Continuing Education Activity
Calcium is the most abundant cation found in the human body and plays an integral role in neural transmission, enzyme activity, myocardial function, coagulation, and other cellular functions. Systemic acidosis decreases calcium binding to albumin increasing serum levels while alkalosis causes the opposite effect. Serum calcium concentrations are highest in neonates and infants, decrease during childhood and adolescence, and stabilize at adult values by 17 years of age. Hypercalcemia is defined as serum calcium concentration two standard deviations above the mean values. This activity reviews the cause, pathophysiology, and presentation of hypercalcemia and highlights the role of the interprofessional team in its management.
Objectives:
- Identify the etiology of hypercalcemia.
- Review the presentation of a patient with hypercalcemia.
- Outline the treatment and management options available for hypercalcemia.
- Describe some interprofessional team strategies for improving care coordination and outcomes in patients with hypercalcemia.
Introduction
Calcium is the most abundant cation found in the human body and plays an integral role in neural transmission, enzyme activity, myocardial function, coagulation and other cellular functions. Most of the calcium is found in the bones as calcium phosphate while a small percentage is found in the cells and extracellular fluids. In the serum, about 45% of calcium is bound to proteins, 45% exists as free or ionized calcium that is the active form, while 10% is bound to anions. Systemic acidosis decreases calcium binding to albumin increasing serum levels while alkalosis causes the opposite effect. Serum calcium concentrations are highest in neonates and infants, decrease during childhood and adolescence, and stabilize at adult values by 17 years of age. Hypercalcemia is defined as serum calcium concentration two standard deviations above the mean values. The normal serum calcium ranges from 8.8 mg/dL-10.8 mg/dL. Primary hyperparathyroidism and malignancy accounts for 90% of the cases of hypercalcemia. [1][2]
Etiology
The main cause of hypercalcemia is an excess parathyroid hormone (PTH). PTH-mediated causes include adenoma/hyperplasia of the gland, familial hypocalciuric hypercalcemia, and multiple endocrine neoplasia syndromes (type1, 2A). Familial hypocalciuric hypercalcemia is an autosomal dominant condition due to an inactivating mutation in the calcium-sensing receptor gene.
Although more common in adults than pediatric patients, the next important etiology to consider is a malignancy. Renal carcinomas, leukemias, lymphomas, and rhabdomyosarcoma can be associated with elevated calcium levels mediated by the action of PTH-related peptides.
Hypervitaminosis D can be iatrogenic, secondary to excessive milk intake, or related to granulomatous conditions such as sarcoidosis, tuberculosis or fungal infections.
Endocrine causes such as thyrotoxicosis (due to stimulation of osteoclast function by thyroid hormone), hypoadrenalism and also pheochromocytomas can be associated with hypercalcemia.
Other miscellaneous causes include Williams syndrome, Murk Jansen syndrome, hypophosphatasia, hypervitaminosis A, acute/chronic renal failure, use of thiazide diuretics or lithium, milk-alkali syndrome, subcutaneous fat necrosis, and prolonged immobilization. Williams-Beuren syndrome is characterized by elfin facies, subvalvular aortic stenosis, short stature, and developmental delay. Features of Murk Jansen syndrome include extreme short stature (short-limbed dwarfism), prominent eyes, high-arched palate, wide open fontanelles and micrognathia.[3][4]
Epidemiology
The prevalence of hypercalcemia in the general population is approximately 1% to 2%. Most of the cases (90%) of hypercalcemia are due to primary hyperparathyroidism and malignancy-associated hypercalcemia. The prevalence of primary hyperparathyroidism in the general population ranges from 0.2% to 0.8% and increases with age. Overall, 2% of all cancers are associated with hypercalcemia, but in the pediatric age group, the prevalence is about 0.4% to 1.3%.[5]
Pathophysiology
Calcium concentration is regulated by plasma membrane calcium receptor, PTH and its receptor, calcitonin and its receptor, and by the actions of vitamin D on kidneys, bone, and intestines. PTH mobilizes calcium directly by enhancing bone resorption, and indirectly, by stimulating one alpha-hydroxylase which increases vitamin D3 production, in turn, leading to increased absorption of calcium from the gut and increased bone resorption. Primary hyperparathyroidism is due to a solitary adenoma or diffuse hyperplasia of the gland. In this condition, there is an abnormal set point in the relation between calcium and PTH levels and calcium-independent PTH secretion. Familial hypocalciuric hypercalcemia is inherited autosomal dominant pattern and is due to an inactivating mutation in the calcium-sensing receptor gene. Granulomatous lesions cause ectopic vitamin D production. Transient neonatal hypercalcemia can rarely be seen in infants born to mothers with hypoparathyroidism.[6]
Hypercacemia from hyperparathyroidism is mild and could go one for years and be asymptomatic. Hypercalcemia from malignancy is assoicated with rapidly increasing calcium levels
History and Physical
Symptoms of hypercalcemia are usually seen when serum calcium levels are more than 12 mg/dl. Irrespective of the etiology, the broad signs and symptoms can be summarized as "groans, bones, stones, moans, thrones and psychic overtones".
