Introduction
The foramen magnum is of interest for various scientific disciplines: forensic and physical anthropology; comparative anatomy; biology; surgery of the foramen structures and for the craniovertebral junction. There are morphological differences between men and women. The sagittal diameter is greater in the male, as is the transverse diameter.
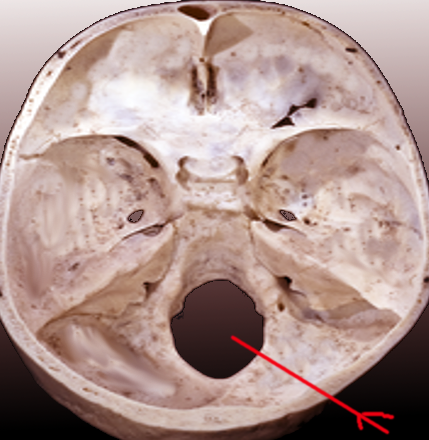
The foramen magnum is the largest foramen of the skull. It is located in the most inferior portion of the cranial fossa as a part of the occipital bone. Its contents include the medulla oblongata, meninges, spinal root of cranial nerve XI, vertebral arteries, anterior and posterior spinal arteries, the tectorial membrane, and alar ligaments.
On the foramen magnum, there are two craniometric points: the basion, the median point of the front edge of the hole, and the opisthion, posterior correspondence.
Structure and Function
The foramen magnum functions as a passage of the central nervous system through the skull connecting the brain with the spinal cord. On either side of the foramen magnum is an occipital condyle. These condyles form joints with the first cervical vertebra.
The position of the foramen is essential for posture in orthostatism, as it allows a correct relationship between the skull and the cervical spine.In fossil hominins, the position of the foramen is indicative of the bipedalism of our ancestors.
Embryology
All the bones of the skull develop from paraxial mesoderm and neural crest cells. During gastrulation in the third week, mesenchymal cells migrate through the primitive streak to form the mesoderm. At the end of the third week, the paraxial mesoderm is segmented into somites that differentiate to become sclerotomes.[1] During the fourth week, the occipital sclerotomes concentrate around the notochord under the influence of signals from the rhombencephalon.
These sclerotomes derive membrane and cartilage, the developmental origin of the occipital bone. Four primary cartilaginous centers, the anterior basioccipital (basilar), the lateral exoccipitals (condylars), and the posterior supraoccipital (squamous), in addition to a fifth membranous element, referred to as the interparietal combine around the foramen magnum to form the occipital bone. The mendosal suture runs horizontally between the developing inferior supraoccipital and superior interparietal bones.[2]
Fetuses at 9 weeks gestation have an ossification center around the hypoglossal canal in each exoccipital part and a single median ossification center in the basioccipital cartilage.[3] At 12 weeks gestation, a pair of ossification centers in the supraoccipital cartilage fuse together to form the supraoccipital bone. Rostral to the supraoccipital bone, the second pair of ossification centers in the membranous portion fuse to form the interparietal bone.[4] The intraparietal portion ossifies intramembranous while the rest of the occipital bone ossifies endochondrally utilizing cartilage as a precursor.[1] The supraoccipital and interparietal bones then fuse midline, but at this point in development are still separated laterally by the mendosal suture. At 14 weeks, ossification of the basioccipital occurs and advances laterally into the ventral portion of the condylars, while concurrently the ventral portions advance into the dorsal portions. Also, during the 14th week, the fusion of the supraoccipital and interparietal bones progress almost to completion. The full union of these segments does not occur until between 2 and 4 years of age.[3] By the 16th week of fetal development, all intramembranous ossification centers are generally fused forming a lattice of trabeculae overlaying the external surface of the occipital squama.[4] The exoccipitals remain separated from both the basioccipital and the supraoccipital segments by synchondroses and will not fuse until between 2 and 4 years of age.[3]
Blood Supply and Lymphatics
The vertebral arteries and the anterior and posterior spinal arteries traverse the foramen magnum. The vertebral artery branches off of the subclavian artery and has four segments. The first preforaminal segment (V1) constitutes the portion from the subclavian to the transverse foramen of C6. The second foraminal segment (V2) travels through the transverse foramen of C6 through C2. When the artery emerges from the foramen of C2, it is considered V3. This third segment, the extradural segment, continues through the transverse foramen of C1, the suboccipital triangle, and the foramen magnum. The point at which the artery penetrates the dura and arachnoid mater indicates the beginning of the fourth and final segment of the vertebral artery, the intradural segment. The right and left fourth segments of the vertebral artery combine to form the basilar artery at the level of the pons.[5]
In addition to the basilar artery, the fourth segment of the vertebral arteries gives rise to the anterior spinal artery, the posterior spinal artery, perforating branches to the medulla, and the posterior inferior cerebellar artery. The anterior spinal artery feeds the upper cervical spinal cord and inferior medulla while the posterior spinal artery feeds the dorsal spinal cord and conus medullaris. The lateral medulla, cerebellar tonsils, inferior vermis, and choroid plexus depend on the posterior inferior cerebellar arteries, and the penetrating branches supply portions of the medulla, the olives, and the inferior cerebellar peduncle.
