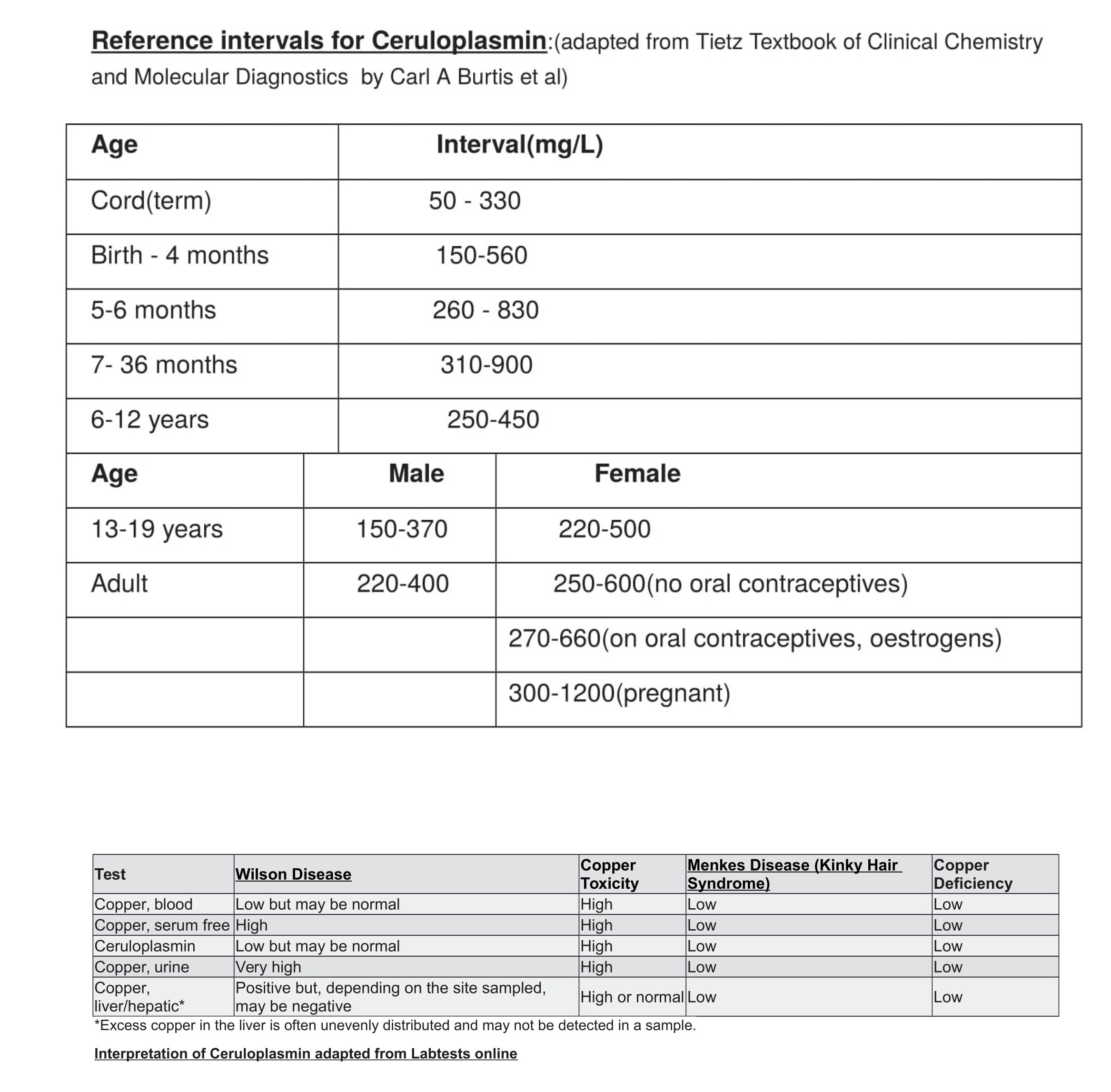
[1]
Walshe JM, Cause of death in Wilson disease. Movement disorders : official journal of the Movement Disorder Society. 2007 Nov 15;
[PubMed PMID: 17712859]
[2]
Vasilyev VB, Interactions of caeruloplasmin with other proteins participating in inflammation. Biochemical Society transactions. 2010 Aug;
[PubMed PMID: 20658982]
[3]
Twomey PJ,Viljoen A,House IM,Reynolds TM,Wierzbicki AS, Relationship between serum copper, ceruloplasmin, and non-ceruloplasmin-bound copper in routine clinical practice. Clinical chemistry. 2005 Aug
[PubMed PMID: 16040861]
[4]
Squitti R,Quattrocchi CC,Forno GD,Antuono P,Wekstein DR,Capo CR,Salustri C,Rossini PM, Ceruloplasmin (2-D PAGE) Pattern and Copper Content in Serum and Brain of Alzheimer Disease Patients. Biomarker insights. 2007 Feb 7
[PubMed PMID: 19690651]
[5]
Squitti R,Quattrocchi CC,Salustri C,Rossini PM, Ceruloplasmin fragmentation is implicated in 'free' copper deregulation of Alzheimer's disease. Prion. 2008 Jan-Mar
[PubMed PMID: 19164917]
[6]
Sharp P, The molecular basis of copper and iron interactions. The Proceedings of the Nutrition Society. 2004 Nov;
[PubMed PMID: 15831128]
[7]
Maio N,Polticelli F,De Francesco G,Rizzo G,Bonaccorsi di Patti MC,Musci G, Role of external loops of human ceruloplasmin in copper loading by ATP7B and Ccc2p. The Journal of biological chemistry. 2010 Jul 2
[PubMed PMID: 20430895]
[8]
Sharonov BP,Govorova NIu, [Oxidation of ceruloplasmin by hypochlorite. The loss of blue color and preservation of oxidase activity]. Biokhimiia (Moscow, Russia). 1990 Jun;
[PubMed PMID: 2169906]
[9]
Hurd RW,Van Rinsvelt HA,Wilder BJ,Karas B,Maenhaut W,De Reu L, Selenium, zinc, and copper changes with valproic acid: possible relation to drug side effects. Neurology. 1984 Oct;
[PubMed PMID: 6435012]
[11]
Vlasova II,Sokolov AV,Kostevich VA,Mikhalchik EV,Vasilyev VB, Myeloperoxidase-Induced Oxidation of Albumin and Ceruloplasmin: Role of Tyrosines. Biochemistry. Biokhimiia. 2019 Jun;
[PubMed PMID: 31238865]
[14]
Bento I,Peixoto C,Zaitsev VN,Lindley PF, Ceruloplasmin revisited: structural and functional roles of various metal cation-binding sites. Acta crystallographica. Section D, Biological crystallography. 2007 Feb
