1*03 has also been found to be associated with AA. A few polymorphisms have been identified that are more highly associated with AT and AU than AF; these include HLA-DRB1*1104 and HLA-DQB1*0301.[6][7]
In addition to genetic influence, environmental factors such as illness, infection, drugs, and stress may contribute to flares or relapse of disease.[4]
Epidemiology
There is approximately a 2% lifetime risk of developing AA, but the development of AT is less common. AT is estimated to affect 0.03% of the population, and the prevalence has been increasing in recent years.[8] For patients with AF, less than 10% will go on to develop AT.[9] Children and young adults are more commonly affected, but AT can present at any age.[10] There is no sex predilection for this disease.[11]
Pathophysiology
There are multiple theories on the pathogenesis of AA. Central to these is the loss of the immune privilege of the hair follicle, leading to an attack of the follicle by the immune system. The loss of immune privilege may occur secondary to an environmental insult such as a virus, or it may be a spontaneous autoimmune phenomenon. In normal conditions, healthy anagen hair follicles express major histocompatibility complex (MHC) class I at low levels, which prevents molecular presentation as autoantigens. When this process is disturbed, and MHC expression is increased, immune privilege is lost, as previously hidden antigens may be presented to T-cells.[4]
CD8+ T-cells, in particular, are known to play a key role in the pathogenesis. These T-cells produce interferon (IFN)-gamma, which activates interleukin (IL)-2, IL-7, IL-15, and IL-21 cytokines. IL-15 suppresses regulatory T cells, promoting the proliferation of natural killer cells. Downstream, these cytokines signal the Janus kinase/ signal transducer and activator of the transcription (JAK/STAT) pathway. JAK3 is overexpressed in patients with AA.[12]
Histopathology
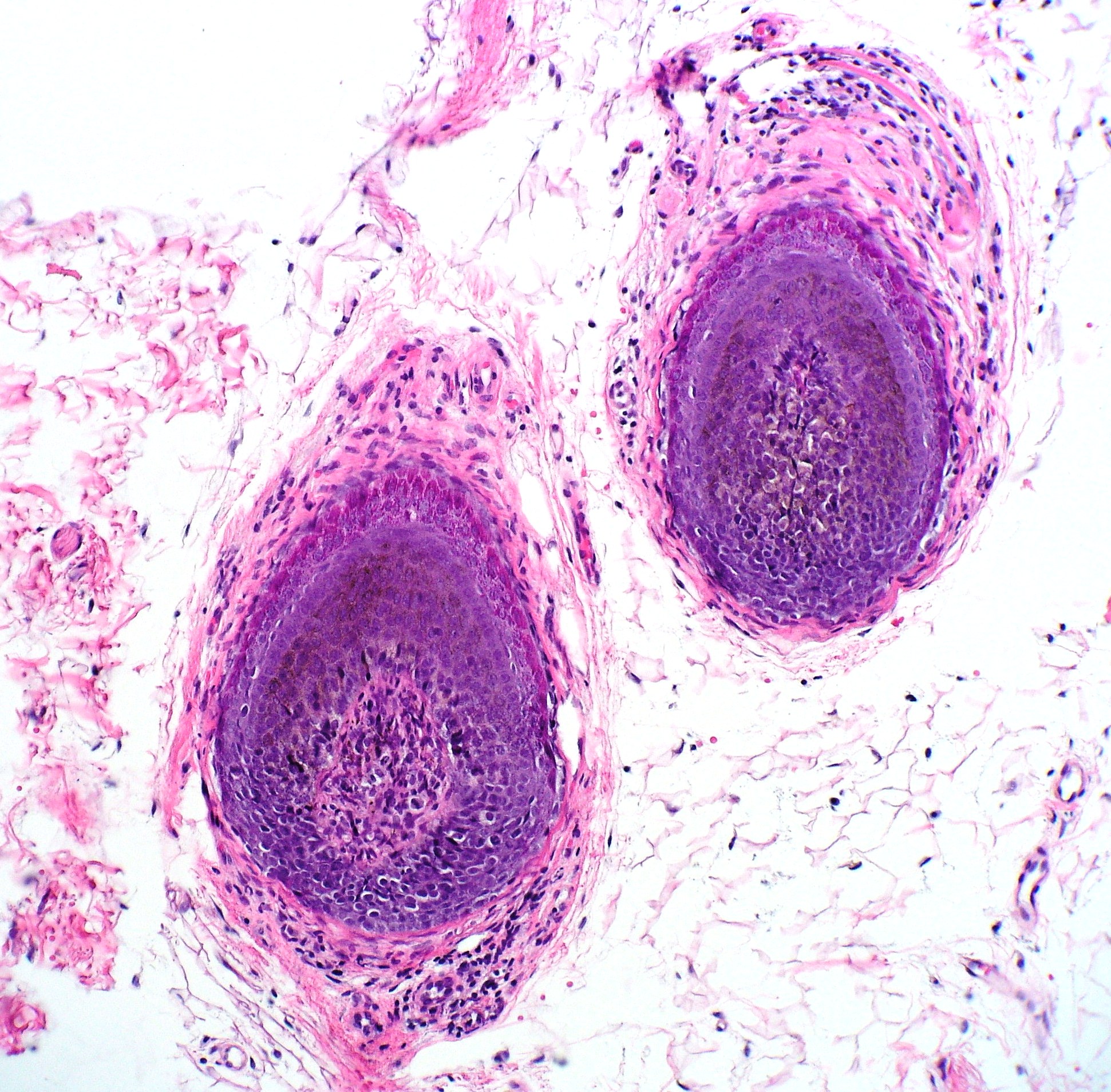
Early in the disease course, histologic examination reveals a normal total number of hair follicles. Anagen hairs often have lymphocytic inflammation clustered around the bulbar portion of the follicle, said to resemble a "swarm of bees." Eosinophils may be seen as well. In longstanding disease, histologic examination reveals mostly catagen and telogen hair follicles, often displaying miniaturization. Fibrous tract remnants known as "follicular streamers" may be seen, but these can also be a finding in scarring alopecias.[13][14] See Image. Alopecia Areata Biopsy.
History and Physical
Elucidating a thorough history is critical in patients presenting with extensive hair loss. Most patients presenting with AT will have a history of discrete, patchy hair loss, which has progressed to total scalp involvement, though only 5%-10% of patients with AF progress to AT or AU.