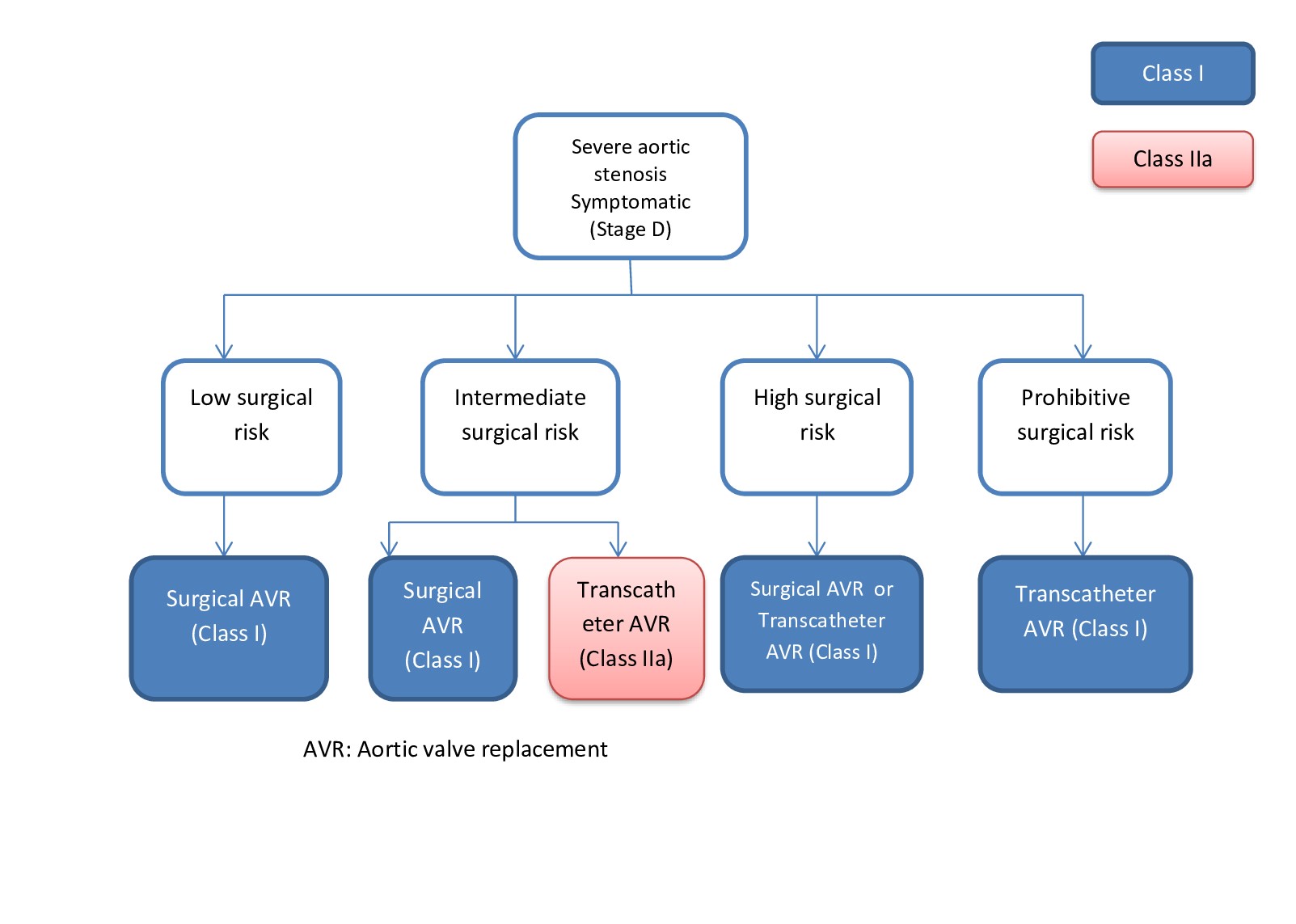
[1]
El-Hamamsy I, Balachandran K, Yacoub MH, Stevens LM, Sarathchandra P, Taylor PM, Yoganathan AP, Chester AH. Endothelium-dependent regulation of the mechanical properties of aortic valve cusps. Journal of the American College of Cardiology. 2009 Apr 21:53(16):1448-55. doi: 10.1016/j.jacc.2008.11.056. Epub
[PubMed PMID: 19371829]
[2]
Rozeik M, Wheatley D, Gourlay T. The aortic valve: structure, complications and implications for transcatheter aortic valve replacement. Perfusion. 2014 Jul:29(4):285-300
[PubMed PMID: 24570076]
[3]
Fedak PW, Verma S, David TE, Leask RL, Weisel RD, Butany J. Clinical and pathophysiological implications of a bicuspid aortic valve. Circulation. 2002 Aug 20:106(8):900-4
[PubMed PMID: 12186790]
[4]
Iung B, Baron G, Butchart EG, Delahaye F, Gohlke-Bärwolf C, Levang OW, Tornos P, Vanoverschelde JL, Vermeer F, Boersma E, Ravaud P, Vahanian A. A prospective survey of patients with valvular heart disease in Europe: The Euro Heart Survey on Valvular Heart Disease. European heart journal. 2003 Jul:24(13):1231-43
[PubMed PMID: 12831818]
Level 3 (low-level) evidence
[5]
Freeman RV, Otto CM. Spectrum of calcific aortic valve disease: pathogenesis, disease progression, and treatment strategies. Circulation. 2005 Jun 21:111(24):3316-26
[PubMed PMID: 15967862]
[6]
Bonow RO, Carabello BA, Chatterjee K, de Leon AC Jr, Faxon DP, Freed MD, Gaasch WH, Lytle BW, Nishimura RA, O'Gara PT, O'Rourke RA, Otto CM, Shah PM, Shanewise JS, 2006 Writing Committee Members, American College of Cardiology/American Heart Association Task Force. 2008 Focused update incorporated into the ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1998 Guidelines for the Management of Patients With Valvular Heart Disease): endorsed by the Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation. 2008 Oct 7:118(15):e523-661. doi: 10.1161/CIRCULATIONAHA.108.190748. Epub 2008 Sep 26
[PubMed PMID: 18820172]
Level 1 (high-level) evidence
[7]
Varadarajan P,Kapoor N,Bansal RC,Pai RG, Survival in elderly patients with severe aortic stenosis is dramatically improved by aortic valve replacement: Results from a cohort of 277 patients aged > or =80 years. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2006 Nov;
[PubMed PMID: 16950629]
[8]
Iung B, Cachier A, Baron G, Messika-Zeitoun D, Delahaye F, Tornos P, Gohlke-Bärwolf C, Boersma E, Ravaud P, Vahanian A. Decision-making in elderly patients with severe aortic stenosis: why are so many denied surgery? European heart journal. 2005 Dec:26(24):2714-20
[PubMed PMID: 16141261]
[9]
Kvidal P, Bergström R, Malm T, Ståhle E. Long-term follow-up of morbidity and mortality after aortic valve replacement with a mechanical valve prosthesis. European heart journal. 2000 Jul:21(13):1099-111
