Continuing Education Activity
This activity reviews the presurgical cardiac risk assessment and highlights the role of the interprofessional team in optimizing presurgical fitness and management of patients and enhancing postsurgical outcomes.
Objectives:
Identify risk factors for perioperative adverse cardiac events.
Summarize preoperative cardiac risk using the American College of Cardiology/American Heart Association algorithm.
Outline situations when elective surgeries should be delayed.
Explain the importance of the interprofessional team in communicating with patients with cardiac disease to optimize presurgical fitness and in turn enhance postsurgical outcomes.
Introduction
Many patients undergoing major noncardiac as well as vascular surgery are at risk for a major adverse cardiac event (MACE). In the United States, 27 million patients undergo non-cardiac surgery annually, 50,000 of which suffer a postoperative myocardial infarction (MI). It's estimated that cardiovascular complications are responsible for roughly one-half of all the mortality experienced by patients undergoing noncardiac surgery and occur in 1% to 5% of patients undergoing vascular surgery.[1] To minimize perioperative complications and reduce morbidity and mortality, a thorough preoperative evaluation of patients with heart disease with appropriate risk stratification is necessary.[2] The American College of Cardiology (ACC) and the American Heart Association (AHH) have collaborated to develop a set of clinical practice guidelines using scientific evidence to standardize the preoperative cardiac evaluation of patients undergoing non-cardiac surgery. Recommendations have their basis on a known history of or risk factors for cardiac disease, the patient’s functional status, as well as the urgency of the procedure. The resulting individualized cardiac risk assessment reduces the risk of MACE, as it determines the need for further optimization of the patient with diagnostic testing, medical therapy, and even coronary interventions.[3]
Anatomy and Physiology
When it comes to preoperative cardiac risk assessment, the essential anatomical considerations present on the patient’s physical exam. A thorough exam, along with non-invasive testing can provide information on three cardiac risk markers: LV dysfunction, myocardial ischemia, and heart valve abnormalities, all of which, are significant determinants of adverse postoperative outcome.[3]
Objective examination includes the evaluation of vital signs such as blood pressure, heart rate, respiratory rate, temperature and oxygen saturation, as well as height, weight, and body mass index (BMI).
The physical examination focuses on the cardiovascular and pulmonary systems and includes auscultation of the heart and lungs, inspection of arterial pulses and veins, abdominal palpation and examination of the extremities for edema and vascular integrity.
Critical physical findings include third or fourth heart sounds, rales, jugular venous distention, ascites, hepatomegaly, and dependent edema, which could be evidence of heart failure or a murmur suspicious for hemodynamically significant valvular heart disease.
Indications
Even though heart disease may occur for various reasons in younger age groups, perioperative cardiac mortality and morbidity are predominantly an issue in the adult population undergoing major non-cardiac surgery.[3] Canadian Cardiovascular Society (CCS) Guidelines recommend cardiac risk assessment in patients over the age of 45 or patients 18 to 44 with significant cardiovascular disease.[4]
Risk Factors
Clinical risk factors for a perioperative major adverse cardiac event (MACE) include the following[5]:
- Reduced functional status (< 4 METs)
- Ischemic heart disease (history of MI, angina pectoris, etc.)
