Continuing Education Activity
Thoracic ultrasound has rapidly gained popularity over the past 10 years. This is in part because ultrasound equipment is available in many settings, more training programs are educating trainees on point of care ultrasound (POCUS), and ultrasound can be done rapidly without exposure to radiation or contrast. Studies have shown equivalent or even improved sensitivity and specificity of POCUS compared with conventional chest radiography, and bedside ultrasounds may aid in differentiating between pathologies that conventional radiographs cannot. Following trauma, thoracic ultrasound can assist with diagnosing pneumothorax, hemothorax, rib fractures, pulmonary contusions, and chest wall hematomas. Non-traumatic uses for thoracic ultrasound include evaluation for pleural effusions, infections such as pneumonia or empyema, pulmonary edema, chronic obstructive pulmonary disease, pulmonary embolism, and acute respiratory distress syndrome. This activity reviews the indications, contraindications, and technique involved in performing thoracic ultrasound and highlights the role of the interprofessional team in the care of patients undergoing this procedure.
Objectives:
- Outline the indications for thoracic ultrasound.
- Describe the technique involved in performing a thoracic ultrasound.
- Review the complications associated with thoracic ultrasound.
- Explain interprofessional team strategies for enhancing coordination and communication to advance the utilization of thoracic ultrasound for the diagnosis of pleural cavity pathology to improve patient outcomes.
Introduction
Thoracic ultrasound has rapidly gained popularity over the past 10 years, mainly due to its wide availability in emergency and trauma settings, as well as trends toward the point of care ultrasound (POCUS) usage in training programs. Additionally, ultrasound circumvents many of the issues that arrive with traditional radiography, such as delay of care and radiation exposure. In an unstable patient, who is unfit for extended delays due to transport to the CT scanner or even bedside chest radiography, bedside ultrasound is readily available to physicians. In addition, numerous studies have shown equivalent or often improved sensitivity and specificity of POCUS compared with conventional radiography, namely chest x-rays. Bedside ultrasound (US) may also aid in differentiating between pathologies that conventional radiography may be unable to determine. [1][2]
Anatomy and Physiology
The thoracic anatomy is composed of the chest wall that is made of musculature including intercostal muscles and pectoralis muscles, the sternum, ribs, costal cartilage, the pleura, including the parietal pleura as well as the visceral pleura, and the lungs themselves. The heart and great vessels are located centrally. Important inferior borders of the thoracic cavity are the diaphragm and liver on the right, and spleen on the left. Interestingly, much of thoracic ultrasound relies less on the lung anatomy, and more on the artifacts that are resultant of the ultrasound physics of air-filled structures. Visualization of the lung parenchyma tends to be more indicative of pathology. Under normal conditions, clinicians are unable to differentiate the parietal and visceral pleura, and only the "pleural line" and its associated artifacts secondary to air scatter pattern can be noted.
Indications
Indications for thoracic ultrasound vary. Trauma is a common history, with bedside thoracic ultrasound useful for diagnosis of pneumothorax, hemothorax, rib fractures, pulmonary contusions, and chest wall hematomas, comprising of the extended form of the focused assessment with sonography for trauma (FAST) exam, known as EFAST. Non-traumatic uses for thoracic ultrasound include evaluation for pleural effusions, infectious causes such as pneumonia and empyema, pulmonary edema, chronic obstructive pulmonary disease (COPD), pulmonary embolism, and ARDS. Symptoms and exam findings that can warrant ultrasonographic evaluation of patients include chest pain, dyspnea, fever, and hypoxia, just to name a few. [3][4][5]
Contraindications
There are no significant contraindications for ultrasonography. Although the demand to transport a patient in need to the operating room in certain situations need not be delayed, the rapid availability of POCUS in most emergency departments, and the limited time needed to complete an evaluation make ultrasound a quick and reasonable mechanism for a rapid assessment before operative management in most cases.
Equipment
Evaluation of the thoracic wall and the pleura are usually evaluated using the 9 to 12 MHz linear-array transducer, allowing more detailed visualization of the more superficial nature of the anatomic structures. In patients with significant thoracic musculature or subcutaneous adipose tissue, transducers with lower frequency may be needed. Evaluation of more distal structures, as in the cases of in pulmonary edema, pneumonia, or pleural effusions, require a lower frequency transducer, such as a 3.5 to 5.0 MHz phased array, or curvilinear transducer are optimal.
Personnel
Any trained personnel is capable of performing thoracic ultrasound evaluation. Currently, specific requirements in thoracic ultrasound are vague in most programs, although it is standard practice that trainees should complete a benchmark of 25 to 50 quality reviewed exams in a particular application.