[PubMed PMID: 10843828]
[10]
Sadee AS, Becker AE, Verheul HA, Bouma B, Hoedemaker G. Aortic valve regurgitation and the congenitally bicuspid aortic valve: a clinico-pathological correlation. British heart journal. 1992 Jun:67(6):439-41
[PubMed PMID: 1622690]
[11]
Wise JR Jr, Cleland WP, Hallidie-Smith KA, Bentall HH, Goodwin JF, Oakley CM. Urgent aortic-valve replacement for acute aortic regurgitation due to infective endocarditis. Lancet (London, England). 1971 Jul 17:2(7716):115-21
[PubMed PMID: 4104458]
[12]
Shibayama K, Watanabe H, Murai T, Sasaki S, Tabata M, Fukui T, Umemura J, Takanashi S, Sumiyoshi T. Aortic regurgitation caused by cusp bending of aortic valve leaflet. Journal of echocardiography. 2012 Mar:10(1):21-3. doi: 10.1007/s12574-011-0105-x. Epub 2012 Jan 5
[PubMed PMID: 27277925]
[13]
Kai H, Koyanagi S, Takeshita A. Aortic valve prolapse with aortic regurgitation assessed by Doppler color-flow echocardiography. American heart journal. 1992 Nov:124(5):1297-304
[PubMed PMID: 1442499]
[14]
Bonow RO, Carabello BA, Chatterjee K, de Leon AC Jr, Faxon DP, Freed MD, Gaasch WH, Lytle BW, Nishimura RA, O'Gara PT, O'Rourke RA, Otto CM, Shah PM, Shanewise JS, American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2008 focused update incorporated into the ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to revise the 1998 guidelines for the management of patients with valvular heart disease). Endorsed by the Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Journal of the American College of Cardiology. 2008 Sep 23:52(13):e1-142. doi: 10.1016/j.jacc.2008.05.007. Epub
[PubMed PMID: 18848134]
Level 1 (high-level) evidence
[15]
Ellis JT, Healy TM, Fontaine AA, Saxena R, Yoganathan AP. Velocity measurements and flow patterns within the hinge region of a Medtronic Parallel bileaflet mechanical valve with clear housing. The Journal of heart valve disease. 1996 Nov:5(6):591-9
[PubMed PMID: 8953436]
[16]
Nazarov VM, Zheleznev SI, Bogachev-Prokophiev AV, Afanasyev AV, Nemchenko EV, Jeltovskiy YV, Lavinyukov SO. CardiaMed mechanical valve: mid-term results of a multicenter clinical trial. Asian cardiovascular & thoracic annals. 2014 Jan:22(1):9-17. doi: 10.1177/0218492312464035. Epub 2013 Jul 9
[PubMed PMID: 24585637]
Level 1 (high-level) evidence
[17]
Ghanbari H, Viatge H, Kidane AG, Burriesci G, Tavakoli M, Seifalian AM. Polymeric heart valves: new materials, emerging hopes. Trends in biotechnology. 2009 Jun:27(6):359-67. doi: 10.1016/j.tibtech.2009.03.002. Epub 2009 May 4
[PubMed PMID: 19406497]
[18]