- Groans: Gastrointestinal symptoms like pain, nausea, and vomiting. Hypercalcemia can lead to peptic ulcer disease and pancreatitis.
- Bones: Bone related complications like bone pain. Hypercalcemia can lead to osteoporosis, osteomalacia, arthritis and pathological fractures.
- Stones: Renal stones causing pain.
- Moans: Refers to fatigue and malaise.
- Thrones: Polyuria, polydipsia, and constipation (sitting on the toilet as you are sitting on a throne).
- Psychic overtones: Lethargy, confusion, depression and memory loss.
Severe hypercalcemia inhibits neuromuscular and myocardial depolarization leading to muscle weakness and arrhythmias. Cardiovascular effects include prolonged PR interval, short QT interval, widened QRS complex, and bradycardia. Increased thirst with polydipsia and polyuria is seen initially, progressing to nephrolithiasis and nephrocalcinosis in chronic cases. Neurologic features include impaired concentration, altered mental status ranging from confusion to irritability. Levels of more than 14 mg/dL can cause encephalopathy and levels above 15 mg/dL is a medica emergency. Severe acute abdominal pain should be a clue to evaluate for pancreatitis. Osseous changes can lead to bone pain, gait abnormalities, and fractures and are seen radiologically as subperiosteal and endosteal bone resorption. In infants and young children, hypercalcemia can cause poor weight gain and failure to thrive.
Evaluation
Hypercalcemia can be classified into
- Mild hypercalcemia: 10.5 to 11.9 mg/dL
- Moderate hypercalcemia: 12.0 to 13.9 mg/dL
- Hypercalecemic crisis: 14.0 to 16.0 mg/dL
Most cases of hypercalcemia are detected on routine testing. Work up for etiology includes obtaining serum PTH, calcitonin, Vitamin D, ionized calcium, phosphorus, magnesium, alkaline phosphatase levels, renal functions, and urinary calcium-creatinine ratio. Sensitive and specific immunochemiluminometric assays should be used to measure parathyroid hormone (PTH) (i-PTH). Hyperparathyroidism is characterized by high calcium, high i-PTH levels with low phosphorous levels. Familial hypocalciuric hypercalcemia has inappropriately normal PTH levels with high magnesium and low calcium creatinine clearance. Other causes of hypercalcemia typically have low PTH levels. Patients with hypercalcemia and hypervitaminosis D have high serum concentrations of calcidiol, whereas, patients with a granulomatous disease have high levels of calcitriol. In patients with hypercalcemia associated with malignancy, the PTH-related peptide levels are elevated. Imaging is indicated to identify nephrolithiasis and nephrocalcinosis. Neonates and infants who are found to be hypercalcemic in the presence of dysmorphic features should prompt evaluation for Williams or Murk Jansen syndrome.[1][7]
Imaging studies are done to rule out sarcoidosis or lung cancer. A mammogram is used to rule out breast cancer and a CT scan is done to rule out renal cancer. The parathyroid glands are investigated with ultrasound and MRI.
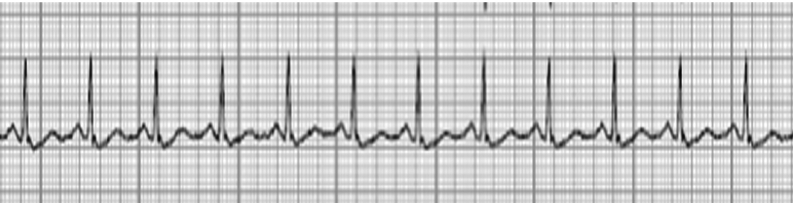
ECG features of hypercalcemia include:
- T wave flattening or inversion
- Mild prolongation of the QRS and PR intervals
- ST-elevation
- Presence of J wave at the end of the QRS complex
Treatment / Management
Treatment for hypercalcemia is required if the patient is symptomatic or if the calcium level is more than 15 mg/dL, even in asymptomatic patients. The goals of treating hypercalcemia include increased elimination from the extracellular fluid, reducing gastrointestinal (GI) absorption and decreasing bone resorption. Immediate therapy is directed at restoring intravascular volume and promoting calcium excretion in the urine with an infusion of 0.9% saline at twice the maintenance rate until any fluid deficit is replaced and diuresis occurs (urine output ≥ 200 mL/h to 300 mL/h). Hemodialysis is the treatment of choice to rapidly decrease serum calcium in patients with heart failure or renal insufficiency. Loop diuretics should be used with caution as even though they may enhance renal excretion, paradoxical hypercalcemia can occur due to bone resorption.