- Heart failure
- Cardiomyopathy
- Severe valvular heart disease (severe aortic stenosis, symptomatic mitral regurgitation)
- Significant arrhythmias (Mobitz II AV block, 3rd-degree block, symptomatic ventricular arrhythmia, symptomatic bradycardia, newly recognized ventricular tachycardia)
- Chronic renal failure
- History of cerebrovascular accident or transient ischemic attack
- Diabetes mellitus requiring Insulin
- Chronic pulmonary dysfunction
- Obesity
- Anemia
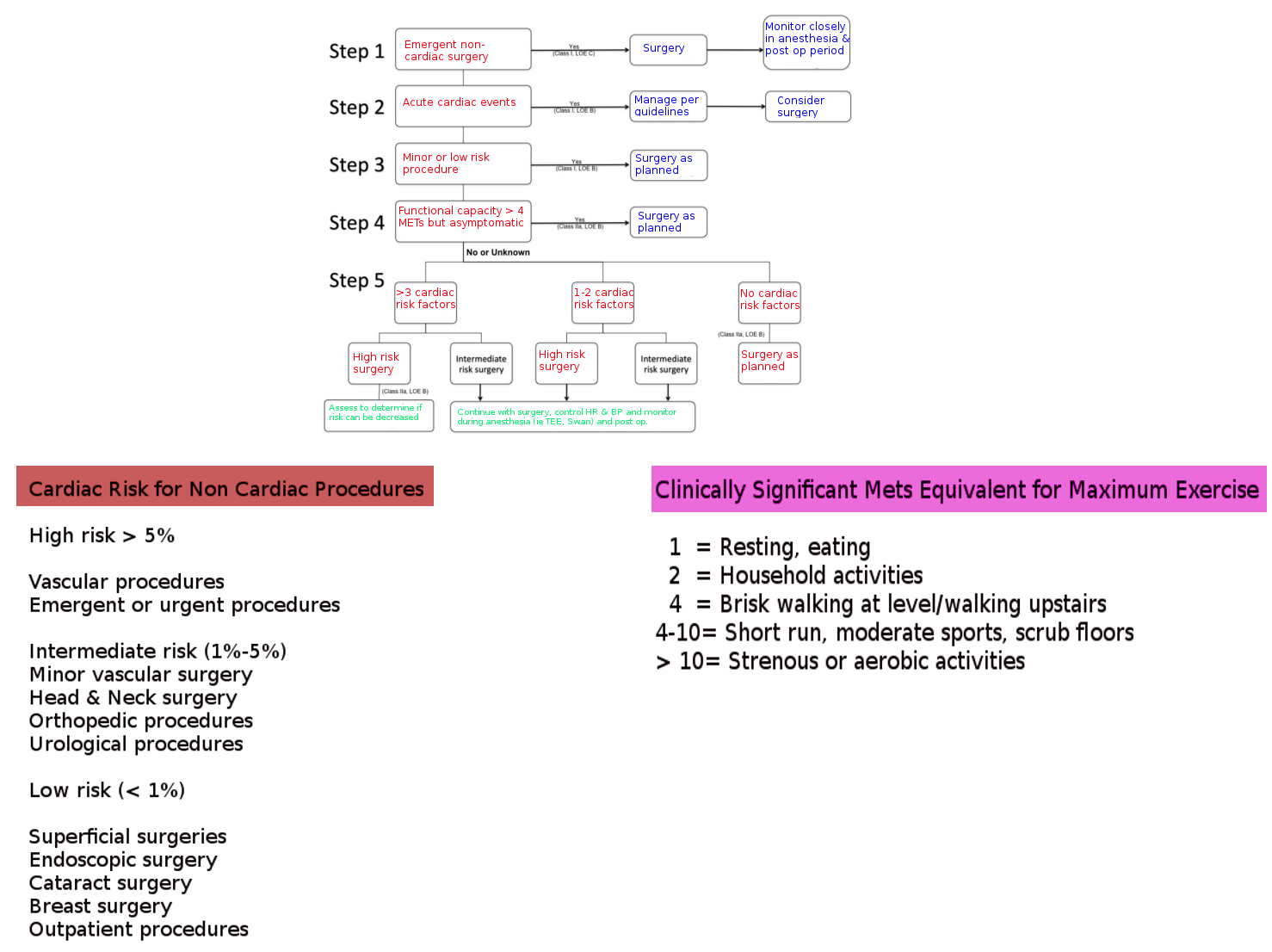
Following a thorough physical exam and evaluation of risk factors, patients undergoing surgery should undergo assessment for additional preoperative interventions by using the American College of Cardiology/American Heart Association algorithm. The guidelines provide a template for assessing cardiac risk, by taking into account both the patient’s condition and the extent of the surgery. It is followed in a stepwise fashion and stops at the first point that applies to the patient.[2] Please see the attached image for the algorithm.