Wheatley DJ, Raco L, Bernacca GM, Sim I, Belcher PR, Boyd JS. Polyurethane: material for the next generation of heart valve prostheses? European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2000 Apr:17(4):440-8
[PubMed PMID: 10773568]
[19]
Vitale N, De Feo M, De Siena P, Cappabianca G, Onorati F, Gregorio R, Branzoli S, de Luca L, Schinosa T, Viganò M, Scardone M, Cotrufo M. Tilting-disc versus bileaflet mechanical prostheses in the aortic position: a multicenter evaluation. The Journal of heart valve disease. 2004 May:13 Suppl 1():S27-34
[PubMed PMID: 15224997]
[20]
STARR A, EDWARDS ML. Mitral replacement: clinical experience with a ball-valve prosthesis. Annals of surgery. 1961 Oct:154(4):726-40
[PubMed PMID: 13916361]
[21]
Chaikof EL. The development of prosthetic heart valves--lessons in form and function. The New England journal of medicine. 2007 Oct 4:357(14):1368-71
[PubMed PMID: 17914037]
[22]
Blot WJ, Ibrahim MA, Ivey TD, Acheson DE, Brookmeyer R, Weyman A, Defauw J, Smith JK, Harrison D. Twenty-five-year experience with the Björk-Shiley convexoconcave heart valve: a continuing clinical concern. Circulation. 2005 May 31:111(21):2850-7
[PubMed PMID: 15927993]
[23]
Zilla P, Brink J, Human P, Bezuidenhout D. Prosthetic heart valves: catering for the few. Biomaterials. 2008 Feb:29(4):385-406
[PubMed PMID: 17950840]
[24]
Braunwald NS. It will work: the first successful mitral valve replacement. The Annals of thoracic surgery. 1989 Sep:48(3 Suppl):S1-3
[PubMed PMID: 2673090]
[25]
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP 3rd, Guyton RA, O'Gara PT, Ruiz CE, Skubas NJ, Sorajja P, Sundt TM 3rd, Thomas JD, ACC/AHA Task Force Members. 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014 Jun 10:129(23):e521-643. doi: 10.1161/CIR.0000000000000031. Epub 2014 Mar 3
[PubMed PMID: 24589853]
Level 1 (high-level) evidence
[26]
Andersen HR, Knudsen LL, Hasenkam JM. Transluminal implantation of artificial heart valves. Description of a new expandable aortic valve and initial results with implantation by catheter technique in closed chest pigs. European heart journal. 1992 May:13(5):704-8
[PubMed PMID: 1618213]
[27]
Mack MJ, Brennan JM, Brindis R, Carroll J, Edwards F, Grover F, Shahian D, Tuzcu EM, Peterson ED, Rumsfeld JS, Hewitt K, Shewan C, Michaels J, Christensen B, Christian A, O'Brien S, Holmes D, STS/ACC TVT Registry. Outcomes following transcatheter aortic valve replacement in the United States. JAMA. 2013 Nov 20:310(19):2069-77. doi: 10.1001/jama.2013.282043. Epub
[PubMed PMID: 24240934]
[28]
Bloomfield GS, Gillam LD, Hahn RT, Kapadia S, Leipsic J, Lerakis S, Tuzcu M, Douglas PS. A practical guide to multimodality imaging of transcatheter aortic valve replacement. JACC. Cardiovascular imaging. 2012 Apr:5(4):441-55. doi: 10.1016/j.jcmg.2011.12.013. Epub
