Introduction
The abdominal aorta's first major branch, the celiac trunk, arises anteriorly at approximately the T12 vertebral level. This short artery is the primary blood supply of the foregut and foregut-derived organs in the superior abdominal cavity. The celiac trunk is an important blood vessel that may vary in form and branching pattern. Pancreatic dysfunction, liver failure, and hemorrhage are some of the conditions that can arise from celiac trunk pathology. Knowledge of this vessel's branches and their variants helps to interpret abdominal imaging studies and limit surgical complications appropriately. This topic discusses the anatomy and clinical importance of the celiac trunk.
Structure and Function
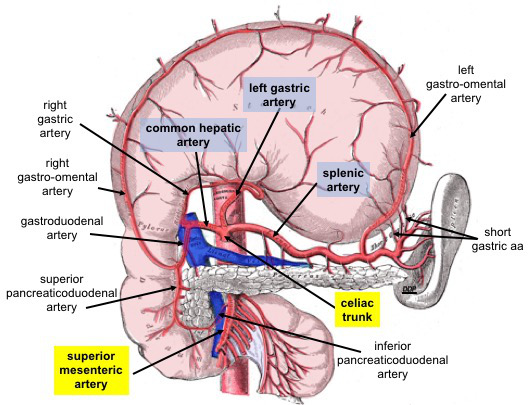
The celiac artery, also known as the celiac trunk, is a short vessel arising from the aorta just as it enters the abdomen at approximately the level of the T12 vertebra. The celiac trunk courses inferiorly deep to the median arcuate ligament, typically dividing into 3 main branches (see Image. Celiac Trunk Branches):
- Left gastric artery: normally the smallest of the 3 celiac trunk branches. The left gastric artery supplies the proximal part of the stomach's lesser curvature and the distal esophagus. This vessel anastomoses with the right gastric artery, a branch of the common hepatic artery.
- Common hepatic artery: branches of this vessel supply the liver, stomach pylorus, distal part of the greater and lesser stomach curvatures, gallbladder, proximal duodenum, and pancreatic head.
- Splenic artery: branches of this artery supply the proximal and middle sections of the greater stomach curvature, the stomach fundus, a large portion of the pancreas, and the spleen. The splenic artery ends its course at the spleen.
The celiac artery typically measures about 1.5 to 2 cm in length and supplies the foregut and foregut-derived structures, which include the following:
- Distal esophagus
- Stomach
- Proximal one-third of the duodenum
- Liver
- Pancreas
- Gallbladder
- Spleen [1][2]
Maintaining these organs' functions entails good blood flow in the celiac trunk.
Embryology
The arterial system undergoes numerous modifications in utero. Intraembryonically, the arterial system includes paired dorsal aortae that eventually fuse to form the abdominal aorta. Paired ventral segmental arteries arise from the dorsal aorta, with some fusing to form the median vessels. The median vessels then give rise to the 3 main arteries serving the abdominal viscera: the celiac trunk, superior mesenteric artery, and inferior mesenteric artery.
By the 4th week of embryonic development, the gastrointestinal tract has differentiated into the foregut, midgut, and hindgut. The foregut spans from the esophagus superiorly to the proximal third of the duodenum inferiorly, just past the hepatopancreatic ampulla (ampulla of Vater). The midgut extends from the proximal duodenum to the proximal two-thirds of the transverse colon, terminating at the left colic or splenic flexure. The hindgut extends from approximately the left colic flexure to the rectum. Each of the gut segments has a unique blood supply arising from the abdominal aorta. Additionally, the blood supply to the gut segments distinguishes the foregut, midgut, and hindgut.
Blood Supply and Lymphatics
Blood Supply
The 3 major celiac trunk branches are the left gastric, splenic, and common hepatic arteries. These arteries supply the foregut, foregut derivatives, and spleen. The left gastric artery gives off branches that supply the inferior part of the esophagus. The vessel then travels along the lesser stomach curvature and anastomoses with the right gastric artery. This anastomosis is an important collateral circulatory route.
The splenic artery travels posterior to the stomach and gives rise to the left gastroepiploic artery, which delivers blood to the proximal greater stomach curvature. The splenic artery also gives off branches to the tail and body of the pancreas.
The short gastric arteries, which supply the stomach fundus, also diverge from the splenic artery. These blood vessels do not anastomose with any other arteries. Splenic artery obstruction or rupture may cause ischemia of the stomach fundus, which lacks collateral blood supply.
The common hepatic artery is the liver's only arterial source. This blood vessel gives off the proper hepatic, gastroduodenal, and right gastric arteries. The proper hepatic artery gives rise to the right and left hepatic arteries. Some texts list the right gastric artery as a branch of the proper hepatic artery. However, the anatomic definition of "proper" requires that the structure only serves its namesake, which is the liver in this case.
The right gastric artery delivers blood to the pylorus and the distal portion of the lesser curvature of the stomach. The right and left hepatic arteries supply the corresponding liver lobes. The cystic artery, usually a right hepatic artery branch, supplies the gallbladder. This blood vessel originates from the left gastric artery approximately 1.2% of the time.[3]
The gastroduodenal artery gives off the right gastroepiploic and superior pancreaticoduodenal arteries. The right gastroepiploic artery supplies the distal portion of the greater curvature of the stomach. The superior pancreaticoduodenal artery delivers blood to the proximal head of the pancreas and part of the duodenum. The pancreatic head is the most common site of pancreatic adenocarcinoma and is typically removed in a Whipple procedure.
