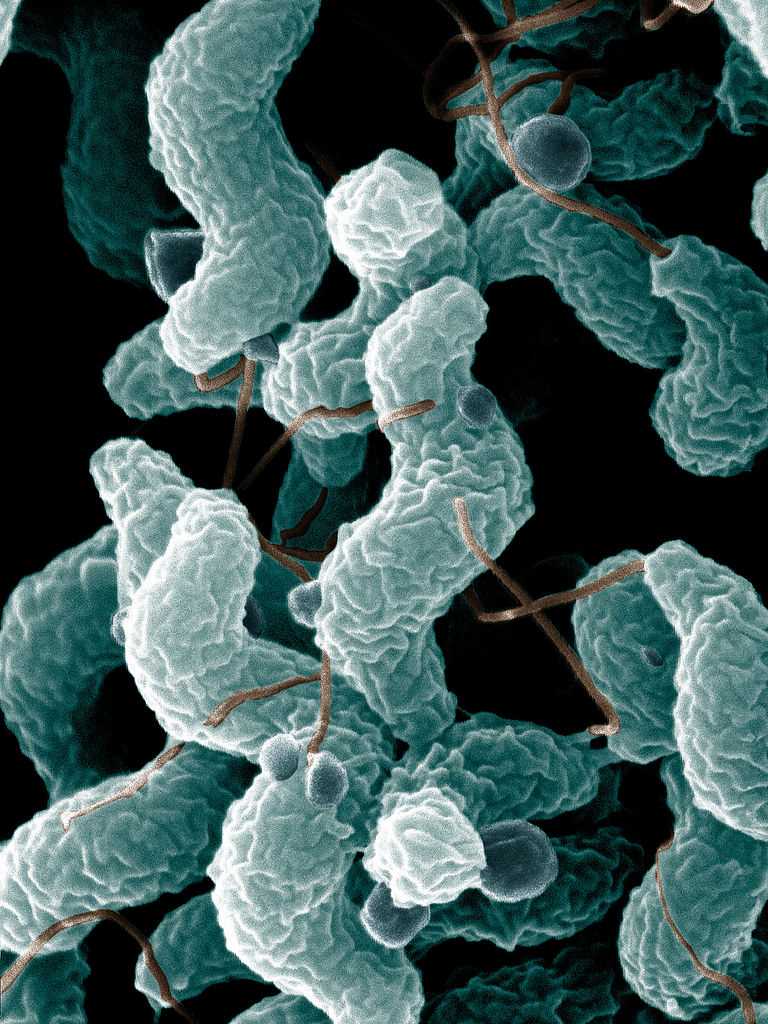
[1]
Kaakoush NO, Castaño-Rodríguez N, Mitchell HM, Man SM. Global Epidemiology of Campylobacter Infection. Clinical microbiology reviews. 2015 Jul:28(3):687-720. doi: 10.1128/CMR.00006-15. Epub
[PubMed PMID: 26062576]
[2]
Serichantalergs O, Ruekit S, Pandey P, Anuras S, Mason C, Bodhidatta L, Swierczewski B. Incidence of Campylobacter concisus and C. ureolyticus in traveler's diarrhea cases and asymptomatic controls in Nepal and Thailand. Gut pathogens. 2017:9():47. doi: 10.1186/s13099-017-0197-6. Epub 2017 Aug 17
[PubMed PMID: 28824712]
Level 3 (low-level) evidence
[3]
Sanders JW, Isenbarger DW, Walz SE, Pang LW, Scott DA, Tamminga C, Oyofo BA, Hewitson WC, Sanchez JL, Pitarangsi C, Echeverria P, Tribble DR. An observational clinic-based study of diarrheal illness in deployed United States military personnel in Thailand: presentation and outcome of Campylobacter infection. The American journal of tropical medicine and hygiene. 2002 Nov:67(5):533-8
[PubMed PMID: 12479558]
[4]
Newell DG, Fearnley C. Sources of Campylobacter colonization in broiler chickens. Applied and environmental microbiology. 2003 Aug:69(8):4343-51
[PubMed PMID: 12902214]
[5]
Sibanda N, McKenna A, Richmond A, Ricke SC, Callaway T, Stratakos AC, Gundogdu O, Corcionivoschi N. A Review of the Effect of Management Practices on Campylobacter Prevalence in Poultry Farms. Frontiers in microbiology. 2018:9():2002. doi: 10.3389/fmicb.2018.02002. Epub 2018 Aug 24
[PubMed PMID: 30197638]
[6]
Kuhn KG, Hvass AK, Christiansen AH, Ethelberg S, Cowan SA. Sexual Contact as Risk Factor for Campylobacter Infection, Denmark. Emerging infectious diseases. 2021 Apr:27(4):1133-1140. doi: 10.3201/eid2704.202337. Epub
[PubMed PMID: 33754996]
[7]
Rincé A, Balière C, Hervio-Heath D, Cozien J, Lozach S, Parnaudeau S, Le Guyader FS, Le Hello S, Giard JC, Sauvageot N, Benachour A, Strubbia S, Gourmelon M. Occurrence of Bacterial Pathogens and Human Noroviruses in Shellfish-Harvesting Areas and Their Catchments in France. Frontiers in microbiology. 2018:9():2443. doi: 10.3389/fmicb.2018.02443. Epub 2018 Oct 11
[PubMed PMID: 30364306]
[8]
Black RE, Levine MM, Clements ML, Hughes TP, Blaser MJ. Experimental Campylobacter jejuni infection in humans. The Journal of infectious diseases. 1988 Mar:157(3):472-9
[PubMed PMID: 3343522]
[9]
Epps SV, Harvey RB, Hume ME, Phillips TD, Anderson RC, Nisbet DJ. Foodborne Campylobacter: infections, metabolism, pathogenesis and reservoirs. International journal of environmental research and public health. 2013 Nov 26:10(12):6292-304. doi: 10.3390/ijerph10126292. Epub 2013 Nov 26
[PubMed PMID: 24287853]
[10]
Murphy H, Cogan T, Humphrey T. Direction of neutrophil movements by Campylobacter-infected intestinal epithelium. Microbes and infection. 2011 Jan:13(1):42-8. doi: 10.1016/j.micinf.2010.09.007. Epub 2010 Oct 8
[PubMed PMID: 20934530]
[11]