[PubMed PMID: 22498335]
[29]
Poulin F, Carasso S, Horlick EM, Rakowski H, Lim KD, Finn H, Feindel CM, Greutmann M, Osten MD, Cusimano RJ, Woo A. Recovery of left ventricular mechanics after transcatheter aortic valve implantation: effects of baseline ventricular function and postprocedural aortic regurgitation. Journal of the American Society of Echocardiography : official publication of the American Society of Echocardiography. 2014 Nov:27(11):1133-42. doi: 10.1016/j.echo.2014.07.001. Epub 2014 Aug 7
[PubMed PMID: 25125314]
[30]
Di Bello V, Giannini C, De Carlo M, Delle Donne MG, Nardi C, Palagi C, Cucco C, Dini FL, Guarracino F, Marzilli M, Petronio AS. Acute improvement in arterial-ventricular coupling after transcatheter aortic valve implantation (CoreValve) in patients with symptomatic aortic stenosis. The international journal of cardiovascular imaging. 2012 Jan:28(1):79-87. doi: 10.1007/s10554-010-9772-3. Epub 2011 Jan 8
[PubMed PMID: 21222040]
[31]
Ben-Dor I, Goldstein SA, Pichard AD, Satler LF, Maluenda G, Li Y, Syed AI, Gonzalez MA, Gaglia MA Jr, Wakabayashi K, Delhaye C, Belle L, Wang Z, Collins SD, Torguson R, Okubagzi P, Aderotoye A, Xue Z, Suddath WO, Kent KM, Epstein SE, Lindsay J, Waksman R. Clinical profile, prognostic implication, and response to treatment of pulmonary hypertension in patients with severe aortic stenosis. The American journal of cardiology. 2011 Apr 1:107(7):1046-51. doi: 10.1016/j.amjcard.2010.11.031. Epub 2011 Feb 4
[PubMed PMID: 21296325]
[32]
Kodali SK, O'Neill WW, Moses JW, Williams M, Smith CR, Tuzcu M, Svensson LG, Kapadia S, Hanzel G, Kirtane AJ, Leon MB. Early and late (one year) outcomes following transcatheter aortic valve implantation in patients with severe aortic stenosis (from the United States REVIVAL trial). The American journal of cardiology. 2011 Apr 1:107(7):1058-64. doi: 10.1016/j.amjcard.2010.11.034. Epub
[PubMed PMID: 21419887]
[33]
Yared K, Garcia-Camarero T, Fernandez-Friera L, Llano M, Durst R, Reddy AA, O'Neill WW, Picard MH. Impact of aortic regurgitation after transcatheter aortic valve implantation: results from the REVIVAL trial. JACC. Cardiovascular imaging. 2012 May:5(5):469-77. doi: 10.1016/j.jcmg.2012.02.008. Epub
[PubMed PMID: 22595154]
[34]
Toggweiler S, Gurvitch R, Leipsic J, Wood DA, Willson AB, Binder RK, Cheung A, Ye J, Webb JG. Percutaneous aortic valve replacement: vascular outcomes with a fully percutaneous procedure. Journal of the American College of Cardiology. 2012 Jan 10:59(2):113-8. doi: 10.1016/j.jacc.2011.08.069. Epub
[PubMed PMID: 22222073]
[35]
