Continuing Education Activity
Acoustic neuromas are tumors that develop from the sheath of Schwann cells. They tend to occupy the cerebellopontine angle and are usually found adjacent to the cochlear or vestibular nerve, either intracranially or extra-axially. This activity reviews the etiology, evaluation, and management of acoustic neuromas and highlights the collaboration among the interprofessional team in caring for patients with this condition.
Objectives:
- Identify the signs and symptoms of acoustic neuromas, such as gradual hearing loss, tinnitus, and balance problems.
- Assess the size, location, and growth rate of acoustic neuromas to determine the appropriate treatment approach.
- Utilize various treatment modalities, including surgical resection, stereotactic radiosurgery, or medical management, to develop individualized treatment plans for patients with acoustic neuromas.
- Collaborate with a multidisciplinary team, including neurosurgeons, radiation oncologists, audiologists, and rehabilitation specialists, to provide comprehensive care and optimize patient outcomes.
Introduction
Acoustic neuroma is also known by other names such as vestibular schwannoma (VS), acoustic neurinoma, vestibular neuroma, and acoustic neurofibroma. These tumors originate from the Schwann cells in the sheath surrounding the vestibular and cochlear nerves. Acoustic neuroma can occur either intracranially or extra-axially.
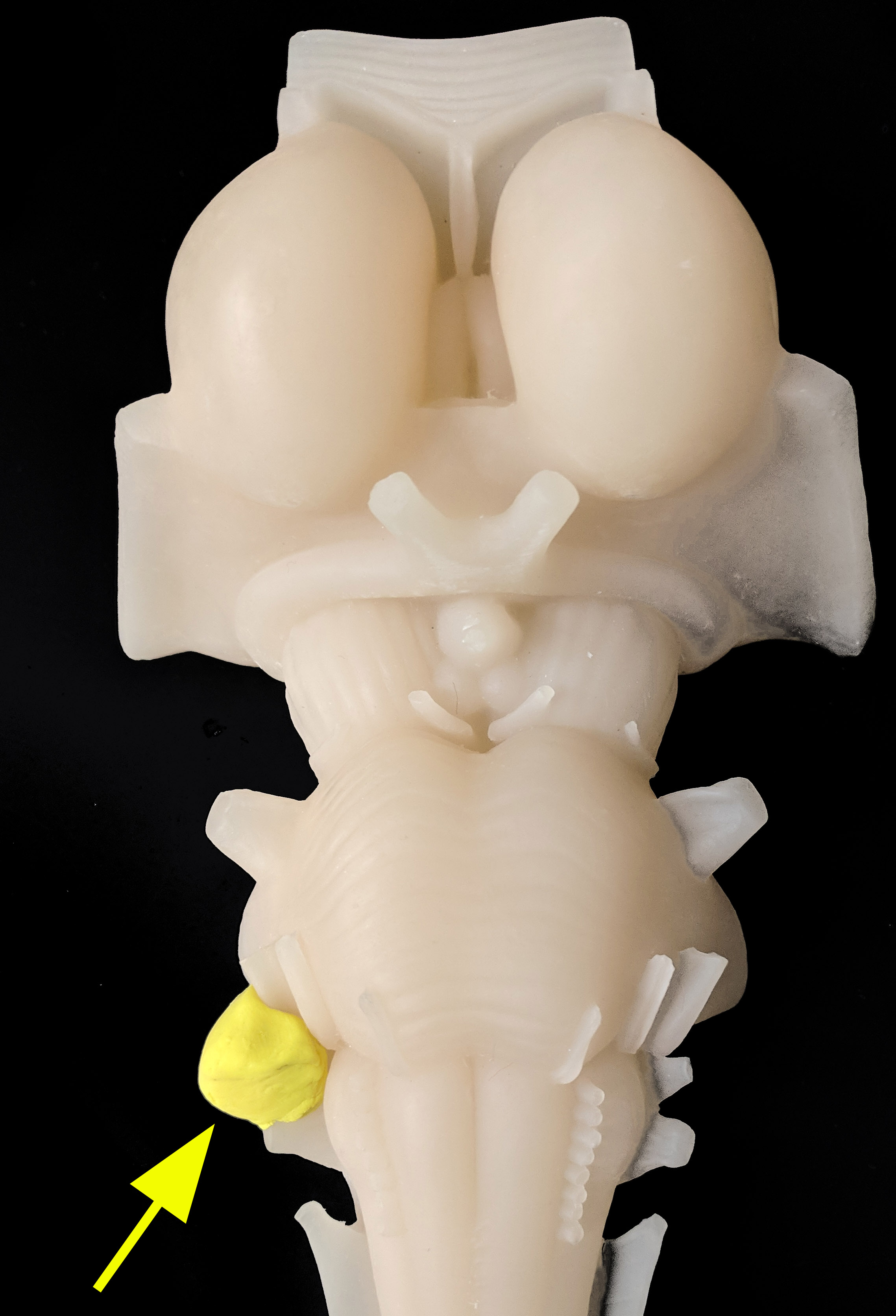
Acoustic neuromas are commonly located in the cerebellopontine angle, near the cochlear and vestibular nerves, particularly the inferior division of the vestibular nerve. About 5% to 10% of tumors in the cerebellopontine angle (CPA) are meningiomas, which may occur elsewhere in the brain. While most cases are sporadic, bilateral acoustic neuromas are typically associated with type 2 neurofibromatosis.[1][2][3]
Etiology
Bilateral acoustic neuromas can be associated with neurofibromatosis type 2, which is caused by a defect on chromosome 22q12.2 at the location of the neurofibromin 2 gene, responsible for encoding the merlin protein. Studies have indicated a predisposing mutation for the development of acoustic neuroma. Additionally, radiation exposure may increase the likelihood of developing this condition.[4]
While there have been concerns regarding mobile phone radiation, several studies have failed to establish a direct causal relationship between mobile phone radiation and the development of vestibular schwannomas.[5]
Epidemiology
Schwannomas account for approximately 8% of all clinically manifested intracranial tumors. Most acoustic neuromas are unilateral and occur sporadically. However, genetic factors contribute to developing bilateral acoustic neuromas, constituting less than 5% of all schwannomas.
Generally, acoustic neuromas are diagnosed between the fourth to sixth decades of life. However, individuals with neurofibromatosis type 2 (NF II) tend to present earlier, with the peak incidence occurring in the third decade of life. Although rare, acoustic schwannomas can occur in children.