Hadden RD, Karch H, Hartung HP, Zielasek J, Weissbrich B, Schubert J, Weishaupt A, Cornblath DR, Swan AV, Hughes RA, Toyka KV, Plasma Exchange/Sandoglobulin Guillain-Barré Syndrome Trial Group. Preceding infections, immune factors, and outcome in Guillain-Barré syndrome. Neurology. 2001 Mar 27:56(6):758-65
[PubMed PMID: 11274311]
[12]
Smith KE, Besser JM, Hedberg CW, Leano FT, Bender JB, Wicklund JH, Johnson BP, Moore KA, Osterholm MT. Quinolone-resistant Campylobacter jejuni infections in Minnesota, 1992-1998. Investigation Team. The New England journal of medicine. 1999 May 20:340(20):1525-32
[PubMed PMID: 10332013]
[13]
Kapperud G, Lassen J, Ostroff SM, Aasen S. Clinical features of sporadic Campylobacter infections in Norway. Scandinavian journal of infectious diseases. 1992:24(6):741-9
[PubMed PMID: 1287808]
[14]
Blaser MJ. Epidemiologic and clinical features of Campylobacter jejuni infections. The Journal of infectious diseases. 1997 Dec:176 Suppl 2():S103-5
[PubMed PMID: 9396691]
[15]
Tracz DM, Keelan M, Ahmed-Bentley J, Gibreel A, Kowalewska-Grochowska K, Taylor DE. pVir and bloody diarrhea in Campylobacter jejuni enteritis. Emerging infectious diseases. 2005 Jun:11(6):838-43
[PubMed PMID: 15963277]
[16]
Platts-Mills JA, Liu J, Gratz J, Mduma E, Amour C, Swai N, Taniuchi M, Begum S, Peñataro Yori P, Tilley DH, Lee G, Shen Z, Whary MT, Fox JG, McGrath M, Kosek M, Haque R, Houpt ER. Detection of Campylobacter in stool and determination of significance by culture, enzyme immunoassay, and PCR in developing countries. Journal of clinical microbiology. 2014 Apr:52(4):1074-80. doi: 10.1128/JCM.02935-13. Epub 2014 Jan 22
[PubMed PMID: 24452175]
[17]
Bonilauri P, Bardasi L, Leonelli R, Ramini M, Luppi A, Giacometti F, Merialdi G. Detection of Food Hazards in Foods: Comparison of Real Time Polymerase Chain Reaction and Cultural Methods. Italian journal of food safety. 2016 Jan 18:5(1):5641
[PubMed PMID: 27800434]
[18]
Lin S, Wang X, Zheng H, Mao Z, Sun Y, Jiang B. Direct detection of Campylobacter jejuni in human stool samples by real-time PCR. Canadian journal of microbiology. 2008 Sep:54(9):742-7. doi: 10.1139/w08-064. Epub
[PubMed PMID: 18772937]
[19]
Bessède E, Delcamp A, Sifré E, Buissonnière A, Mégraud F. New methods for detection of campylobacters in stool samples in comparison to culture. Journal of clinical microbiology. 2011 Mar:49(3):941-4. doi: 10.1128/JCM.01489-10. Epub 2011 Jan 5
[PubMed PMID: 21209172]
[20]
Buss JE, Cresse M, Doyle S, Buchan BW, Craft DW, Young S. Campylobacter culture fails to correctly detect Campylobacter in 30% of positive patient stool specimens compared to non-cultural methods. European journal of clinical microbiology & infectious diseases : official publication of the European Society of Clinical Microbiology. 2019 Jun:38(6):1087-1093. doi: 10.1007/s10096-019-03499-x. Epub 2019 Feb 19
[PubMed PMID: 30783889]
[21]
Couturier BA, Couturier MR, Kalp KJ, Fisher MA. Detection of non-jejuni and -coli Campylobacter species from stool specimens with an immunochromatographic antigen detection assay. Journal of clinical microbiology. 2013 Jun:51(6):1935-7. doi: 10.1128/JCM.03208-12. Epub 2013 Apr 3