[8] For patients with AT, the most common initial presentation is multifocal AA, defined as 5 or more discrete patches. The time from the initial development of AA to progression to AT is, on average 1 year, and within 4 years in 90% of patients.[4]
Family history and past medical history are also important to obtain. There is a positive family history of hair loss in about 20% of cases. Comorbid conditions are common in patients with AA and might be slightly more frequent in those with AT, affecting about 20% of patients. Comorbidities in order of highest frequency include autoimmune thyroid disease, vitiligo, type I diabetes mellitus, atopy, and inflammatory bowel disease.[4]
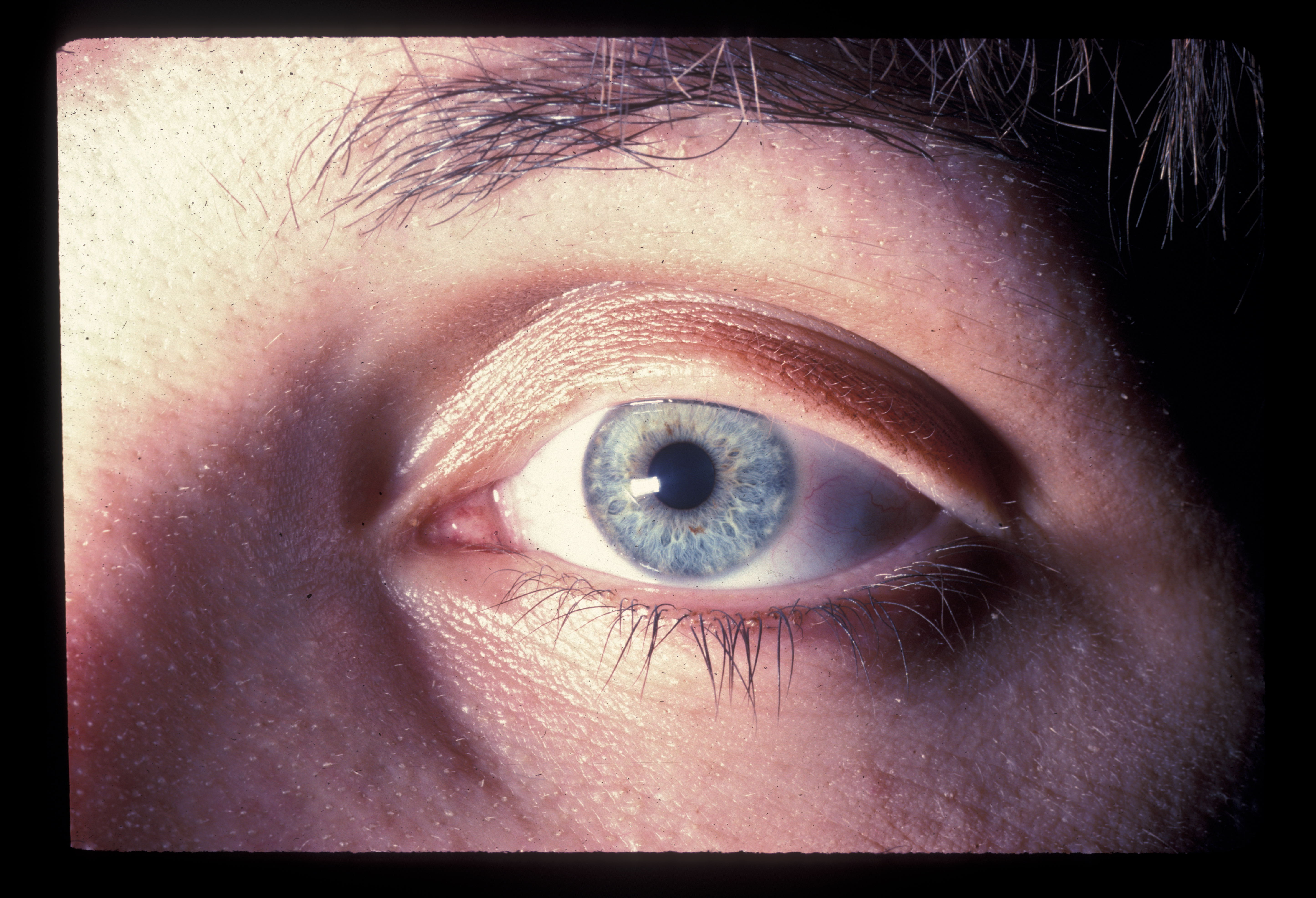
On physical examination, there is a near-complete loss of scalp hair, with preservation of hair elsewhere, including the eyelashes, eyebrows, axilla, and pubic area (see Image. Alopecia Areata With Eyelash Loss). On the scalp, follicular orifices should be visible, in contrast to scarring alopecia, where they are absent. There should be minimal scalp erythema or scale. At the periphery of affected areas, exclamation point hairs may be seen. These are thick at the apex of the hair shaft and thinner towards the base of the shaft. In previously affected areas where spontaneous regrowth has occurred, the hair may initially be nonpigmented, but repigmentation usually occurs in weeks to months.