[PubMed PMID: 17242517]
[15]
Harazono A,Kawasaki N,Itoh S,Hashii N,Ishii-Watabe A,Kawanishi T,Hayakawa T, Site-specific N-glycosylation analysis of human plasma ceruloplasmin using liquid chromatography with electrospray ionization tandem mass spectrometry. Analytical biochemistry. 2006 Jan 15
[PubMed PMID: 16321355]
[16]
Ibragimova MI,Chushnikov AI,Moiseev VN,Petukhov VIu,Zheglov EP,Pogorel'tsev VI,Khardina NB, [Electron paramagnetic resonance determination of ceruloplasmin concentration in the blood of patients for the diagnosis of urological cancer diseases]. Klinicheskaia laboratornaia diagnostika. 2010 Apr
[PubMed PMID: 20524342]
[17]
Levitan I,Volkov S,Subbaiah PV, Oxidized LDL: diversity, patterns of recognition, and pathophysiology. Antioxidants & redox signaling. 2010 Jul 1
[PubMed PMID: 19888833]
[18]
Moriya M,Ho YH,Grana A,Nguyen L,Alvarez A,Jamil R,Ackland ML,Michalczyk A,Hamer P,Ramos D,Kim S,Mercer JF,Linder MC, Copper is taken up efficiently from albumin and alpha2-macroglobulin by cultured human cells by more than one mechanism. American journal of physiology. Cell physiology. 2008 Sep;
[PubMed PMID: 18579803]
[19]
González-Jiménez E,Schmidt-Riovalle J,Sinausía L,Carmen Valenza M,Perona JS, Predictive value of ceruloplasmin for metabolic syndrome in adolescents. BioFactors (Oxford, England). 2016 Mar-Apr
[PubMed PMID: 27083758]
[20]
Yada T,Nagae M,Moriyama S,Azuma T, Effects of prolactin and growth hormone on plasma immunoglobulin M levels of hypophysectomized rainbow trout, Oncorhynchus mykiss. General and comparative endocrinology. 1999 Jul
[PubMed PMID: 10375463]
Level 2 (mid-level) evidence
[21]
Hermann W, Classification and differential diagnosis of Wilson's disease. Annals of translational medicine. 2019 Apr
[PubMed PMID: 31179300]
[22]
Gautam B,Deb K,Banerjee M,Ali MS,Akhter S,Shahidullah SM,Hoque MR, Serum zinc and copper level in children with protein energy malnutrition. Mymensingh medical journal : MMJ. 2008 Jul;
[PubMed PMID: 18946444]
[23]
Kalra R,Kalra VB,Sareen PM,Khandelwal R, Serum copper and ceruloplasmin in pregnancy with anaemia. Indian journal of pathology & microbiology. 1989 Jan
[PubMed PMID: 2592032]
[24]