Pang PY, Chiam PT, Chua YL, Sin YK. A survivor of late prosthesis migration and rotation following percutaneous transcatheter aortic valve implantation. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery. 2012 May:41(5):1195-6. doi: 10.1093/ejcts/ezr195. Epub 2012 Jan 6
[PubMed PMID: 22228843]
[36]
Lauten A, Hamadanchi A, Doenst T, Figulla HR. Late migration of balloon-expandable transcatheter aortic valve. European heart journal. 2013 Aug:34(32):2509. doi: 10.1093/eurheartj/eht204. Epub 2013 Jun 11
[PubMed PMID: 23761393]
[37]
Tamburino C, Capodanno D, Ramondo A, Petronio AS, Ettori F, Santoro G, Klugmann S, Bedogni F, Maisano F, Marzocchi A, Poli A, Antoniucci D, Napodano M, De Carlo M, Fiorina C, Ussia GP. Incidence and predictors of early and late mortality after transcatheter aortic valve implantation in 663 patients with severe aortic stenosis. Circulation. 2011 Jan 25:123(3):299-308. doi: 10.1161/CIRCULATIONAHA.110.946533. Epub 2011 Jan 10
[PubMed PMID: 21220731]
[38]
Kodali S, Pibarot P, Douglas PS, Williams M, Xu K, Thourani V, Rihal CS, Zajarias A, Doshi D, Davidson M, Tuzcu EM, Stewart W, Weissman NJ, Svensson L, Greason K, Maniar H, Mack M, Anwaruddin S, Leon MB, Hahn RT. Paravalvular regurgitation after transcatheter aortic valve replacement with the Edwards sapien valve in the PARTNER trial: characterizing patients and impact on outcomes. European heart journal. 2015 Feb 14:36(7):449-56. doi: 10.1093/eurheartj/ehu384. Epub 2014 Oct 1
[PubMed PMID: 25273886]
[39]
Sinning JM, Vasa-Nicotera M, Chin D, Hammerstingl C, Ghanem A, Bence J, Kovac J, Grube E, Nickenig G, Werner N. Evaluation and management of paravalvular aortic regurgitation after transcatheter aortic valve replacement. Journal of the American College of Cardiology. 2013 Jul 2:62(1):11-20. doi: 10.1016/j.jacc.2013.02.088. Epub 2013 May 1
[PubMed PMID: 23644081]
[40]
Barbanti M, Yang TH, Rodès Cabau J, Tamburino C, Wood DA, Jilaihawi H, Blanke P, Makkar RR, Latib A, Colombo A, Tarantini G, Raju R, Binder RK, Nguyen G, Freeman M, Ribeiro HB, Kapadia S, Min J, Feuchtner G, Gurtvich R, Alqoofi F, Pelletier M, Ussia GP, Napodano M, de Brito FS Jr, Kodali S, Norgaard BL, Hansson NC, Pache G, Canovas SJ, Zhang H, Leon MB, Webb JG, Leipsic J. Anatomical and procedural features associated with aortic root rupture during balloon-expandable transcatheter aortic valve replacement. Circulation. 2013 Jul 16:128(3):244-53. doi: 10.1161/CIRCULATIONAHA.113.002947. Epub 2013 Jun 7
[PubMed PMID: 23748467]
[41]
Nombela-Franco L, Rodés-Cabau J, DeLarochellière R, Larose E, Doyle D, Villeneuve J, Bergeron S, Bernier M, Amat-Santos IJ, Mok M, Urena M, Rheault M, Dumesnil J, Côté M, Pibarot P, Dumont E. Predictive factors, efficacy, and safety of balloon post-dilation after transcatheter aortic valve implantation with a balloon-expandable valve. JACC. Cardiovascular interventions. 2012 May:5(5):499-512. doi: 10.1016/j.jcin.2012.02.010. Epub