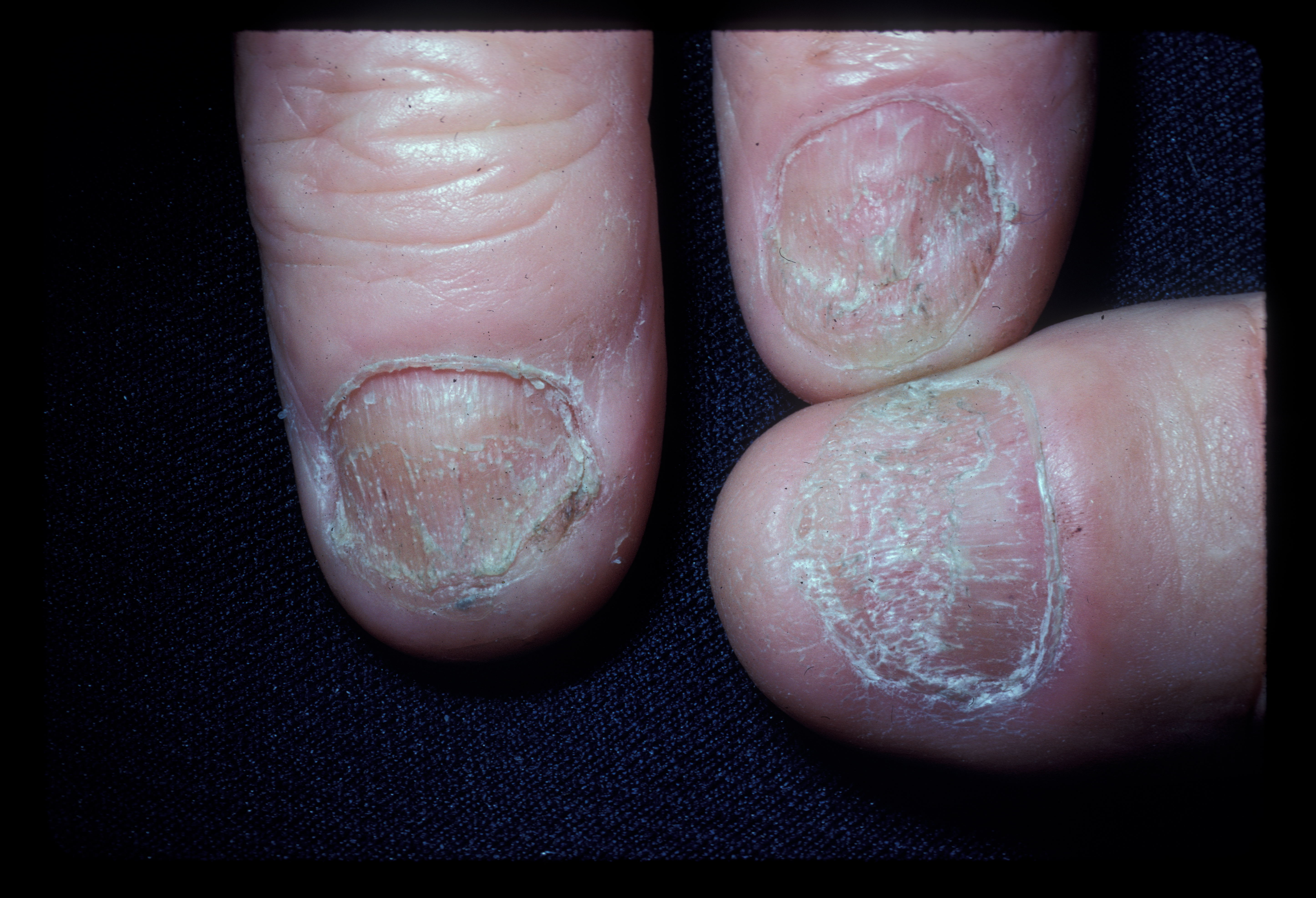
The nails should be closely evaluated as nail involvement is common in alopecia areata. This is even more frequent in patients with AT than AF and is seen in about 20%.[4] Nail pitting is the most common abnormality. Brittle nails, trachyonychia, and onycholysis are also seen.[15] See Image. Alopecia Areata Dystrophic Nails.
