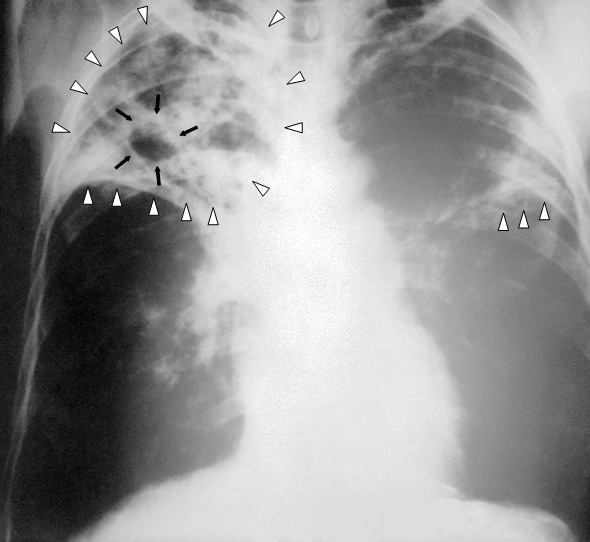
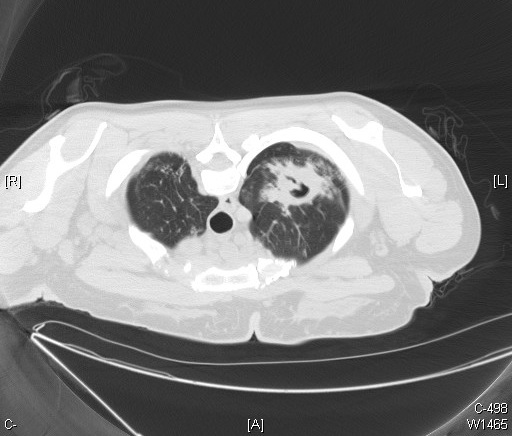
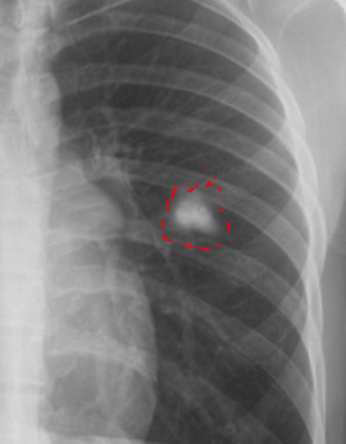
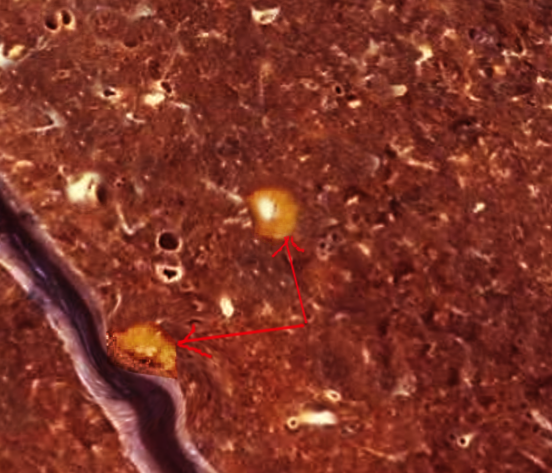
[1]
Donald PR, Diacon AH, Thee S. Anton Ghon and His Colleagues and Their Studies of the Primary Focus and Complex of Tuberculosis Infection and Their Relevance for the Twenty-First Century. Respiration; international review of thoracic diseases. 2021:100(7):557-567. doi: 10.1159/000509522. Epub 2020 Dec 15
[PubMed PMID: 33321506]
[2]
Lyon SM, Rossman MD. Pulmonary Tuberculosis. Microbiology spectrum. 2017 Jan:5(1):. doi: 10.1128/microbiolspec.TNMI7-0032-2016. Epub
[PubMed PMID: 28185620]
[3]
Concepcion NDP, Laya BF, Andronikou S, Daltro PAN, Sanchez MO, Uy JAU, Lim TRU. Standardized radiographic interpretation of thoracic tuberculosis in children. Pediatric radiology. 2017 Sep:47(10):1237-1248. doi: 10.1007/s00247-017-3868-z. Epub 2017 Aug 29
[PubMed PMID: 29052771]
[4]
Stephenson L, Byard RW. An atlas overview of characteristic features of tuberculosis that may be encountered at autopsy. Forensic science, medicine, and pathology. 2020 Mar:16(1):143-151. doi: 10.1007/s12024-019-00161-y. Epub 2019 Aug 30
[PubMed PMID: 31471869]
Level 3 (low-level) evidence
[5]
Skoura E, Zumla A, Bomanji J. Imaging in tuberculosis. International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases. 2015 Mar:32():87-93. doi: 10.1016/j.ijid.2014.12.007. Epub
[PubMed PMID: 25809762]
[6]
Bell LCK, Noursadeghi M. Pathogenesis of HIV-1 and Mycobacterium tuberculosis co-infection. Nature reviews. Microbiology. 2018 Feb:16(2):80-90. doi: 10.1038/nrmicro.2017.128. Epub 2017 Nov 7
[PubMed PMID: 29109555]
[7]
de Campos EP, Bertoli CJ, Barbosa KS. [Pulmonary lymph node in acute juvenile paracoccidioidomycosis (a case report)]. Revista da Sociedade Brasileira de Medicina Tropical. 1992 Jul-Sep:25(3):195-200
[PubMed PMID: 1308953]
Level 3 (low-level) evidence
[8]
Glaziou P, Floyd K, Raviglione MC. Global Epidemiology of Tuberculosis. Seminars in respiratory and critical care medicine. 2018 Jun:39(3):271-285. doi: 10.1055/s-0038-1651492. Epub 2018 Aug 2
[PubMed PMID: 30071543]
[9]
Suárez I, Fünger SM, Kröger S, Rademacher J, Fätkenheuer G, Rybniker J. The Diagnosis and Treatment of Tuberculosis. Deutsches Arzteblatt international. 2019 Oct 25:116(43):729-735. doi: 10.3238/arztebl.2019.0729. Epub
[PubMed PMID: 31755407]
[10]
Floyd K, Glaziou P, Zumla A, Raviglione M. The global tuberculosis epidemic and progress in care, prevention, and research: an overview in year 3 of the End TB era. The Lancet. Respiratory medicine. 2018 Apr:6(4):299-314. doi: 10.1016/S2213-2600(18)30057-2. Epub
[PubMed PMID: 29595511]
Level 3 (low-level) evidence
[11]
Vynnycky E, Fine PE. Lifetime risks, incubation period, and serial interval of tuberculosis. American journal of epidemiology. 2000 Aug 1:152(3):247-63
[PubMed PMID: 10933272]
[12]
Lönnroth K, Raviglione M. The WHO's new End TB Strategy in the post-2015 era of the Sustainable Development Goals. Transactions of the Royal Society of Tropical Medicine and Hygiene. 2016 Mar:110(3):148-50. doi: 10.1093/trstmh/trv108. Epub