[PubMed PMID: 23554192]
[22]
Fitzgerald C, Patrick M, Gonzalez A, Akin J, Polage CR, Wymore K, Gillim-Ross L, Xavier K, Sadlowski J, Monahan J, Hurd S, Dahlberg S, Jerris R, Watson R, Santovenia M, Mitchell D, Harrison C, Tobin-D'Angelo M, DeMartino M, Pentella M, Razeq J, Leonard C, Jung C, Achong-Bowe R, Evans Y, Jain D, Juni B, Leano F, Robinson T, Smith K, Gittelman RM, Garrigan C, Nachamkin I. Multicenter Evaluation of Clinical Diagnostic Methods for Detection and Isolation of Campylobacter spp. from Stool. Journal of clinical microbiology. 2016 May:54(5):1209-15. doi: 10.1128/JCM.01925-15. Epub 2016 Mar 9
[PubMed PMID: 26962088]
[23]
Nolan CM, Johnson KE, Coyle MB, Faler K. Campylobacter jejuni enteritis: efficacy of antimicrobial and antimotility drugs. The American journal of gastroenterology. 1983 Oct:78(10):621-6
[PubMed PMID: 6624735]
[24]
Ruiz-Palacios GM. The health burden of Campylobacter infection and the impact of antimicrobial resistance: playing chicken. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2007 Mar 1:44(5):701-3
[PubMed PMID: 17278063]
[25]
Luangtongkum T, Jeon B, Han J, Plummer P, Logue CM, Zhang Q. Antibiotic resistance in Campylobacter: emergence, transmission and persistence. Future microbiology. 2009 Mar:4(2):189-200. doi: 10.2217/17460913.4.2.189. Epub
[PubMed PMID: 19257846]
[26]
Gupta A, Nelson JM, Barrett TJ, Tauxe RV, Rossiter SP, Friedman CR, Joyce KW, Smith KE, Jones TF, Hawkins MA, Shiferaw B, Beebe JL, Vugia DJ, Rabatsky-Ehr T, Benson JA, Root TP, Angulo FJ, NARMS Working Group. Antimicrobial resistance among Campylobacter strains, United States, 1997-2001. Emerging infectious diseases. 2004 Jun:10(6):1102-9
[PubMed PMID: 15207064]
[27]
Allos BM. Campylobacter jejuni Infections: update on emerging issues and trends. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2001 Apr 15:32(8):1201-6
[PubMed PMID: 11283810]
[28]
Rohner P, Pittet D, Pepey B, Nije-Kinge T, Auckenthaler R. Etiological agents of infectious diarrhea: implications for requests for microbial culture. Journal of clinical microbiology. 1997 Jun:35(6):1427-32
[PubMed PMID: 9163457]
[29]
Slack A. Parasitic causes of prolonged diarrhoea in travellers - diagnosis and management. Australian family physician. 2012 Oct:41(10):782-6
[PubMed PMID: 23210100]
[30]
Mee AS, Shield M, Burke M. Campylobacter colitis: differentiation from acute inflammatory bowel disease. Journal of the Royal Society of Medicine. 1985 Mar:78(3):217-23
[PubMed PMID: 3973886]
[31]
Nielsen H, Hansen KK, Gradel KO, Kristensen B, Ejlertsen T, Østergaard C, Schønheyder HC. Bacteraemia as a result of Campylobacter species: a population-based study of epidemiology and clinical risk factors. Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 2010 Jan:16(1):57-61. doi: 10.1111/j.1469-0691.2009.02900.x. Epub
[PubMed PMID: 19673969]
[32]