Step 1 in the algorithm considers the urgency of surgery.
Step 2 focuses on active cardiac conditions such as acute MI, unstable or severe angina, decompensated heart failure, severe valvular disease, or significant arrhythmias. Active cardiac conditions warrant postponement of surgery for all except lifesaving emergency procedures.
Step 3 considers the surgical risk or severity. Surgical interventions divide into low-risk, intermediate-risk, and high-risk groups with estimated 30-day cardiac event rates (cardiac death and MI) of <1, 1–5, and >5%, respectively. Please see the attached image for a more detailed division of surgical risk.
Step 4 assesses the patient’s functional capacity based on Metabolic Equivalents (METs). One MET equals the basal metabolic rate. Exercise testing provides an objective assessment of functional capacity. Without testing, functional capacity is estimated by the ability to perform the activities of daily living. Please see the attached image for examples of Metabolic Equivalents of functional capacity.
Step 5 considers patients who have an inadequate or indeterminate functional capacity and need intermediate-risk or vascular surgery. The number of patient-level clinical predictors from the RCRI (CAD, heart failure, cerebrovascular disease, diabetes, renal insufficiency) determines the likely benefit of further cardiac testing among individuals who reach step 5. Patients with no clinical predictors proceed to surgery. Those with risk predictors may benefit from further testing, but only if the results alter management.
Contraindications
Emergency Surgery
Patients who require emergent (<6 hours) or urgent (6 to 24) surgery are not able to undergo a preoperative cardiac workup as the time lost to complete the workup could be detrimental to the overall outcome if it delays surgery.[2][6] Due to the urgent or emergent need for surgery, these patients are at an increased risk of a perioperative cardiovascular event, regardless of baseline risk. In these cases, perioperative surveillance for cardiac events is key, as well as the continuation of chronic cardiovascular medical therapy.[3]
High-risk Patients
Patients with a recent history of cardiac disease, uncontrolled comorbidities or recent interventions have specific guidelines set by the ACC/AHA for subsequently undergoing surgery.
Hypertension
When evaluating a hypertensive patient in the preoperative setting, it is imperative to assess not only the patient’s current blood pressure but their baseline pressure before surgery. This can be accomplished by obtaining a thorough history as well as looking through medical records for information on their anti-hypertensive regimen as well as the adequacy of blood pressure control. The assessment should also search for evidence of chronic or acute end-organ damage.[3]
Continuation of antihypertensive therapy on the day of surgery is a topic of debate. Observational studies have demonstrated more frequent episodes of transient intraoperative hypotension if angiotensin-converting enzyme inhibitors or diuretics are taken the same day of surgery. Patients who have discontinued therapy the morning of surgery have shown no major adverse outcome. If discontinued, they should restart them promptly in the post-operative period.[7] Additionally, there have been multiple studies assessing the utility of beta-blockers in the perioperative period, with the consensus that beta-blockers should be continued only in patients taking them chronically. The POISE (Perioperative Ischemic Evaluation Study) trial demonstrated that starting them in a beta-blocker naïve patient can result in a higher incidence of hypotension, stroke, and death from non-cardiac complications.[7]
The major limitation in hypertensive patients is in patients with grade 3 hypertension (systolic blood pressure =180 mmHg and/or diastolic blood pressure =110 mmHg). In these cases, the potential benefits of delaying surgery to optimize medical therapy should be weighed against the risk of delaying the surgical procedure.[3]
Acute Coronary Syndrome
