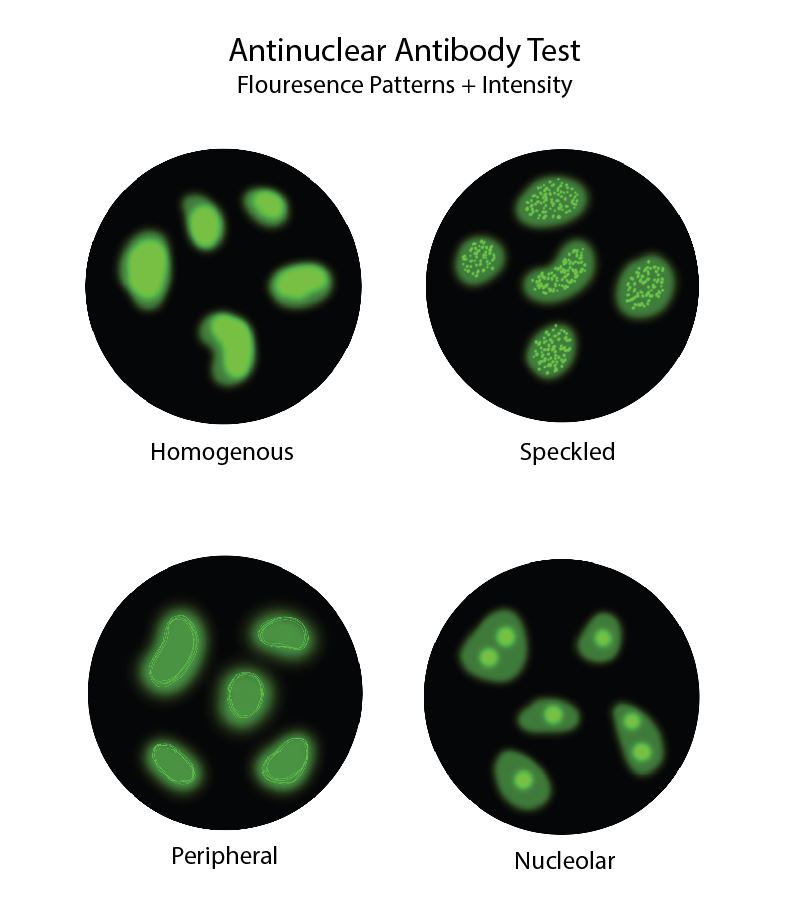
[2]
Satoh M,V�zquez-Del Mercado M,Chan EK, Clinical interpretation of antinuclear antibody tests in systemic rheumatic diseases. Modern rheumatology. 2009
[PubMed PMID: 19277826]
[3]
Kumar Y,Bhatia A,Minz RW, Antinuclear antibodies and their detection methods in diagnosis of connective tissue diseases: a journey revisited. Diagnostic pathology. 2009 Jan 2
[PubMed PMID: 19121207]
[4]
Yoshimi R,Ueda A,Ozato K,Ishigatsubo Y, Clinical and pathological roles of Ro/SSA autoantibody system. Clinical & developmental immunology. 2012
[PubMed PMID: 23304190]
[5]
Prince HE,Hogrefe WR, Evaluation of a line immunoblot assay for detection of antibodies recognizing extractable nuclear antigens. Journal of clinical laboratory analysis. 1998
[PubMed PMID: 9773966]
[6]
Tebo AE, Recent Approaches To Optimize Laboratory Assessment of Antinuclear Antibodies. Clinical and vaccine immunology : CVI. 2017 Dec
[PubMed PMID: 29021301]
[7]
Pisetsky DS,Lipsky PE, New insights into the role of antinuclear antibodies in systemic lupus erythematosus. Nature reviews. Rheumatology. 2020 Oct
[PubMed PMID: 32884126]
[8]
Dieker JW,van der Vlag J,Berden JH, Deranged removal of apoptotic cells: its role in the genesis of lupus. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association. 2004 Feb
[PubMed PMID: 14736945]
[9]
Migliorini P,Baldini C,Rocchi V,Bombardieri S, Anti-Sm and anti-RNP antibodies. Autoimmunity. 2005 Feb
[PubMed PMID: 15804705]
[10]
Gussin HA,Ignat GP,Varga J,Teodorescu M, Anti-topoisomerase I (anti-Scl-70) antibodies in patients with systemic lupus erythematosus. Arthritis and rheumatism. 2001 Feb;
[PubMed PMID: 11229469]
[11]
Powell FC,Winkelmann RK,Venencie-Lemarchand F,Spurbeck JL,Schroeter AL, The anticentromere antibody: disease specificity and clinical significance. Mayo Clinic proceedings. 1984 Oct;
[PubMed PMID: 6384675]
[12]
Zampieri S,Ghirardello A,Iaccarino L,Tarricone E,Gambari PF,Doria A, Anti-Jo-1 antibodies. Autoimmunity. 2005 Feb
[PubMed PMID: 15804708]
[13]
Fuchs G,Stein AJ,Fu C,Reinisch KM,Wolin SL, Structural and biochemical basis for misfolded RNA recognition by the Ro autoantigen. Nature structural
[PubMed PMID: 17041599]
[14]