Esme H,Cemek M,Sezer M,Saglam H,Demir A,Melek H,Unlu M, High levels of oxidative stress in patients with advanced lung cancer. Respirology (Carlton, Vic.). 2008 Jan
[PubMed PMID: 18197920]
[25]
Wolf TL,Kotun J,Meador-Woodruff JH, Plasma copper, iron, ceruloplasmin and ferroxidase activity in schizophrenia. Schizophrenia research. 2006 Sep
[PubMed PMID: 16842975]
[26]
Manolaki N,Nikolopoulou G,Daikos GL,Panagiotakaki E,Tzetis M,Roma E,Kanavakis E,Syriopoulou VP, Wilson disease in children: analysis of 57 cases. Journal of pediatric gastroenterology and nutrition. 2009 Jan;
[PubMed PMID: 19172127]
Level 3 (low-level) evidence
[27]
Weiss KH,Merle U,Schaefer M,Ferenci P,Fullekrug J,Stremmel W, Copper toxicosis gene MURR1 is not changed in Wilson disease patients with normal blood ceruloplasmin levels. World journal of gastroenterology. 2006 Apr 14
[PubMed PMID: 16610028]
[28]
Kroll CA,Ferber MJ,Dawson BD,Jacobson RM,Mensink KA,Lorey F,Sherwin J,Cunningham G,Rinaldo P,Matern D,Hahn SH, Retrospective determination of ceruloplasmin in newborn screening blood spots of patients with Wilson disease. Molecular genetics and metabolism. 2006 Sep-Oct;
[PubMed PMID: 16644258]
Level 2 (mid-level) evidence
[30]
McNeill A,Pandolfo M,Kuhn J,Shang H,Miyajima H, The neurological presentation of ceruloplasmin gene mutations. European neurology. 2008
[PubMed PMID: 18667828]
[31]
Jafri SK,Kumar R,Lashari SK,Chand P, Menkes disease: A rare disorder. JPMA. The Journal of the Pakistan Medical Association. 2017 Oct;
[PubMed PMID: 28955085]
[32]
Prohaska JR, Role of copper transporters in copper homeostasis. The American journal of clinical nutrition. 2008 Sep
[PubMed PMID: 18779302]
[33]
Target cells for transformation with avian leukosis viruses., Graf T,Royer-Pokora B,Beug H,, Hamatologie und Bluttransfusion, 1976
[PubMed PMID: 16791614]
[36]
Growth regulation and suppression of metastasis in the congenitally athymic nude mouse., Stiles CD,Roberts PE,Saier MH Jr,Sato G,, Hamatologie und Bluttransfusion, 1976
[PubMed PMID: 30190489]
[37]
Cell-mediated immunity to leukemia associated antigens in experimental models and in man., Herberman RB,, Hamatologie und Bluttransfusion, 1976
[PubMed PMID: 29979436]