Patients with recent myocardial infarction(MI) or unstable angina are at higher risk for developing a major adverse cardiac event. To reduce the risk of incurring a MACE, the American College of Cardiology (ACC) recommends waiting a minimum of 4-6 weeks after an MI before proceeding with elective surgery.[8] There is a substantial decrease in 30-day mortality as the length of time from MI to operation increases.[7] If the time between the initial MI and surgery is less than 30 days, the risk of developing an adverse cardiac event is 32.8%. If surgery takes place between 31 and 60 days, the risk is 18.7%, and after 60 days, it declines to less than 10%.[6]
Heart Failure
The prevalence of heart failure (HF) in the United States is currently 5.7 million and continues to increase. Heart failure is an important predictor of a major adverse cardiac event and has a higher 30-day postoperative mortality rate (9.2 to 9.3%) when compared to those with CAD (2.9%).[7] The risk elevates when HF is decompensated or is worsening. Perioperative mortality increases to 13% when patients have an exacerbation of HF within 4 weeks of intended surgery. Using the New York Heart Association (NYHA) categories to assess the severity of heart failure can be useful in determining if a patient requires an echocardiogram (Class III and IV) preoperatively to assess left ventricular function, or further evaluation by a cardiologist. Optimization of fluid status and improvement of symptoms prior to surgery can improve outcomes.[6]
Valvular Disease
Aortic Stenosis
Another key component of the cardiac risk assessment is the evaluation of patients with underlying aortic stenosis (AS). Since patients with moderate to severe aortic stenosis have been found to have double the rate of 30-day mortality and can suffer from postoperative MI 3 times more frequently than patients without AS, preoperative determination of the degree of stenosis, as well as the presence of symptoms, is critical.[3][7]. This is achievable through a thorough review of symptoms as well as reviewing or obtaining an echocardiogram performed within 1 year of surgery or more recently if there is a change in clinical status.[6][7]
Patients with severe aortic stenosis that require an urgent procedure will require invasive hemodynamic monitoring intraoperatively.[7] If the surgery is elective, the presence of symptoms will dictate management.[3]
Asymptomatic patients can proceed with low to intermediate risk surgery, and invasive hemodynamic monitoring is a consideration. If the surgery is high risk, the patient should undergo evaluation for the possibility of an aortic valve replacement (AVR), or if ineligible for AVR, can proceed with invasive hemodynamic monitoring.[3]
In symptomatic patients or those with hemodynamically significant AS, AVR should be performed prior to surgery. If patients are not a candidate for AVR or refuse, and the noncardiac surgery is essential, then there are a few options. First, the surgery could proceed under invasive hemodynamic monitoring with adjustment of the anesthetic plan to prevent a significant drop in preload to maintain adequate cardiac output.[7] In this case, the risk of developing a MACE is 10 to 20%.[6] Second, patients could be evaluated for a transcatheter aortic valve replacement (TAVR), as this procedure has been shown to decrease the 1-year post-operative mortality rate. Finally, patients could undergo percutaneous balloon valvuloplasty, which is less optimal as recurrence and mortality rates approach 50% within 6 months.[3][7]
Mitral Stenosis
Patients with mitral stenosis are also at increased risk for a MACE when undergoing elective noncardiac surgery.[7] If they have non-significant mitral stenosis or have severe mitral stenosis but are asymptomatic, they can proceed with low to intermediate risk surgery without further intervention.[3] If the mitral stenosis is severe, they should be managed similarly to those with aortic stenosis,[7] with special care when it comes to intraoperative fluid management and maintaining cardiac output, as large increases in left atrial and pulmonary capillary wedge pressure can lead to pulmonary edema.[7]
Aortic and Mitral Regurgitation
Nonsignificant aortic and mitral regurgitation (AR and MR) do not correlate with an increased risk of developing a MACE in the perioperative period. Those with moderate to severe AR and MR with preserved left ventricle function can proceed with noncardiac surgery without intervention.[3] Patients who are symptomatic or have a decreased left ventricular ejection fraction (<35%), are at high risk of developing a cardiovascular complication. These surgeries should only be performed if essential and should include invasive hemodynamic monitoring, echocardiography, and admittance to an intensive care unit postoperatively.[3][7] Optimization of pharmacologic therapy to achieve maximum hemodynamic stability before high-risk surgery can also be beneficial in improving postoperative outcomes.[3]