Evaluation
The history and physical examination are often sufficient for making the diagnosis. In most cases, the patient will have a history of patchy hair loss, which has progressed to complete loss of hair on the scalp, with the physical exam findings described above. Dermatoscopy can aid in examination. Trichoscopy, a term for dermoscopic evaluation of hair, reveals perifollicular yellow dots, black dots, tapering hairs, and vellus hairs.[16][17]
If the diagnosis remains unclear, a biopsy with histologic examination can be utilized. One or 2 4-mm punch biopsies of the scalp are often performed. The specimens may be sectioned horizontally or vertically, so some practitioners perform 2 sets of punch biopsies, one for vertical sectioning and another for horizontal sectioning, for a thorough evaluation of the follicles.[4][13] Sometimes a single bisected punch can have one half sectioned vertically and the other half horizontally, but then the sample size is smaller.
In certain patients, lab work may be indicated. Thyroid testing, such as thyroid-stimulating hormone (TSH) with reflex to free thyroxine (T4), is the most important screening laboratory test. Other tests sometimes ordered with alopecia, in general, include a complete blood count (CBC), vitamin and mineral levels, rapid plasma reagin (RPR), plasma testosterone, dehydroepiandrosterone sulfate (DHEA-S), and antinuclear antibody (ANA).
Treatment / Management
There are numerous therapies available for AA, including intralesional, topical, and systemic modalities. Due to the extent of involvement, intralesional administration is often impractical in AT, and treatment is initiated with topical or systemic agents. There are many more studies on the treatment of AF than studies specifically evaluating the treatment of AT. The most efficacious treatments were found to be topical immunotherapy, phototherapy, pulsed systemic steroids, and immunosuppressive agents. There have also been successful case reports of JAK inhibitors and the combination of simvastatin and ezetimibe, though clinical trials with outcomes for patients with AT are lacking.[18]
Topical Steroids
Topical steroids are an initial conservative treatment for AT. These are most often utilized in children under 10 years old presenting with extensive disease. Therapy with a super potent topical steroid such as clobetasol 0.05% solution or foam is recommended. Children with a history of hair loss for less than one year have shown better responses than those with longer disease durations.[12]
Topical Immunotherapy
Topical immunotherapy can be used in extensive AA and is performed with diphenylcyclopropenone (DPCP), also known as diphencyprone, or squaric acid dibutyl ester (SADBE). Dinitrochlorobenzene (DNCB) has also been used, but has largely been abandoned because of mutagenicity by the Ames test.[19] These agents cause mild allergic contact dermatitis of the scalp and work through an incompletely understood mechanism. Some authorities prefer to try DPCP first; if the patient fails treatment, SADBE may be tried. These treatments are administered regularly, usually weekly. The most common side effects are localized irritation, itching, or erythema.[18] Upon discontinuation, rates of relapse are lower than those for many other treatment modalities.
Systemic Steroids
Systemic steroids are another frequently used initial therapy to stimulate hair regrowth with good results. Monthly intramuscular triamcinolone injections have shown the most success, followed by pulsatile oral prednisone or methylprednisolone.[18] A variety of pulsed oral steroid regimens have been studied, without a significant difference in results. These vary from 2 consecutive daily pulses every 3 weeks to every 3 months. Adverse effects of systemic steroid treatment include weight gain, acne, dysmenorrhea, and moon facies. Relapse after discontinuation is common.