There is a slight female preponderance, and symptoms can worsen during pregnancy. The hereditary form of acoustic neuroma is more commonly associated with NF II than neurofibromatosis type 1 (NF I), despite NFI being more prevalent. NF I is exclusively linked to unilateral acoustic neuroma in 24% of cases, while bilateral acoustic schwannoma is a hallmark feature of NF II. Both NF I and NF II are autosomal dominant, with genetic defects localized to chromosome 17 and chromosome 22, respectively.[6][7]
Histopathology
Acoustic neuromas originate near the porus acusticus, where the transition between glial and Schwann cells, known as the Obersteiner-Redlich zone, can be observed.
On gross examination, acoustic neuromas typically exhibit a rubbery-firm consistency and appear pale with a gray color. They may display varying degrees of vascularity and possess a well-defined capsule. The presence of the tumor can lead to the displacement and stretching of nerve fibers, which may result in discernible changes to the capsule's appearance.
When an acoustic neuroma is sectioned, the cut surface reveals a pale gray and firm appearance with a finely trabeculated pattern. Evidence of cystic degeneration, hemorrhage, xanthomatous changes, and calcification points may be present in larger tumors. These changes contribute to a variegated appearance in terms of consistency and color of the giant tumors. The blood supply to the tumor primarily originates from the internal auditory artery, which branches into several tiny vessels on the tumor's surface. In the case of larger tumors, there may be blood supply from small branches of neighboring cerebellar and pontine arteries.
On light microscopy examination, the tumor reveals a cellular composition of spindle cells with elongated nuclei and fibrillary cytoplasm. Those cells are arranged in two ways: Antoni A and Antoni B.
Antoni A tissue is characterized by a compact and organized structure consisting of an interwoven arrangement of elongated bipolar cells. The nuclei and fibers in Antoni A tissue may demonstrate a distinctive pattern, occasionally forming a spiral framework that resembles the appearance observed in meningiomas.
Antoni B, commonly observed in large acoustic neuromas, is characterized by a less organized arrangement of cells. In Antoni B tissue, cells are randomly dispersed and often clustered around areas of cystic changes, necrosis, old hemorrhage, and blood vessels. In addition, this tissue type has varying amounts of lymphocytic infiltration. The Antoni B tissue type, generally seen in larger tumors, is thought to be the outcome of ischemia.
The relative proportions of Antoni A and Antoni B tissue types determine the consistency of the acoustic neuroma. Although these changes are benign and malignant transformation is rare, nuclear pleomorphism is a typical feature of schwannomas. Mitotic figures, which are indicative of cell division, are relatively rare. Necrosis, if present, is attributed to poor blood supply rather than rapid tumor growth.