Percutaneous Coronary Intervention
Although several patients presenting for elective surgery have a history of coronary artery disease, preoperative coronary angiography with revascularization is rarely indicated.[9] Data demonstrably shows the procedural risk associated with PCI as well as the lack of evidence supporting improved perioperative outcomes outweighs any benefit from the procedure. According to the 2011 coronary artery bypass grafting (CABG) and percutaneous coronary intervention (PCI) guidelines, indications for PCI are the same as in a non-surgical setting, and should only occur in the presence of acute coronary syndrome or when there is evidence of ischemia on non-invasive testing.[7]
In the event of stent placement, patients are required to comply with dual antiplatelet therapy (DAPT) with aspirin and a platelet receptor inhibitor, to prevent stent thrombosis. Discontinuing DAPT is one of the strongest risk factors when it comes to stent thrombosis and mortality.[7] Also, the newly exposed coronary arteriolar endothelium, combined with the physiological stress of surgery, places patients in a hypercoagulable state, which further increases the risk of stent thrombosis.[10]
To maximize DAPT, the 2016 ACC/AHA guidelines recommend patients treated with balloon angioplasty, a bare metal stent, or a drug-eluting stent, should postpone elective noncardiac surgery for 14 days, 30 days and 3 - 12 months, respectively.[9][7]
After the appropriate timeframe, the platelet receptor inhibitor may be held perioperatively due to bleeding risk. In that case, guidelines recommend continuation of aspirin.[7]
Coronary Artery Bypass Grafting
Similar to coronary angiography, patients should not prophylactically undergo coronary artery bypass grafting (CABG), as there is no increase in perioperative outcomes.[10] The ACC/AHA guideline recommendations are that patients who are revascularized by CABG within the past 5 years can proceed with noncardiac surgery without further intervention.[10] According to the Bypass Angioplasty Revascularization Investigation (BARI) trial, the determination is that these patients, as well as those who have undergone PCI, have a lower risk of developing postoperative cardiac complications when compared to those who have not been revascularized.
Equipment
Pacemaker
Preoperatively, it is imperative to understand the indication for the device, battery status, and current settings. Patients undergoing surgery who have a permanent pacemaker may experience interference with function due to use of intraoperative electrocautery.[3][9] This can be avoided by strategically positioning the ground plate for the electrical circuit so that the current travels away from the pacemaker. Additionally, using brief bursts and using a low amplitude may decrease interference.[3] In pacemaker-dependent patients, a magnet can also be placed on the skin over the pacemaker to set it to an asynchronous mode.[3] In cases of magnet placement, the pacemaker must be interrogated after surgery, to ensure appropriate pacing.
Automatic Implantable Cardioverter Defibrillator (AICD)
Patients with an AICD who are undergoing surgery require deactivation of the device to avoid accidental defibrillation as a result of interference from electrocautery.[3] Placement of a magnet over an AICD disables its ability to defibrillate and converts it to pacemaker mode.
During the period of deactivation, the patient's electrocardiogram requires constant monitoring and the ability for prompt external cardioversion must be available.[9] All AICDs must be reactivated following surgery.
Preparation
A significant component of cardiac risk assessment is preoperative testing. There are specific evidence-based guidelines regarding when to obtain non-invasive testing in patients before undergoing surgery.
Electrocardiogram
A preoperative electrocardiogram (ECG) is a valuable tool to assess for the possibility of myocardial ischemia, left ventricular hypertrophy, conduction blocks or other arrhythmias.