[PubMed PMID: 22625188]
[42]
Beohar N, Kirtane AJ, Blackstone E, Waksman R, Holmes D Jr, Minha S, Alli O, Suri RM, Svensson LG, Leon M, Kodali S. Trends in Complications and Outcomes of Patients Undergoing Transfemoral Transcatheter Aortic Valve Replacement: Experience From the PARTNER Continued Access Registry. JACC. Cardiovascular interventions. 2016 Feb 22:9(4):355-363. doi: 10.1016/j.jcin.2015.10.050. Epub 2016 Jan 20
[PubMed PMID: 26803420]
[43]
Dvir D, Leipsic J, Blanke P, Ribeiro HB, Kornowski R, Pichard A, Rodés-Cabau J, Wood DA, Stub D, Ben-Dor I, Maluenda G, Makkar RR, Webb JG. Coronary obstruction in transcatheter aortic valve-in-valve implantation: preprocedural evaluation, device selection, protection, and treatment. Circulation. Cardiovascular interventions. 2015 Jan:8(1):. pii: e002079. doi: 10.1161/CIRCINTERVENTIONS.114.002079. Epub
[PubMed PMID: 25593122]
[44]
Ribeiro HB, Webb JG, Makkar RR, Cohen MG, Kapadia SR, Kodali S, Tamburino C, Barbanti M, Chakravarty T, Jilaihawi H, Paradis JM, de Brito FS Jr, Cánovas SJ, Cheema AN, de Jaegere PP, del Valle R, Chiam PT, Moreno R, Pradas G, Ruel M, Salgado-Fernández J, Sarmento-Leite R, Toeg HD, Velianou JL, Zajarias A, Babaliaros V, Cura F, Dager AE, Manoharan G, Lerakis S, Pichard AD, Radhakrishnan S, Perin MA, Dumont E, Larose E, Pasian SG, Nombela-Franco L, Urena M, Tuzcu EM, Leon MB, Amat-Santos IJ, Leipsic J, Rodés-Cabau J. Predictive factors, management, and clinical outcomes of coronary obstruction following transcatheter aortic valve implantation: insights from a large multicenter registry. Journal of the American College of Cardiology. 2013 Oct 22:62(17):1552-62. doi: 10.1016/j.jacc.2013.07.040. Epub 2013 Aug 14
[PubMed PMID: 23954337]
Level 2 (mid-level) evidence
[45]
Dvir D, Waksman R, Barbash IM, Kodali SK, Svensson LG, Tuzcu EM, Xu K, Minha S, Alu MC, Szeto WY, Thourani VH, Makkar R, Kapadia S, Satler LF, Webb JG, Leon MB, Pichard AD. Outcomes of patients with chronic lung disease and severe aortic stenosis treated with transcatheter versus surgical aortic valve replacement or standard therapy: insights from the PARTNER trial (placement of AoRTic TraNscathetER Valve). Journal of the American College of Cardiology. 2014 Jan 28:63(3):269-79. doi: 10.1016/j.jacc.2013.09.024. Epub 2013 Oct 16
[PubMed PMID: 24140659]
[46]