Nerves
Cranial nerve XI, or the accessory nerve, originates from the upper spinal cord and medulla and enters the skull through the foramen magnum. This nerve innervates the sternocleidomastoid and trapezius muscles and provides motor function.
Muscles
The sternocleidomastoid muscle acts to flex and extend the neck and turn the head to the contralateral side. The trapezius muscle shrugs the shoulders.
Physiologic Variants
There are a few significant anatomical measurements concerning the foramen magnum: the transverse diameter, the anteroposterior diameter, and the foramen magnum index calculated by dividing the anteroposterior diameter by the transverse diameter. These measurements exhibit physiologic variance between skulls.[6] For example, a foramen magnum index greater than 1.2 is considered an ovoid variant.[7] Other named shapes of the foramen magnum include rhomboid, circle, heart, pear, and hexagon. Specific names are inconsistent between studies.[6] Additionally, the foramen may be asymmetrical. The protrusion of the occipital condyles into the foramen magnum demonstrates another source of anatomical variance. The hypocondylar arch is a feature of embryologic skulls that normally regresses at birth but rarely is maintained.[7]
Surgical Considerations
The anatomical variance of the foramen magnum may have some impact on certain surgical procedures such as vertebral artery and posterior inferior cerebellar artery aneurysm repairs, foramen magnum meningioma resections, and foramen magnum decompression among others. In an ovoid type foramen magnum, it is difficult for a surgeon to adequately expose the anterior portion of the foramen. Additionally, the occipital condyle and jugular tubercle are the main bony prominence obstructing the anterolateral portion of the foramen magnum. Extension of the occipital condyles into the foramen magnum may indicate the need for more extensive bony removal in certain procedures.[7]
Clinical Significance
There are a number of pathological states directly associated with the foramen magnum. Elevated intracranial pressure (greater than 20 mm Hg) often results from sources of edema such as stroke, trauma, mass effect, and infection and represents a serious prognosis. As the total volume in the skull is fixed and consists of blood, cerebrospinal fluid (CSF), and brain tissue, the increased pressure may lead to compensation via cerebellar tonsil herniation through the foramen magnum. The clinical picture of any herniation includes hypertension, bradycardia, and respiratory depression, but cerebellar herniation specifically causes compression of the respiratory centers of the medulla and may be fatal. The anatomy of the foramen magnum may influence the displacement of the herniated tissues.[6]
In another subset of clinical associations with this anatomic structure, patients with Chiari malformations have statistically larger foramen magnums compared to the normal population. Chiari I is the herniation of the cerebellar tonsils (a single tonsil 5 mm or both tonsils 3 mm) through the foramen magnum. While some patients are asymptomatic, there is a wide range of severity of clinical symptoms. The mildest being infrequent exertional headaches. Severe cases present with significant myelopathy and brainstem compromise. Common complications include syringomyelia and hydrocephalus. In babies, there are associated sleep apnea and feeding difficulties.[8]
Chiari II is the inferior displacement of the vermis, cerebellar tonsils, medulla, and fourth ventricle through the foramen magnum. Clinical presentation is more severe in Chiari II than Chiari I and is strongly associated with lumbar myelomeningocele and supratentorial anomalies such as corpus callosal dysgenesis, heterotopias, and sulcation abnormalities. Approximately 80% to 90% of children with Chiari II malformations present with hydrocephalus secondary to fourth ventricle obstruction requiring shunt placement. Patients with Chiari III malformations present with encephalocele in addition to the abnormalities of Chiari II.[8]
A mass represents another form of compression adjacent to the foramen magnum. Meningiomas are a benign slow-growing central nervous system tumors of the arachnoid cells of the dura mater most often arising in the basal region of the cerebrum. Rarely they occur in the foramen magnum presenting with posterior headache, paresthesias, and motor deficits. The overall symptomology may be varied due to the anatomic proximity to the cerebellar tonsils, caudal medulla, lower cranial nerves, rostral spinal cord, and upper cervical nerves.[9]
A complication of chronic rheumatoid arthritis demonstrates another example of pathology associated with the foramen magnum. Rheumatoid arthritis-associated C1-C2 facet erosion and laxity of ligamentous restrains results in vertical atlantoaxial subluxation. This defect may further lead to a protrusion of the odontoid through the foramen magnum and compression of the midbrain. Other etiologies of atlantoaxial subluxation include trauma and congenital conditions such as Down syndrome. Clinical presentation varies widely as some patients experience few symptoms, but others experience significant instability and neurologic compromise.[10]
The occipital condyles comprise the osseous lateral boundaries of the foramen magnum. In patients presenting with high-energy trauma mechanisms of injury or high impact axial loads during various sporting events (i.e. American football, mountain climbing, extreme sports events), due diligence to rule out injury to the occiput-cervical junction, as well as the rest of the cervical spine is warranted via accurate history taking, physical examination, and appropriate imaging. At advanced (i.e. Level I) and regional trauma centers, computed tomogram imaging is performed as part of the advanced imaging protocol. It is important to recognize that even displaced occipital condyle fractures can be missed on initial radiographs. In the appropriate clinical scenario, consideration should be given to transferring the patient complaining of persistent neck pain even in the absence of neurologic symptoms upon presentation.[11]