Lymphatics
Celiac lymph nodes around the celiac trunk collect lymph from the liver, gallbladder, stomach, spleen, and pancreas. Lymph vessels in this region drain to the cisterna chyli, which ultimately empties into the thoracic duct.
Nerves
The celiac ganglia are 2 large and irregularly shaped collections of neuronal cell bodies lateral to the celiac trunk at approximately the T12 to L1 spinal levels. These ganglia are part of the autonomic nervous system, innervating the foregut and foregut-derived abdominal organs. The celiac ganglia carry sympathetic input to the abdominal parenchyma and are vital in coordinating digestive functions like motility, secretion, and absorption.[4]
Physiologic Variants
Celiac trunk branching variations have been reported in the literature. They include the following:[5][6]
- Bifurcation into the common hepatic and splenic arteries
- Formation of the celiacomesenteric trunk, where the celiac and superior mesenteric arteries have a common origin
- Inferior phrenic arteries branching out from the celiac trunk (normal: these arteries originate from the abdominal aorta)
- Left gastric artery branching directly from the abdominal aorta
Abdominal anastomoses are crucial during surgical procedures requiring blood vessel ligation. Knowledge of the normal celiac trunk anatomy and its variants helps surgeons limit complications from abdominal procedures. Additionally, a thorough understanding of abdominal vasculature helps radiologists accurately interpret imaging studies of this region.
Surgical Considerations
Celiac vascular anomalies are mostly asymptomatic. However, iatrogenic injuries may occur by ligating an aberrant arterial branch or inadvertently cutting an accessory vessel. A thorough knowledge of this abdominal vasculature segment and its possible variations helps reduce blood loss during abdominal surgical procedures.[6][7]
Clinical Significance
Celiac artery pathology can lead to hemorrhage or dysfunction of the organs it supplies. Symptoms range from mild to potentially fatal, depending on factors like injury type, clinical course, and patient's health status. Below are some diseases that can affect this vital arterial structure.
Celiac Artery Compression Syndrome
Celiac artery compression syndrome (CACS) is known by other names: celiac axis syndrome, median arcuate ligament syndrome (MALS), Marable syndrome, and Dunbar syndrome. This condition characteristically presents as pain localized to the upper abdomen due to celiac artery compression by the diaphragmatic crura.
The median arcuate ligament is a fibrous arch connecting the right and left diaphragmatic crura around the aorta at the base of the diaphragm. A low-lying median arcuate ligament can impinge on the celiac trunk inferiorly, resulting in excruciating epigastric pain. The pain is often relieved in the standing position and triggered in the supine position.
The condition commonly presents in individuals 20 to 40 years old. CACS may result in mesenteric ischemia in some cases, which presents with epigastric postprandial pain and weight loss.
Computed tomography (CT) angiography will reveal focal stenosis of the vessel with a characteristic hook-shaped appearance, representing celiac trunk distortion on its superior surface. The celiac artery can have a similar appearance in healthy individuals. Thus, imaging findings must be correlated with patient history.
CACS treatment is via laparoscopic surgical decompression, specifically by dividing the median arcuate ligament. This procedure is usually reserved for symptomatic patients.
Celiac Aneurysm
Although rare, celiac artery aneurysm can occur as a form of splanchnic artery aneurysm. A celiac aneurysm is typically asymptomatic unless it ruptures. Early recognition of this vascular condition is crucial since celiac aneurysm rupture carries high morbidity and mortality rates. Celiac aneurysm is usually an incidental finding in diagnostic imaging studies like arteriography or discovered in post-rupture autopsies.
Celiac Trunk Dissection
Celiac trunk dissection is most commonly iatrogenic. However, the condition may also develop in people with atherosclerosis, hypertension, and pre-existing vascular diseases like fibromuscular dysplasia. Other predisposing factors include pregnancy, trauma, and sudden intra-abdominal pressure elevation.
Celiac trunk dissection is frequently asymptomatic. However, pain may occur when bowel ischemia develops from the rupture of the celiac artery's foregut branches or nearby vessels like the renal and superior mesenteric arteries.
CT with contrast or CT angiography is the optimal means of diagnosing celiac trunk dissection. Magnetic resonance (MR) angiography or sonography may also be considered. The most common findings on diagnostic imaging of celiac trunk dissection are intimal flaps and intraluminal mural thrombi.
Surgery is the definitive treatment for celiac trunk dissection, as it can prevent acute complications like rupture and intestinal ischemia and chronic complications like arterial stenosis. However, a conservative approach may be taken if the arterial dissection is limited. Prevention of thromboembolic events is key. Patients may be managed with anticoagulation and antihypertensives. Endovascular stenting may be considered if the collateral circulation is healthy.[8]
Peptic Ulcer Disease
Gastric or duodenal ulcers are painful sores that often result from Helicobacter pylori infection, long-term non-steroidal anti-inflammatory drug (NSAID) use, or tobacco smoking. Patients usually present with epigastric pain and early satiety. Gastric ulcers are associated with pain aggravated by meals. In contrast, duodenal ulcers present with pain relieved by food intake. Gastric ulcers most commonly appear in the lesser curvature of the stomach, which is supplied by the right and left gastric arteries. Duodenal ulcers most commonly occur on the posterior duodenal wall, an area supplied by the gastroduodenal artery.
Peptic ulcer perforation may damage celiac artery branches. Erosion of the superior mesenteric artery is another potential complication of this condition.
The gold standard for peptic ulcer disease diagnosis is esophagogastroduodenoscopy (EGD). Management typically includes H2-receptor antagonists and proton pump inhibitors (PPIs).