Edema, formation of micro or macrocysts, xanthomatous alteration, and areas of calcification are considered degenerative changes within the tumor tissue.
In electron microscopy (EM), the characteristic basement membrane of Schwann cells can be observed. Additionally, wide-spaced collagen is often present, indicating abnormalities in collagen fiber arrangement.[8]
History and Physical
The signs and symptoms of acoustic neuroma are primarily caused by the compression of the surrounding structures, including cranial nerve VIII (the vestibulocochlear nerve), adjacent cranial nerves, cerebellum, brainstem, and the resulting increase in intracranial pressure (ICP). Most individuals with acoustic neuromas present unilateral hearing loss due to cochlear nerve interruption or impaired blood supply to the nerve. Other clinical features include tinnitus, decreased word understanding, vertigo, headaches, and numbness. As the acoustic neuroma grows, the mass within the CPA will eventually compress the brainstem. This compression of the brainstem can result in gait abnormalities. The following is a summary of the clinical features of acoustic neuroma.
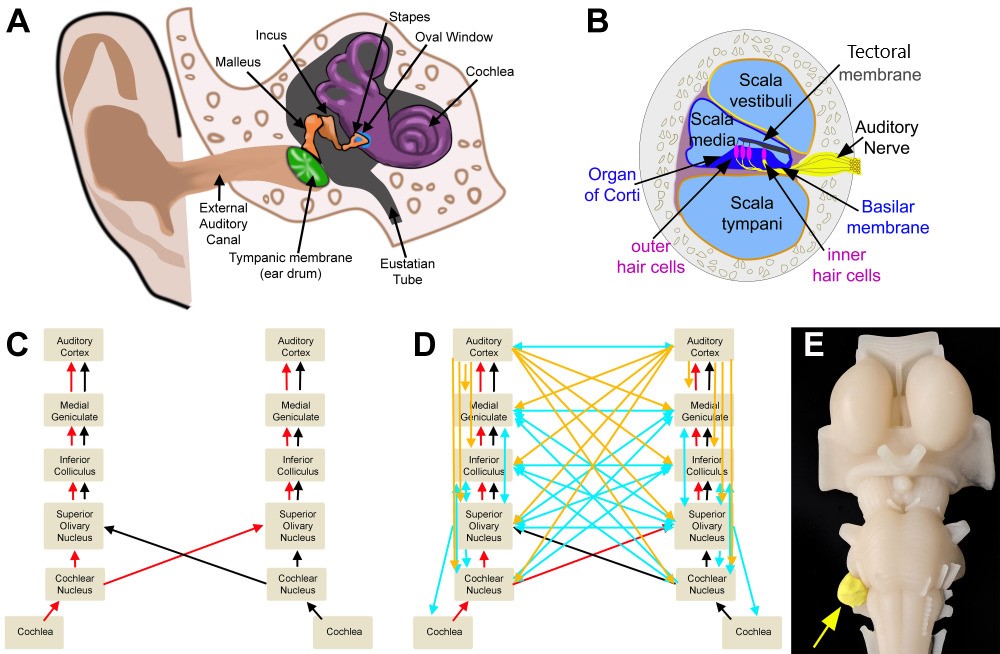
Involvement of Cranial Nerve VIII
- Auditory
- Hearing impairment is the most common and earliest symptom of acoustic neuroma. The impairment typically presents as slowly progressive, high-frequency retro-cochlear sensorineural hearing loss. Due to its gradual onset, the hearing loss may go unnoticed or be attributed to other factors. Several tests can be conducted to assess hearing impairment in the physical examination. These tests include the following:
- Speech discrimination tests evaluate the ability to understand speech.
- The use of tuning forks with wide-range frequencies
- Weber test can help determine if hearing loss is unilateral or bilateral.
- Rinne test can assess sound conduction through bone and air.
- Tinnitus refers to the perception of ringing or buzzing sounds in the ears and is another common symptom associated with acoustic neuroma. Tinnitus can manifest as a constant or intermittent sensation.
- Vestibular symptoms are related to the involvement of the vestibular portion of the eighth cranial nerve (vestibulocochlear nerve) and can include instability while moving the head and nystagmus.
Compression of Other Cranial Nerves
- Facial nerve (cranial nerve VII) symptoms are often minimal or subtle during the early stages, leading to late presentation, except in the case of very large tumors. Depending on the degree of nerve engagement, the symptoms may include twitching, increased lacrimation, and facial weakness.
