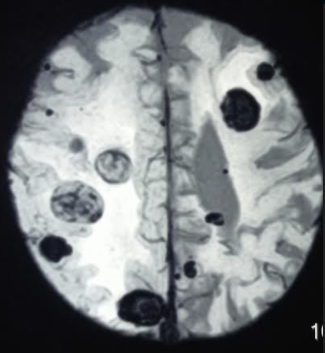
[1]
Nayak L, Lee EQ, Wen PY. Epidemiology of brain metastases. Current oncology reports. 2012 Feb:14(1):48-54. doi: 10.1007/s11912-011-0203-y. Epub
[PubMed PMID: 22012633]
[2]
Smedby KE, Brandt L, Bäcklund ML, Blomqvist P. Brain metastases admissions in Sweden between 1987 and 2006. British journal of cancer. 2009 Dec 1:101(11):1919-24. doi: 10.1038/sj.bjc.6605373. Epub 2009 Oct 13
[PubMed PMID: 19826419]
[3]
Quattrocchi CC, Errante Y, Gaudino C, Mallio CA, Giona A, Santini D, Tonini G, Zobel BB. Spatial brain distribution of intra-axial metastatic lesions in breast and lung cancer patients. Journal of neuro-oncology. 2012 Oct:110(1):79-87. doi: 10.1007/s11060-012-0937-x. Epub 2012 Jul 18
[PubMed PMID: 22802020]
[4]
Mahajan A, Ahmed S, McAleer MF, Weinberg JS, Li J, Brown P, Settle S, Prabhu SS, Lang FF, Levine N, McGovern S, Sulman E, McCutcheon IE, Azeem S, Cahill D, Tatsui C, Heimberger AB, Ferguson S, Ghia A, Demonte F, Raza S, Guha-Thakurta N, Yang J, Sawaya R, Hess KR, Rao G. Post-operative stereotactic radiosurgery versus observation for completely resected brain metastases: a single-centre, randomised, controlled, phase 3 trial. The Lancet. Oncology. 2017 Aug:18(8):1040-1048. doi: 10.1016/S1470-2045(17)30414-X. Epub 2017 Jul 4
[PubMed PMID: 28687375]
Level 1 (high-level) evidence
[5]
Brown PD, Ballman KV, Cerhan JH, Anderson SK, Carrero XW, Whitton AC, Greenspoon J, Parney IF, Laack NNI, Ashman JB, Bahary JP, Hadjipanayis CG, Urbanic JJ, Barker FG 2nd, Farace E, Khuntia D, Giannini C, Buckner JC, Galanis E, Roberge D. Postoperative stereotactic radiosurgery compared with whole brain radiotherapy for resected metastatic brain disease (NCCTG N107C/CEC·3): a multicentre, randomised, controlled, phase 3 trial. The Lancet. Oncology. 2017 Aug:18(8):1049-1060. doi: 10.1016/S1470-2045(17)30441-2. Epub 2017 Jul 4
[PubMed PMID: 28687377]
Level 1 (high-level) evidence
[6]
A Dose-Volume Response Model for Brain Metastases Treated With Frameless Single-Fraction Robotic Radiosurgery: Seeking to Better Predict Response to Treatment., Amsbaugh MJ,Yusuf MB,Gaskins J,Dragun AE,Dunlap N,Guan T,Woo S,, Technology in cancer research & treatment, 2017 Jun
[PubMed PMID: 28027696]
[7]
Chang EL, Hassenbusch SJ 3rd, Shiu AS, Lang FF, Allen PK, Sawaya R, Maor MH. The role of tumor size in the radiosurgical management of patients with ambiguous brain metastases. Neurosurgery. 2003 Aug:53(2):272-80; discussion 280-1
[PubMed PMID: 12925241]
[8]
Minniti G, Scaringi C, Paolini S, Lanzetta G, Romano A, Cicone F, Osti M, Enrici RM, Esposito V. Single-Fraction Versus Multifraction (3 × 9 Gy) Stereotactic Radiosurgery for Large (}2 cm) Brain Metastases: A Comparative Analysis of Local Control and Risk of Radiation-Induced Brain Necrosis. International journal of radiation oncology, biology, physics. 2016 Jul 15:95(4):1142-8. doi: 10.1016/j.ijrobp.2016.03.013. Epub 2016 Mar 19
[PubMed PMID: 27209508]
Level 2 (mid-level) evidence
[9]
Yamamoto M, Serizawa T, Higuchi Y, Sato Y, Kawagishi J, Yamanaka K, Shuto T, Akabane A, Jokura H, Yomo S, Nagano O, Aoyama H. A Multi-institutional Prospective Observational Study of Stereotactic Radiosurgery for Patients With Multiple Brain Metastases (JLGK0901 Study Update): Irradiation-related Complications and Long-term Maintenance of Mini-Mental State Examination Scores. International journal of radiation oncology, biology, physics. 2017 Sep 1:99(1):31-40. doi: 10.1016/j.ijrobp.2017.04.037. Epub 2017 Aug 7
[PubMed PMID: 28816158]
Level 2 (mid-level) evidence
[10]
Mulvenna P, Nankivell M, Barton R, Faivre-Finn C, Wilson P, McColl E, Moore B, Brisbane I, Ardron D, Holt T, Morgan S, Lee C, Waite K, Bayman N, Pugh C, Sydes B, Stephens R, Parmar MK, Langley RE. Dexamethasone and supportive care with or without whole brain radiotherapy in treating patients with non-small cell lung cancer with brain metastases unsuitable for resection or stereotactic radiotherapy (QUARTZ): results from a phase 3, non-inferiority, randomised trial. Lancet (London, England). 2016 Oct 22:388(10055):2004-2014. doi: 10.1016/S0140-6736(16)30825-X. Epub 2016 Sep 4
[PubMed PMID: 27604504]
Level 1 (high-level) evidence
[11]
Matsuo S, Amano T, Kawauchi S, Nakamizo A. Multiple Brain Metastases from Pancreatic Adenocarcinoma Manifesting with Simultaneous Intratumoral Hemorrhages. World neurosurgery. 2019 Mar:123():221-225. doi: 10.1016/j.wneu.2018.12.036. Epub 2018 Dec 20
[PubMed PMID: 30579022]
[12]
Rastogi K, Bhaskar S, Gupta S, Jain S, Singh D, Kumar P. Palliation of Brain Metastases: Analysis of Prognostic Factors Affecting Overall Survival. Indian journal of palliative care. 2018 Jul-Sep:24(3):308-312. doi: 10.4103/IJPC.IJPC_1_18. Epub
[PubMed PMID: 30111944]