Immunosuppressive Agents
Immunosuppressive agents are often used second-line after a patient has failed standard therapy. These include cyclosporine, methotrexate, or azathioprine, alone or in combination with systemic steroids. Methotrexate can be used in both children and adults and is dosed weekly, usually at 15 to 25mg. Cyclosporine may be used as monotherapy in adults and is usually dosed at 3 to 5 mg/kg/d. Treatment duration should be limited to 6 to 12 months.[12][20]
JAK Inhibitors
JAK inhibitors are newer medications that are currently being studied in alopecia areata and totalis. These include tofacitinib, ruxolitinib, and baricitinib. These agents have promising therapeutic benefits due to our current understanding of the pathogenesis of AA. JAK inhibitors have proven efficacious in treating other inflammatory conditions such as rheumatoid arthritis and psoriasis. There are a few open-label trials for AA patients, which have shown impressive results with significant hair regrowth. However, many patients have a recurrence of hair loss after therapy discontinuation. Side effects of oral JAK inhibitors include viral reactivation, infection, transaminitis, and bone marrow suppression.[12] JAK inhibitors have also been used topically.[18] Topical 2% tofacitinib has been reported to have good results, though relapse upon discontinuation is also high.[21]
Phototherapy and Photodynamic Therapy
Phototherapy with psoralen plus ultraviolet A (PUVA) and photodynamic therapy (PDT) have been used in cases of extensive AA with reported success. PUVA involves oral or topical administration of psoralen, a photosensitizing agent, followed by UVA light exposure. The adverse effects of PUVA include cutaneous malignancy, so this therapy should be utilized cautiously. PDT is a similar therapy with the application of a photosensitizer, such as 5-aminolaevulinic acid (ALA), followed by light exposure. This therapy has mixed results, and clinical trials are lacking.[22][23]
Simvastatin
There are case reports of the use of daily simvastatin in combination with ezetimibe, with reported success, though no trials have been conducted.[18]
Nonmedical Interventions
In addition to medical intervention, products to conceal hair loss, such as wigs, hairpieces, and scalp prostheses, may be used instead of (or in conjunction with) other treatments. Topical and systemic therapies take time to achieve results, and wigs or prostheses provide immediate coverage. These can be purchased readymade or customized for the patient. There are options made with human hair and others with synthetic fibers. Due to the extensive nature of the disease and psychological impact, many patients prefer to keep their scalp covered while undergoing medical therapy.[12]
Differential Diagnosis
Some differentials for AT include:
- Telogen effluvium
- Anagen effluvium
- Androgenetic alopecia
Prognosis
AT generally has a worse prognosis than the more common patchy type of AA, with up to 23% of patients not responding to any therapy. In AT, the initial recovery rates are often higher than long-term rates due to the high rate of relapse.[4] Spontaneous recovery occurs in less than 10% of patients, and long-term complete recovery is about 8.5%.[24][25] Risk factors for a poor prognosis include a positive family history of AA and the baseline extent of AA.[4] There is a recently described AT subtype, acute diffuse and total alopecia (ADTA), which occurs in young adults and portends a more favorable prognosis.[24]
Complications
Psychological disorders such as anxiety, phobias, depression, and panic disorders are more common in patients with AA. There is a correlation between the extent of disease and the amount of psychological distress, so patients with AT are at higher risk for these comorbidities.[26]
Deterrence and Patient Education
Patients should be educated on the chronic nature of this disease. In addition to discussing therapeutic options, side effects, as well as the rate of relapse, should be discussed before initiating therapy. Patients should be counseled on the high degree of psychological distress that can accompany this condition and be referred to mental health providers when appropriate. Patients should be given information about organizations such as the National Alopecia Areata Foundation (NAAF), where they can learn more about this disease and connect with other affected individuals.
Pearls and Other Issues
Key facts to keep in mind regarding AT are as follows:
- Be sure to recognize and address psychosocial conditions that are often associated with AT. Referral to a support group may be helpful.
- Patients often prefer to try some treatment even though the overall complete recovery rate is only 8.5%. A discussion of realistic goals is helpful.
- Thyroid testing (such as a TSH with reflex to free T4) is the most important screening laboratory test. [27] Other tests sometimes ordered with alopecia, in general, include a CBC, vitamin and mineral levels, RPR, plasma testosterone, DHEA-S, and ANA.
- Telogen effluvium can resemble AT, but it has a more sudden onset and is self-limited. Also, there is no history of localized AF.
Enhancing Healthcare Team Outcomes
AT is best managed with an interprofessional team approach. AA may initially be encountered by the primary care provider. General practitioners need to recognize this condition and refer the patient to a dermatologist for consideration of treatment options. This is particularly imperative in AT, which portends a worse prognosis with a lower likelihood of spontaneous recovery. A referral to a therapist or psychiatrist may also be appropriate due to the high rates of psychological distress secondary to extensive hair loss. Lastly, the provider should be familiar with resources to provide the affected individual and family, as there are numerous associations and support groups that patients may find valuable.