Preparation
Patients are usually placed in a supine or seated position, keeping in mind the physics of the medium which is being an evaluation. For example, pleural effusions will collect in a dependent manner, and a seated position, if the patient is able, allowing gravity to cause effusions to collect at the inferior lung region, just above the diaphragm. A pneumothorax will rise to the apex of the area it is confined. However, a lung consolidation such as community-acquired pneumonia will be seen relatively independent of position.
Technique or Treatment
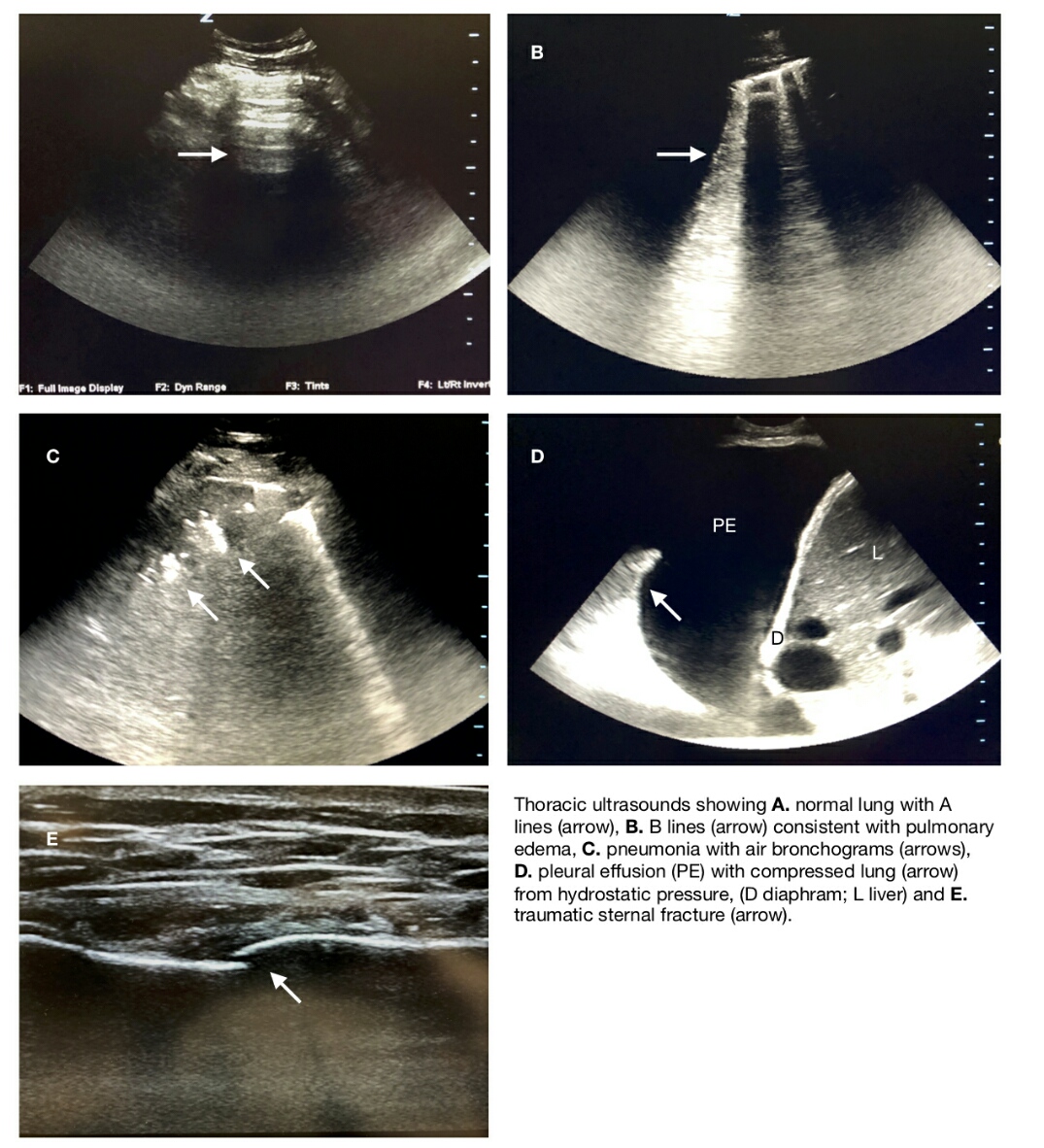
Initial evaluation of the thoracic cavity involves identification of its surrounding borders including the thoracic wall (subcutaneous tissues, musculature, and ribs), the inferior border of the diaphragm, the liver, spleen, the heart and its major vessels. The 9 to 12-MHz linear-array probe is ideal for the initial exam of the chest wall because it provides detailed images of the structure of the underlying soft tissues. Trauma may subject the patient to forces causing significant chest wall contusions and hematomas, especially if rib fractures occur, severing the intercostal vessels. In such cases, the usual regular muscle striations and soft tissue textures will be replaced with tissue edema, resulting in a "cobblestoning" appearance, and in the case of soft tissue hematoma, a localized, hypoechoic fluid collection subcutaneously. Older hematomas in which clotting has initiated may demonstrate a slightly more hyperechoic and fibrous appearance, similar to the surrounding tissue. Subcutaneous emphysema may also be noted in this view, seen as hyperechoic, vertical lines that may obscure the pleural line, known as E lines or "comet tail" artifacts. E lines result from the difference in acoustic impedance between an object and its surroundings, causing the sound wave to resonate, with lag time between successive reverberations interpreted as distance. Rib fractures, especially early on, may often be missed on routine chest x-rays. Occult rib fractures have been noted in 80% of patients physical examined. X-rays are sensitive but lack specificity. Chest radiography has been estimated to miss half of all rib fractures, and one study noted sensitivities of 15% and 90% for chest radiography and ultrasound respectively. Although CT scanning is more sensitive and specific for rib fractures, minor traumas may not always require such radiation exposure. In addition, rib fractures, with the possible exception of flail chest, rarely requires emergent treatment prior to patient stabilization. In such cases, POCUS is ideal for evaluation. The linear probe is placed over the maximal point of tenderness or pain, and scanned, looking for areas of discontinuity along the anterior rib cortex. Normally, the anterior rib cortex should appear hyperechoic with posterior shadowing obscuring the posterior cortex. Fracture discontinuity is more obvious with displacement, and even occult fractures may be suspected if there is surrounding hematoma present. Of note, sternal and clavicular fractures can also be noted on POCUS with this same technique, although incomplete sternal fusion in children may give false positive results when looking from injuries.