HARGRAVES MM,RICHMOND H,MORTON R, Presentation of two bone marrow elements; the tart cell and the L.E. cell. Proceedings of the staff meetings. Mayo Clinic. 1948 Jan 21
[PubMed PMID: 18921142]
[15]
Peene I,Meheus L,Veys EM,De Keyser F, Detection and identification of antinuclear antibodies (ANA) in a large and consecutive cohort of serum samples referred for ANA testing. Annals of the rheumatic diseases. 2001 Dec
[PubMed PMID: 11709455]
[16]
Pisetsky DS, Antinuclear antibody testing - misunderstood or misbegotten? Nature reviews. Rheumatology. 2017 Aug
[PubMed PMID: 28541299]
[17]
Olsen NJ,Choi MY,Fritzler MJ, Emerging technologies in autoantibody testing for rheumatic diseases. Arthritis research
[PubMed PMID: 28738887]
[18]
Satoh M,Tanaka S,Chan EK, The uses and misuses of multiplex autoantibody assays in systemic autoimmune rheumatic diseases. Frontiers in immunology. 2015
[PubMed PMID: 25954274]
[20]
Petri M,Orbai AM,Alarc�n GS,Gordon C,Merrill JT,Fortin PR,Bruce IN,Isenberg D,Wallace DJ,Nived O,Sturfelt G,Ramsey-Goldman R,Bae SC,Hanly JG,S�nchez-Guerrero J,Clarke A,Aranow C,Manzi S,Urowitz M,Gladman D,Kalunian K,Costner M,Werth VP,Zoma A,Bernatsky S,Ruiz-Irastorza G,Khamashta MA,Jacobsen S,Buyon JP,Maddison P,Dooley MA,van Vollenhoven RF,Ginzler E,Stoll T,Peschken C,Jorizzo JL,Callen JP,Lim SS,Fessler BJ,Inanc M,Kamen DL,Rahman A,Steinsson K,Franks AG Jr,Sigler L,Hameed S,Fang H,Pham N,Brey R,Weisman MH,McGwin G Jr,Magder LS, Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis and rheumatism. 2012 Aug
[PubMed PMID: 22553077]
Level 1 (high-level) evidence
[21]
Sobolewski P,Maślińska M,Wieczorek M,Łagun Z,Malewska A,Roszkiewicz M,Nitskovich R,Szymańska E,Walecka I, Systemic sclerosis - multidisciplinary disease: clinical features and treatment. Reumatologia. 2019
[PubMed PMID: 31548749]
[22]
Bent MA,Varacallo M,Fox EJ,Voss S,Frauenhoffer EE, Lipoma Arborescens and Coexisting Psoriatic Arthritis: A Case Report and Review of the Literature. JBJS case connector. 2013 Oct-Dec
[PubMed PMID: 29252521]
Level 3 (low-level) evidence
[23]
Silvy F,Bertin D,Bardin N,Auger I,Guzian MC,Mattei JP,Guis S,Roudier J,Balandraud N, Antinuclear Antibodies in Patients with Psoriatic Arthritis Treated or Not with Biologics. PloS one. 2015
[PubMed PMID: 26230924]