Siontis GC, Jüni P, Pilgrim T, Stortecky S, Büllesfeld L, Meier B, Wenaweser P, Windecker S. Predictors of permanent pacemaker implantation in patients with severe aortic stenosis undergoing TAVR: a meta-analysis. Journal of the American College of Cardiology. 2014 Jul 15:64(2):129-40. doi: 10.1016/j.jacc.2014.04.033. Epub
[PubMed PMID: 25011716]
Level 1 (high-level) evidence
[47]
Kahlert P, Knipp SC, Schlamann M, Thielmann M, Al-Rashid F, Weber M, Johansson U, Wendt D, Jakob HG, Forsting M, Sack S, Erbel R, Eggebrecht H. Silent and apparent cerebral ischemia after percutaneous transfemoral aortic valve implantation: a diffusion-weighted magnetic resonance imaging study. Circulation. 2010 Feb 23:121(7):870-8. doi: 10.1161/CIRCULATIONAHA.109.855866. Epub
[PubMed PMID: 20177005]
[48]
Webb J, Gerosa G, Lefèvre T, Leipsic J, Spence M, Thomas M, Thielmann M, Treede H, Wendler O, Walther T. Multicenter evaluation of a next-generation balloon-expandable transcatheter aortic valve. Journal of the American College of Cardiology. 2014 Dec 2:64(21):2235-43. doi: 10.1016/j.jacc.2014.09.026. Epub 2014 Nov 24
[PubMed PMID: 25456759]
[49]
Amat-Santos IJ, Dahou A, Webb J, Dvir D, Dumesnil JG, Allende R, Ribeiro HB, Urena M, Paradis JM, DeLarochellière R, Dumont E, Bergeron S, Thompson CR, Pasian S, Bilodeau S, Leipsic J, Larose E, Pibarot P, Rodés-Cabau J. Comparison of hemodynamic performance of the balloon-expandable SAPIEN 3 versus SAPIEN XT transcatheter valve. The American journal of cardiology. 2014 Oct 1:114(7):1075-82. doi: 10.1016/j.amjcard.2014.07.019. Epub 2014 Jul 17
[PubMed PMID: 25132330]
[50]
Panchal HB, Ladia V, Amin P, Patel P, Veeranki SP, Albalbissi K, Paul T. A meta-analysis of mortality and major adverse cardiovascular and cerebrovascular events in patients undergoing transfemoral versus transapical transcatheter aortic valve implantation using edwards valve for severe aortic stenosis. The American journal of cardiology. 2014 Dec 15:114(12):1882-90. doi: 10.1016/j.amjcard.2014.09.029. Epub 2014 Sep 28
[PubMed PMID: 25438917]
Level 1 (high-level) evidence
[51]
Urena M, Webb JG, Eltchaninoff H, Muñoz-García AJ, Bouleti C, Tamburino C, Nombela-Franco L, Nietlispach F, Moris C, Ruel M, Dager AE, Serra V, Cheema AN, Amat-Santos IJ, de Brito FS, Lemos PA, Abizaid A, Sarmento-Leite R, Ribeiro HB, Dumont E, Barbanti M, Durand E, Alonso Briales JH, Himbert D, Vahanian A, Immè S, Garcia E, Maisano F, del Valle R, Benitez LM, García del Blanco B, Gutiérrez H, Perin MA, Siqueira D, Bernardi G, Philippon F, Rodés-Cabau J. Late cardiac death in patients undergoing transcatheter aortic valve replacement: incidence and predictors of advanced heart failure and sudden cardiac death. Journal of the American College of Cardiology. 2015 Feb 10:65(5):437-48. doi: 10.1016/j.jacc.2014.11.027. Epub
[PubMed PMID: 25660921]