Disruptions in the normal visceral-parietal pleural interface via the introduction of air or fluid, are also key uses for thoracic ultrasonography. Under normal conditions, the parietal and visceral pleura are visualized as a single hyperechoic line known as the "pleural line." It is approximately 1 cm distal to the ribs. Acoustic impedance due to the underlying lung produces an echo artifact known as A lines, which are characteristically hyperechoic, regularly spaced horizontal lines below the pleural line. There are also Z lines which are short, broad, ill-defined vertical comet tail artifacts arising from the pleural line but do not extend to the edge of the screen. Lung sliding may also be noted as the parietal and visceral pleura slide over each other and are visualized via ultrasound as a "shimmering" back and forth movement appearance of the pleural line and associated artifacts. Introduction of free air into the intrapleural space, as in the case of pneumothorax, disrupts the visualization of the lung sliding, and the pleural line appears static rather than dynamic. The loss of lung sliding is sensitive, but not specific for pneumothorax, as pleural adhesions, history of pleurodesis, atelectasis, mainstem intubation, apnea, and COPD can all give similar appearances. However, the "lung point" sign has been shown to be the most specific for pneumothorax. Lung point can be observed when the transducer is over the boundary between the aerated lung and the initiation of the pneumothorax, in which lung sliding will move in and out of view with the normal respiratory cycle. This may not be seen in the complete collapse of the lung parenchyma, and false positive lung points may be observed over pleural borders at the heart and diaphragm. Also of note, "lung pulse" is the term associated with lung sliding seen in apneic patients resulting from slight movements along a normal pleural line due to the pulsations transmitted from the cardiac motions. Some evaluators may choose to use M mode in their evaluation, showing dynamic changes in the chest wall and underlying parenchyma, giving a "sandy beach" or "seashore" sign appearance. In this view, a normal lung will show a more superficial, linear appearance (small waves in water) of the stationary chest wall. The underlying, mobile, lung parenchyma appears more granular, like the sand on a beach. Disruption of the normal anatomy with a pneumothorax will cause loss of the more distal granule appearance and be replaced with continuing horizontal lines, often named the "barcode" or "stratosphere" sign.
In contrast to air collection in the thoracic pleural cavity, fluid collections transmit sonographic signals well, producing an anechoic or hypoechoic (depending on the cause and duration) space between the parietal and visceral pleura. Hyperechoic debris may be noted within the effusion, and clotted blood may be seen in the case of hemothorax, often in the dependent portion of the space. Traumatic hemothorax may also be accompanied by rib fractures or pulmonary contusions, also visible on ultrasound. Whereas supine chest x-ray has been shown to reveal pleural effusions as low as 175 to 525 ml, ultrasound has been shown to be able to identify 5 to 20 ml of fluid with a sensitivity of 89% to 100%, and a specificity of 96% to 100%. It is imperative that the normal anatomy is identified, for example, the diaphragm, as abdominal ascites or other fluid collections may be confused with pleural effusions. In larger effusions, a lung that has collapsed under the pressure of the fluid collection may be seen waving in a fluid motion with the patient's respiratory efforts. This has been dubbed the "plankton sign." Estimation of the effusion volume may be performed by measuring the largest distance in centimeters from the lung base to the diaphragm and multiplying it by 200 mL/cm. For diagnostic or therapeutic thoracentesis, direct visualization of the effusion and needle may be visualized real-time with the probe, covered in a sterile sheath, to obtain optimum placement so to avoid iatrogenic injury.