- Trigeminal Nerve (cranial nerve V) symptoms include paraesthesia in the trigeminal nerve distribution, tongue-tingling, and corneal reflex impairment. Less commonly, patients may experience pain that may mimic typical trigeminal neuralgia.
- The glossopharyngeal (cranial nerve IX) and vagus nerves (cranial nerve X) involvement can result in palatal paresis, hoarseness of voice, and dysphagia.
Cerebellar Compression
- Cerebellar compression can occur in cases where there are large acoustic tumors.
- Symptoms include unsteady gait, difficulties with coordination and balance, tremors, and problems with fine motor skills. Rarely, dysarthria may also occur.
Brainstem Compression/Torsion
- Symptoms include pyramidal weakness, contralateral cranial nerve involvement, and nystagmus.
Raised ICP
- Raised ICP features include but are not limited to headache, nausea, vomiting, increased blood pressure, decreased mental abilities, confusion, disturbance of level of consciousness, and papilledema.
Evaluation
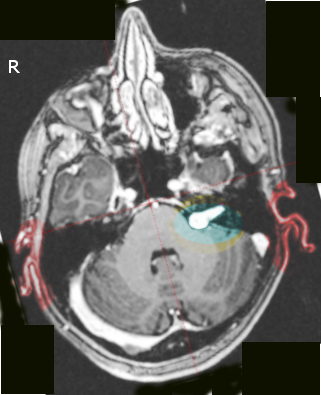
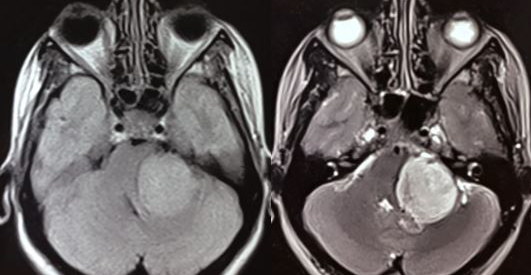
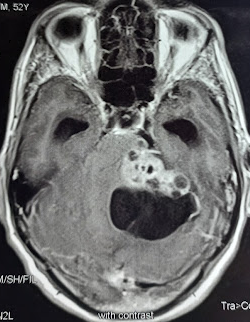
An acoustic neuroma is diagnosed using contrast-enhanced magnetic resonance imaging (MRI) or computed tomography scans. Contrast agents, such as gadolinium, enhance the visualization of tumors and obtain information about the tumor's characteristics and relationship to surrounding structures. Using contrast in these studies is also essential to view smaller tumors. If a patient has a hearing impairment, audiometric tests are often necessary to assess their hearing abilities. While auditory brainstem evoked response is a useful test, it is not commonly used as a primary screening tool for acoustic tumors, as it cannot detect small malignancies reliably.[9][10][11]
An intracanalicular component in the porus acusticus can cause a characteristic finding known as the "trumpeted internal acoustic meatus sign," observed as a widening or enlargement of the opening. Alternatively, when the tumor extends into the extrameatal space, it can produce a distinct appearance known as the "ice cream cone" sign.
Most acoustic neuromas appear hypo to isointense on T1-weighted MRI images and demonstrate heterogeneously hyperintensity on T2-weighted images. Furthermore, these tumors often exhibit significant contrast enhancement when administering a contrast agent, such as gadolinium.
Fast spin-echo MRI may be helpful as a screening test for acoustic neuromas due to its low cost and noninvasiveness.
Treatment / Management
Acoustic neuroma can be treated in several ways. The treatment options may include the following:
- Observation: If the tumor is small and not causing significant symptoms, monitoring the tumor's growth over time is the ideal management. This approach is also considered for older patients with numerous comorbidities.
- Stereotactic radiotherapy: This treatment involves using high-energy radiation to destroy the tumor cells. Gamma Knife, or Cyberknife, is a precise radiation therapy commonly used.
- Surgery: Surgical tumor removal may be necessary for larger tumors or those causing symptoms.
The main surgical approaches for treating acoustic neuroma include the following:
1. Retrosigmoid approach: This involves removing the tumor through an opening at the skull base. This approach provides good access to this region's tumor and cranial nerves. A tumor of any size can be approached while potentially preserving hearing.
2. Middle cranial fossa approach: In this approach, the surgeon gains access to the tumor through the middle cranial fossa of the skull base. This approach is most suitable for tumors with dominant intracanalicular and small cisternal components. Hearing is preserved in most cases. The main disadvantage is the need for temporal lobe retraction, which may produce postoperative seizures and venous infarction if the vein of Labbe is damaged.