Pacanowski J, Lalande V, Lacombe K, Boudraa C, Lesprit P, Legrand P, Trystram D, Kassis N, Arlet G, Mainardi JL, Doucet-Populaire F, Girard PM, Meynard JL, CAMPYL Study Group. Campylobacter bacteremia: clinical features and factors associated with fatal outcome. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2008 Sep 15:47(6):790-6. doi: 10.1086/591530. Epub
[PubMed PMID: 18699745]
[33]
Tee W, Mijch A. Campylobacter jejuni bacteremia in human immunodeficiency virus (HIV)-infected and non-HIV-infected patients: comparison of clinical features and review. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 1998 Jan:26(1):91-6
[PubMed PMID: 9455515]
[34]
Inayat F, Ali NS, Riaz I, Virk HUH. From the Gut to the Heart: Campylobacter jejuni Enteritis Leading to Myopericarditis. Cureus. 2017 Jun 9:9(6):e1326. doi: 10.7759/cureus.1326. Epub 2017 Jun 9
[PubMed PMID: 28690959]
[35]
Pope JE, Krizova A, Garg AX, Thiessen-Philbrook H, Ouimet JM. Campylobacter reactive arthritis: a systematic review. Seminars in arthritis and rheumatism. 2007 Aug:37(1):48-55
[PubMed PMID: 17360026]
Level 1 (high-level) evidence
[36]
Hannu T, Mattila L, Rautelin H, Pelkonen P, Lahdenne P, Siitonen A, Leirisalo-Repo M. Campylobacter-triggered reactive arthritis: a population-based study. Rheumatology (Oxford, England). 2002 Mar:41(3):312-8
[PubMed PMID: 11934969]
[37]
Kwok M, Maurice A, Lisec C, Brown J. Campylobacter colitis: Rare cause of toxic megacolon. International journal of surgery case reports. 2016:27():141-143. doi: 10.1016/j.ijscr.2016.08.030. Epub 2016 Aug 25
[PubMed PMID: 27614338]
Level 3 (low-level) evidence
[38]
Macfarlane S, Furrie E, Macfarlane GT, Dillon JF. Microbial colonization of the upper gastrointestinal tract in patients with Barrett's esophagus. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2007 Jul 1:45(1):29-38
[PubMed PMID: 17554697]
[39]
Lecuit M, Abachin E, Martin A, Poyart C, Pochart P, Suarez F, Bengoufa D, Feuillard J, Lavergne A, Gordon JI, Berche P, Guillevin L, Lortholary O. Immunoproliferative small intestinal disease associated with Campylobacter jejuni. The New England journal of medicine. 2004 Jan 15:350(3):239-48
[PubMed PMID: 14724303]
[40]
Facciolà A, Riso R, Avventuroso E, Visalli G, Delia SA, Laganà P. Campylobacter: from microbiology to prevention. Journal of preventive medicine and hygiene. 2017 Jun:58(2):E79-E92
[PubMed PMID: 28900347]
[41]
Nyati KK, Nyati R. Role of Campylobacter jejuni infection in the pathogenesis of Guillain-Barré syndrome: an update. BioMed research international. 2013:2013():852195. doi: 10.1155/2013/852195. Epub 2013 Aug 13
[PubMed PMID: 24000328]
[42]
Lindsey RL, Gladney LM, Huang AD, Griswold T, Katz LS, Dinsmore BA, Im MS, Kucerova Z, Smith PA, Lane C, Carleton HA. Rapid identification of enteric bacteria from whole genome sequences using average nucleotide identity metrics. Frontiers in microbiology. 2023:14():1225207. doi: 10.3389/fmicb.2023.1225207. Epub 2023 Dec 14
[PubMed PMID: 38156000]