Patients with hyperparathyroidism require surgical exploration and removal of the source of increased PTH secretion. Postoperatively, patients need to be monitored closely for the development of hypocalcemia and tetany. Bisphosphonates such as etidronate, pamidronate, and alendronate are the drugs of choice for hypercalcemia of malignancy as they inhibit osteoclastic activity.
Calcitonin can be administered subcutaneously but in most cases, the effects are mild and limited to a few days. Mithramycin can block the function of osteoclasts and is often administered to patients with malignancy-associated hypercalcemia. but the drug has significant renal, liver, and bone marrow toxicity.
Hypercalcemia associated with excess vitamin D can be treated with steroids as they inhibit one alpha-hydroxylase activity. Ketoconazole, an antifungal agent, has also been used in hypervitaminosis D as it inhibits 1-alpha-hydroxylase activity. Hypercalcemia of immobilization can be prevented by encouraging activity as tolerated and adequate hydration. The specific cause of hypercalcemia needs to be identified, and treatment directed accordingly.[8][9][10]
Differential Diagnosis
- Hypermagnesemia
- Hyperparathyroidism
- Hyperphosphatemia
Prognosis
Hypercalcemia, when it occurs following a benign disorder, has a good prognosis but when the cause is secondary to a malignancy the prognosis is poor. Patients with hypercalcemia associated with malignancy are often symptomatic and need frequent hospitalizations. When hypercalcemia is due to ectopic production of PTH related protein, most patients are dead within a few months. The osteolytic metastatic lesions cause fractures, nerve compression, and paralysis.
Complications
- Depression
- Kidney stones
- Bone pain
- Constipation
- Pancreatitis
- Renal failure
- Gastric ulcers
- Paresthesias
- Syncope and arrhythmias
- Altered mental status
Pearls and Other Issues
Hypercalcemia should be confirmed by taking into account the serum albumin level and measuring an ionized calcium level.
Enhancing Healthcare Team Outcomes
Hypercalcemia is not an uncommon presentation in patients admitted to the hospital. The severity of the symptoms is related to the speed of development and the concentration of calcium. This metabolic problem can present in many ways, and only an interprofessional approach can help reduce the morbidity and mortality of the disorder. Without treatment, the condition carries a very high morbidity.
Besides physicians, pharmacists and nurses play a vital role in the management of patients with hypercalcemia. The nurse plays a vital role in patient hydration and monitoring of vital signs. The pharmacist has to educate the patient on the different drugs available to reduce calcium and their adverse effects. The pharmacist should check the medications to ensure that the cause is not due to a drug. In addition. The patient should be educated about compliance with the bisphosphonates.
All clinicians and nurses should educate the patients on maintaining hydration, changes in diet and limiting calcium intake. The patient and family should be told about the symptoms of hypercalcemia and when to seek assistance. Physical therapy should be involved to ensure that the patient remains mobile and active. If the patient has a metastatic disease associated with hypercalcemia, the hospice, palliative care, and pain teams should be involved to ensure that the quality of life is not compromised.
Finally, a dietitian should be consulted to educate the patient on what type of foods to avoid.[11][12] (Level III) With such a team approach, it is hoped that the patient outcomes will improve. [13] (Level V)
Outcomes
The outcomes depend on the cause of hypercalcemia. When the condition is due to hyperparathyroidism, symptoms tend to be mild but are prolonged. The highest morbidity is due to bone wasting. Mild hypercalcemia does not lead to mortality. However, hypercalcemia from a malignant cause carries a grave prognosis. These patients need aggressive treatment and also require frequent admissions to the hospital. Finally, the morbidity from the malignancy-associated hypercalcemia can be severe leading to many symptoms and a very poor quality of life.[7][14] (Level V)