According to the 2009 ACC/AHA guidelines, an ECG should/should not be obtained in the following situations[3]:
- Pre-operative ECG should be obtained in patients who have risk factors and are scheduled for intermediate- or high-risk surgery
- Pre-operative ECG should be considered for patients who have risk factor(s) and are scheduled for low-risk surgery
- Pre-operative ECG may be considered for patients who have no risk factors and are scheduled for intermediate-risk surgery
- Pre-operative ECG is not recommended for patients who have no risk factor and are scheduled for low-risk surgery
Echocardiography
Echocardiography is not a routine recommendation for all patients undergoing noncardiac surgery. The current ACCF/AHA guidelines recommend a preoperative echocardiogram in patients with dyspnea of unknown origin or recent exacerbation of heart failure, as well as to assess valvular function in patients with a murmur,[7] as these conditions correlate with postoperative cardiac complications.
An echocardiogram is also recommended in patients with a known severe valvular disorder to assess severity and in patients with valvular disorders undergoing intermediate high-risk surgery. It is a consideration in patients undergoing high-risk surgery but is not a recommendation for patients undergoing low to intermediate-risk surgery or in asymptomatic patients.[3]
Non-invasive Stress Testing
Although moderate to large areas of inducible ischemia on noninvasive stress testing have been shown to be a predictor of a major adverse cardiac event,[6] there is no indication for stress testing as a routine part of the preoperative evaluation. Based on the ACC/AHA class IIa recommendations, physicians can consider noninvasive stress testing in patients undergoing high-risk surgery, with reduced or unknown functional capacity, if there is clinical suspicion that it may change management.[6] For these patients, either exercise-induced or pharmacological induced stress testing is appropriate.[7]
Technique or Treatment
Evolution of Cardiac Risk Indices
In 1977, Goldman et al. published the landmark article on cardiac risk index (CRI) which used clinical risk factors to quantify the risk of developing perioperative cardiac complications. These factors include:
- Preoperative third heart sound or jugular venous distention indicating active heart failure
- Myocardial infarction in the past six months
- Premature ventricular complexes/min before surgery
- Cardiac rhythm other than sinus
- Age of greater than or equal to 70 years
- Emergency surgery, significant aortic stenosis
- Intraperitoneal, intrathoracic, or aortic surgery
- Markers of a poor general medical condition such as renal dysfunction, liver disease, lung disease, and electrolyte imbalance
Each of these factors was assigned a point value, and a point system was developed to determine the risk of developing a MACE.
In 1986, Detsky et al. proposed a modified CRI, updating Goldman’s approach by modifying the risk factors and adding angina.[2]
In 1996, the ACC/AHA Taskforce published Practice Guidelines for Perioperative Cardiovascular Evaluation for Noncardiac Surgery and then came out with an update in 2002.[2]