As noted above, lung parenchyma is normally not visualized directly with ultrasonographic evaluation. However, artifacts may be noted in some pathologies giving pathognomonic clues to them. In the case of acute alveolar interstitial syndromes in which there is impaired gas exchange due to a pathologic fluid within the pulmonary interstitium and alveolar space, a vertical, hyperechoic, ring down artifact will be noted. It extends to the bottom of the screen, and moves with respiratory effort, appearing as if a flashlight or multiple flashlights were being swung back and forth. These artifacts, known as B lines or "lung rockets" are secondary to the reverberation of ultrasonographic waves due to the fluid within the interlobular septae. B lines are diagnostic of pulmonary edemata, such as occur in congestive heart failure and will be noted before fluid accumulation in the alveolar spaces. Evaluation for B lines is performed using the phased array or curvilinear probes, evaluating multiple regions of bilateral lung fields, with diagnosis generally including at least 3 or more B lines noted on an exam in at least 2 fields. Thoracic ultrasound diagnosis of pulmonary edema has been shown to precede chest radiographic abnormalities and has a sensitivity and specificity of 97% and 95% respectively. Additional evaluation with cardiac ultrasound can additionally be used to diagnose ejection fraction in cardiogenic pulmonary edema (see cardiac ultrasound section), as can ultrasonographic evaluation of the IVC during respiration. The number of B lines present corresponds to the severity of the pulmonary edema present and correlates with the degree of improvement after treatment. Focal and localized B lines may also be noted in traumatic pulmonary contusion and may be utilized for early detection in trauma evaluations.
Infectious etiologies of dyspnea my be also investigated using thoracic ultrasound technique, given the frequent production of purulent material transforming typically non-echogenic lung parenchyma into an echogenic medium. In the case of consolidated pneumonia, heterogeneous, hyperechoic foci with irregular borders in the subpleural parenchyma may be noted having the appearance similar to the liver and coined "hepatization" of the lung. In addition, static or dynamic air bronchograms, seen as hyperechoic foci that may move with respirations within the lung parenchyma, representing air movement within the diseased lung, may be noted, as well as associated effusions. Of note, atelectasis may appear similar to pneumonia with lung consolidation on both chest x-ray and thoracic ultrasound. Presence of static or dynamic bronchograms may allow differentiation between the 2 conditions, as dynamic air bronchograms, in which the hyperechoic artifacts are mobile, moving greater than 1 mm with respiratory efforts, are more likely in pneumonia than in atelectasis. Additional history and findings such as fever and leukocytosis may strengthen the practitioner's suspicion of pneumonia, as well. Empyema may also be seen with bedside ultrasound, appearing as a hyperechoic fluid collection within the lung parenchyma, often septated or loculated appearance. Early findings may appear hypoechoic without septa. [6][7][8][9]
Complications
Few complications exist with basic thoracic ultrasound short of pain with an applied probe to areas of tenderness, as in rib fractures. Although ultrasound can be performed rapidly at the bedside by qualified personnel, operative management should not be delayed in critical cases where ultrasonographic evaluation will add little to the care of the patient. In the case of bedside ultrasound, use in procedures such as thoracentesis, it is critical that all landmarks (diaphragm, liver, spleen, lung, and chest wall) are identified prior to intervention as to avoid iatrogenic injury. Real-time direct visualization of needle or catheter is recommended.
Clinical Significance
The diagnostic and procedural use of thoracic ultrasound has rapidly grown within the last 10 years due to being largely accepted by emergency physicians' adaptation of POCUS into formal residency training, as well as improvements in the bedside ultrasounds themselves. It allows rapid diagnosis and visualization of pathology and improves accuracy in diagnosis and procedures without the risk of exposure to potentially harmful radiation. [5]
Enhancing Healthcare Team Outcomes
Thoracic ultrasound is sometimes performed by the emergency department physician, internist, intensivist or nurse practitioner to look for pleural fluid or aspiration of a fluid collection from the chest cavity. It is important that that only professionals trained in ultrasound use this technique. If there is any doubt about the diagnosis, a radiologist should be consulted first before inserting any tube or needle in the chest cavity. Once the fluid is removed, a chest x-ray is recommended to ensure that there is no pneumothorax.