[PubMed PMID: 26884490]
[13]
Chakaya J, Petersen E, Nantanda R, Mungai BN, Migliori GB, Amanullah F, Lungu P, Ntoumi F, Kumarasamy N, Maeurer M, Zumla A. The WHO Global Tuberculosis 2021 Report - not so good news and turning the tide back to End TB. International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases. 2022 Nov:124 Suppl 1():S26-S29. doi: 10.1016/j.ijid.2022.03.011. Epub 2022 Mar 20
[PubMed PMID: 35321845]
[14]
Furin J, Cox H, Pai M. Tuberculosis. Lancet (London, England). 2019 Apr 20:393(10181):1642-1656. doi: 10.1016/S0140-6736(19)30308-3. Epub 2019 Mar 20
[PubMed PMID: 30904262]
[15]
Datta S, Shah L, Gilman RH, Evans CA. Comparison of sputum collection methods for tuberculosis diagnosis: a systematic review and pairwise and network meta-analysis. The Lancet. Global health. 2017 Aug:5(8):e760-e771. doi: 10.1016/S2214-109X(17)30201-2. Epub 2017 Jun 15
[PubMed PMID: 28625793]
Level 1 (high-level) evidence
[16]
Nicol MP, Zar HJ. Advances in the diagnosis of pulmonary tuberculosis in children. Paediatric respiratory reviews. 2020 Nov:36():52-56. doi: 10.1016/j.prrv.2020.05.003. Epub 2020 May 25
[PubMed PMID: 32624357]
Level 3 (low-level) evidence
[17]
Stockdale AJ, Duke T, Graham S, Kelly J. Evidence behind the WHO guidelines: hospital care for children: what is the diagnostic accuracy of gastric aspiration for the diagnosis of tuberculosis in children? Journal of tropical pediatrics. 2010 Oct:56(5):291-8. doi: 10.1093/tropej/fmq081. Epub 2010 Sep 3
[PubMed PMID: 20817689]
[18]
Shaw JA, Irusen EM, Diacon AH, Koegelenberg CF. Pleural tuberculosis: A concise clinical review. The clinical respiratory journal. 2018 May:12(5):1779-1786. doi: 10.1111/crj.12900. Epub
[PubMed PMID: 29660258]
[19]
Schito M, Migliori GB, Fletcher HA, McNerney R, Centis R, D'Ambrosio L, Bates M, Kibiki G, Kapata N, Corrah T, Bomanji J, Vilaplana C, Johnson D, Mwaba P, Maeurer M, Zumla A. Perspectives on Advances in Tuberculosis Diagnostics, Drugs, and Vaccines. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2015 Oct 15:61Suppl 3(Suppl 3):S102-18. doi: 10.1093/cid/civ609. Epub
[PubMed PMID: 26409271]
Level 3 (low-level) evidence
[20]
Burrill J, Williams CJ, Bain G, Conder G, Hine AL, Misra RR. Tuberculosis: a radiologic review. Radiographics : a review publication of the Radiological Society of North America, Inc. 2007 Sep-Oct:27(5):1255-73
[PubMed PMID: 17848689]
[21]
Woodring JH, Vandiviere HM, Fried AM, Dillon ML, Williams TD, Melvin IG. Update: the radiographic features of pulmonary tuberculosis. AJR. American journal of roentgenology. 1986 Mar:146(3):497-506
[PubMed PMID: 3484866]
[22]
Rodriguez-Takeuchi SY, Renjifo ME, Medina FJ. Extrapulmonary Tuberculosis: Pathophysiology and Imaging Findings. Radiographics : a review publication of the Radiological Society of North America, Inc. 2019 Nov-Dec:39(7):2023-2037. doi: 10.1148/rg.2019190109. Epub
[PubMed PMID: 31697616]
[23]