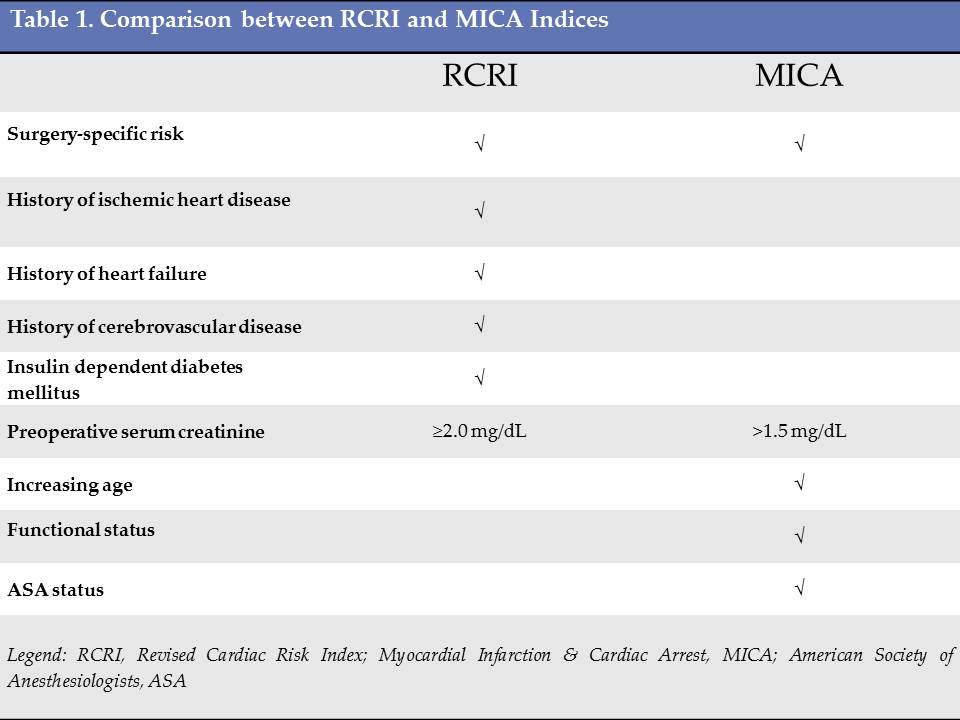
According to the guidelines, there are currently three acceptable risk calculators for assessment of the perioperative risk of MACE: the Revised Cardiac Risk Index (RCRI), the National Surgical Quality Improvement Program Myocardial Infarction and Cardiac Arrest (NSQIP MICA) calculator and the National Surgical Quality Improvement Program risk calculator).[7]
RCRI
The Revised Cardiac Risk Index (RCRI) is developed by Lee et al. in 1999 as a revision of Goldman’s cardiac risk index. It is the most commonly used tool in clinical practice due to its ease of use and reliability. The RCRI uses six clinical risk predictors, each of which are assigned equal weight. These predictors include[6]:
- Coronary heart disease
- Congestive heart failure
- Cerebrovascular disease
- Insulin-dependent diabetes mellitus
- Renal insufficiency with serum creatinine measuring greater than 2 mg/dL
- High-risk surgery (suprainguinal vascular, intraperitoneal, or intrathoracic)
The previously determined risks of MACE determined by Lee, were found to under report it's occurrence. Multiple studies systematically reviewed the data and determined new values. As of January 2019, patients with a score of 0= 3.9% risk of MACE, a score of 1= 6.0% risk of MACE, a score of 2=10.1% risk of MACE and a score of 3 or greater indicate a greater than 15% risk of MACE.[4][11] Patients with at least two predictors of risks would be considered to have an elevated risk of MACE.[6] MACE, in this case, is defined as death, MI or cardiac arrest.
NSQIP MICA
In 2007, Gupta and colleagues used data collected by the National Surgical Quality Improvement Program to create a risk-prediction model using data from 525 US hospitals to predict the risk of MI and cardiac arrest.[7] This model uses the type of surgery, functional status, creatinine concentration, American Society of Anesthesiologists’ physical status classification, and age as predictors. The NSQIP MICA model was found to have better discrimination over the RCRI.[9]
NSQI Surgical Risk Calculator
In 2013, the NSQIP Surgical Risk Calculator was created to provide procedure-specific risk. It is an online calculator which uses 21 patient-specific variables and calculates the risk of developing MI or cardiac arrest.[7]
Clinical Significance
A thorough cardiac risk assessment is essential for patients undergoing non-cardiac surgery, due to the high mortality rate (32-69%) of patients who experience a perioperative MI.[1] Identifying these patients in advance allows for optimization with tailored medical therapy and adjustment of anesthetic and surgical plan, to minimize physiological stress.
Enhancing Healthcare Team Outcomes
To ensure an optimal perioperative course, a unified team approach with close cooperation between all physicians involved (surgeon, anesthesiologist, cardiologist, pulmonologist, primary care) is essential.[3] Despite many advances in evidence-based guidelines, cardiac complications after noncardiac surgery remain a substantial problem, and physicians work together to employ all evidence-based tools available to reduce morbidity and mortality and enhance patient outcomes.[4]