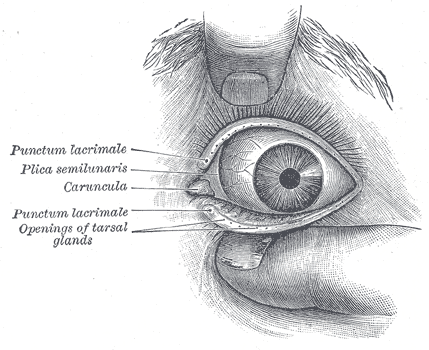
[1]
Kang H, Takahashi Y, Ichinose A, Nakano T, Asamoto K, Ikeda H, Iwaki M, Kakizaki H. Lateral canthal anatomy: a review. Orbit (Amsterdam, Netherlands). 2012 Aug:31(4):279-85. doi: 10.3109/01676830.2012.694957. Epub 2012 Jun 12
[PubMed PMID: 22690873]
[2]
Dantas RR. Lacrimal drainage system obstruction. Seminars in ophthalmology. 2010 May:25(3):98-103. doi: 10.3109/08820538.2010.488577. Epub
[PubMed PMID: 20590420]
[3]
Farmer DT, Nathan S, Finley JK, Shengyang Yu K, Emmerson E, Byrnes LE, Sneddon JB, McManus MT, Tward AD, Knox SM. Defining epithelial cell dynamics and lineage relationships in the developing lacrimal gland. Development (Cambridge, England). 2017 Jul 1:144(13):2517-2528. doi: 10.1242/dev.150789. Epub 2017 Jun 2
[PubMed PMID: 28576768]
[4]
Dean C, Ito M, Makarenkova HP, Faber SC, Lang RA. Bmp7 regulates branching morphogenesis of the lacrimal gland by promoting mesenchymal proliferation and condensation. Development (Cambridge, England). 2004 Sep:131(17):4155-65
[PubMed PMID: 15280212]
[5]
Sherman DD, Gonnering RS, Wallow IH, Lemke BN, Doos WG, Dortzbach RK, Lyon DB, Bindley CD. Identification of orbital lymphatics: enzyme histochemical light microscopic and electron microscopic studies. Ophthalmic plastic and reconstructive surgery. 1993:9(3):153-69
[PubMed PMID: 8217957]
[6]
Scott G, Balsiger H, Kluckman M, Fan J, Gest T. Patterns of innervation of the lacrimal gland with clinical application. Clinical anatomy (New York, N.Y.). 2014 Nov:27(8):1174-7. doi: 10.1002/ca.22447. Epub 2014 Aug 4
[PubMed PMID: 25092807]
[7]
Lorber M, Vidić B. Measurements of lacrimal glands from cadavers, with descriptions of typical glands and three gross variants. Orbit (Amsterdam, Netherlands). 2009:28(2-3):137-46
[PubMed PMID: 19839898]
[8]
Takahashi Y, Watanabe A, Matsuda H, Nakamura Y, Nakano T, Asamoto K, Ikeda H, Kakizaki H. Anatomy of secretory glands in the eyelid and conjunctiva: a photographic review. Ophthalmic plastic and reconstructive surgery. 2013 May-Jun:29(3):215-9. doi: 10.1097/IOP.0b013e3182833dee. Epub
[PubMed PMID: 23381567]
[9]
Scott KR, Tse DT, Kronish JW. Vertically oriented upper eyelid nerve fibers. A clinical, anatomical and immunohistochemical study. Ophthalmology. 1992 Feb:99(2):222-6
[PubMed PMID: 1553211]
[10]
Kim HH, De Paiva CS, Yen MT. Effects of upper eyelid blepharoplasty on ocular surface sensation and tear production. Canadian journal of ophthalmology. Journal canadien d'ophtalmologie. 2007 Oct:42(5):739-42
[PubMed PMID: 17823642]
[11]
Hamawy AH, Farkas JP, Fagien S, Rohrich RJ. Preventing and managing dry eyes after periorbital surgery: a retrospective review. Plastic and reconstructive surgery. 2009 Jan:123(1):353-359. doi: 10.1097/PRS.0b013e31819346ea. Epub
[PubMed PMID: 19116572]
Level 2 (mid-level) evidence
[12]
Tan LT, Davagnanam I, Isa H, Taylor SR, Rose GE, Verity DH, Pusey CD, Lightman S. Clinical and imaging features predictive of orbital granulomatosis with polyangiitis and the risk of systemic involvement. Ophthalmology. 2014 Jun:121(6):1304-9. doi: 10.1016/j.ophtha.2013.12.003. Epub 2014 Feb 20
[PubMed PMID: 24560566]