3. Translabyrinthine: This technique involves removing the tumor through the inner ear and is most commonly used in patients with large tumors and no serviceable hearing. The main advantage of this approach is that facial nerve can be exposed early and be protected. Also, the cerebellum does not need to be retracted. But the access to the contents of the jugular foramen and lower parts of the foramen magnum is limited.
After surgery, an MRI is required within 6 to 12 months to document the extent of tumor removal and establish a baseline image. The most common complications of surgery include injury to the anterior inferior cerebral artery, hemorrhage, cerebellar trauma, facial paralysis, hearing loss (the most common), and hydrocephalus.
Differential Diagnosis
Acoustic neuromas account for around 80% to 90% of CPA lesions. Differential diagnoses of an acoustic neuroma include the following:
- Meningioma (5%-10% of CPA lesions)
- Ectodermal inclusion tumors
- Epidermoid (5%-7% of CPA lesions)
- Dermoid
- Metastases
- Neuroma from cranial nerves other than cranial nerve VIII:
- Trigeminal neuroma.
- Facial nerve neuroma
- Neurinoma of the lowest 4 cranial nerves (IX, X, XI, XII)
- Extensions of nearby lesions in the CPA:
- Dolichobasilar ectasia
- Aneurysm
- Cholesterol granuloma (distinct from epidermoid)
- Neurenteric cyst
- Arachnoid cyst
- Brainstem or cerebellar glioma
- Pituitary adenoma
- Craniopharyngioma
- Chordoma and tumors of the skull base
- Fourth ventricle tumors (ependymoma, medulloblastoma)
- Choroids plexus papilloma from the fourth ventricle through foramen of Luschka
- Glomus jugulare tumor
- Tumors of the temporal bone
Complications
Most complications are related to the surgical procedure and include the following:
- Injury to the anterior or posterior inferior cerebellar arteries
- Neurological injury
- Brain herniation
- Brain hemorrhage
- Injury to the cerebellum
- Facial paralysis
- Hearing loss
After surgery, tinnitus is a common problem in at least 10% to 20% of patients. The recurrence rate after excision is less than 5%. Facial nerve paralysis has been shown to occur in about 15% to 30% of patients, but most make a complete recovery over time. Hearing loss occurs in more than 50% of patients and may not improve. Residual hearing loss has a significant impact on the quality of life.[12][13][14]
Postoperative and Rehabilitation Care
Rehabilitation
Surgical and nonsurgical candidates often have residual vertigo and disequilibrium, which can be addressed through exercises aimed at adaptation, substitution, and habituation exercises.[15][16][17] These exercises include but are not limited to the following:
- Adaptation: gaze stability exercises
- Habituation: repeated movement exercises based on symptom-provoking motions
- Substitution: visual control or active eye-head movements between targets
Following surgery for an acoustic neuroma, patients may experience side effects like facial palsy. However, rehabilitation approaches are available to address these issues and help patients regain function. Two such techniques that can be employed are mirror therapy and motor imagery.[18]
Vestibular rehabilitation programs should be catered to the specific deficits each patient presents with, as it has been demonstrated that a targeted approach is more beneficial for these patients than a general vestibular program.[19]
Pearls and Other Issues
Detecting and diagnosing acoustic neuroma early can improve treatment outcomes and preserve important functions like hearing and facial nerve function. Treatment options, including observation, surgical removal, and radiation therapy, may vary. A multidisciplinary team of neurosurgeons, otolaryngologists, radiation oncologists, and rehabilitation specialists can provide comprehensive care and optimize treatment strategies.
Recurrence is uncommon after surgical removal of an acoustic neuroma; however, it is worth noting that tinnitus may worsen in some patients post-surgery. Hearing loss and facial paralysis may improve over time for many individuals. The preoperative distinction between a meningioma and an acoustic schwannoma is essential for technical and prognostic reasons.
Enhancing Healthcare Team Outcomes
Acoustic neuromas are not common tumors in clinical practice. However, they often present with unilateral hearing loss. Thus healthcare workers should consider the lesion in the differential diagnosis. Once diagnosed, the tumor is best managed by an interprofessional team that may include neurosurgery, otolaryngology (ENT), hearing and rehabilitation specialist, and radiation oncologist.
After surgery, tinnitus is a common problem in at least 10% to 20% of patients. The recurrence rate after excision is less than 5%. Facial nerve paralysis has been shown to occur in about 15% to 30% of patients, but most make a complete recovery over time. Hearing loss occurs in more than 50% of patients and may not improve. Residual hearing loss has a significant impact on the quality of life.[12][13][14]