Wright A, Zignol M, Van Deun A, Falzon D, Gerdes SR, Feldman K, Hoffner S, Drobniewski F, Barrera L, van Soolingen D, Boulabhal F, Paramasivan CN, Kam KM, Mitarai S, Nunn P, Raviglione M, Global Project on Anti-Tuberculosis Drug Resistance Surveillance. Epidemiology of antituberculosis drug resistance 2002-07: an updated analysis of the Global Project on Anti-Tuberculosis Drug Resistance Surveillance. Lancet (London, England). 2009 May 30:373(9678):1861-73. doi: 10.1016/S0140-6736(09)60331-7. Epub 2009 Apr 15
[PubMed PMID: 19375159]
[24]
Caminero JA, Cayla JA, García-García JM, García-Pérez FJ, Palacios JJ, Ruiz-Manzano J. Diagnosis and Treatment of Drug-Resistant Tuberculosis. Archivos de bronconeumologia. 2017 Sep:53(9):501-509. doi: 10.1016/j.arbres.2017.02.006. Epub 2017 Mar 27
[PubMed PMID: 28359606]
[25]
Joshi R, Reingold AL, Menzies D, Pai M. Tuberculosis among health-care workers in low- and middle-income countries: a systematic review. PLoS medicine. 2006 Dec:3(12):e494
[PubMed PMID: 17194191]
Level 1 (high-level) evidence
[26]
McGowan JE Jr. Nosocomial tuberculosis: new progress in control and prevention. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 1995 Sep:21(3):489-505
[PubMed PMID: 8527533]
[27]
Patterson B, Wood R. Is cough really necessary for TB transmission? Tuberculosis (Edinburgh, Scotland). 2019 Jul:117():31-35. doi: 10.1016/j.tube.2019.05.003. Epub 2019 May 28
[PubMed PMID: 31378265]
[29]
Lönnroth K, Jaramillo E, Williams BG, Dye C, Raviglione M. Drivers of tuberculosis epidemics: the role of risk factors and social determinants. Social science & medicine (1982). 2009 Jun:68(12):2240-6. doi: 10.1016/j.socscimed.2009.03.041. Epub 2009 Apr 23
[PubMed PMID: 19394122]
[30]
Carlsson M, Johansson S, Eale RP, Kaboru BB. Nurses' Roles and Experiences with Enhancing Adherence to Tuberculosis Treatment among Patients in Burundi: A Qualitative Study. Tuberculosis research and treatment. 2014:2014():984218. doi: 10.1155/2014/984218. Epub 2014 Aug 19
[PubMed PMID: 25215232]
Level 2 (mid-level) evidence
[31]
Palacios E, Guerra D, Llaro K, Chalco K, Sapag R, Furin J. The role of the nurse in the community-based treatment of multidrug-resistant tuberculosis (MDR-TB). The international journal of tuberculosis and lung disease : the official journal of the International Union against Tuberculosis and Lung Disease. 2003 Apr:7(4):343-6
[PubMed PMID: 12729339]
[32]
Karumbi J, Garner P. Directly observed therapy for treating tuberculosis. The Cochrane database of systematic reviews. 2015 May 29:2015(5):CD003343. doi: 10.1002/14651858.CD003343.pub4. Epub 2015 May 29
[PubMed PMID: 26022367]
Level 1 (high-level) evidence
[33]
Nahid P, Dorman SE, Alipanah N, Barry PM, Brozek JL, Cattamanchi A, Chaisson LH, Chaisson RE, Daley CL, Grzemska M, Higashi JM, Ho CS, Hopewell PC, Keshavjee SA, Lienhardt C, Menzies R, Merrifield C, Narita M, O'Brien R, Peloquin CA, Raftery A, Saukkonen J, Schaaf HS, Sotgiu G, Starke JR, Migliori GB, Vernon A. Executive Summary: Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines: Treatment of Drug-Susceptible Tuberculosis. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2016 Oct 1:63(7):853-67. doi: 10.1093/cid/ciw566. Epub
[PubMed PMID: 27621353]
Level 1 (high-level) evidence