Introduction
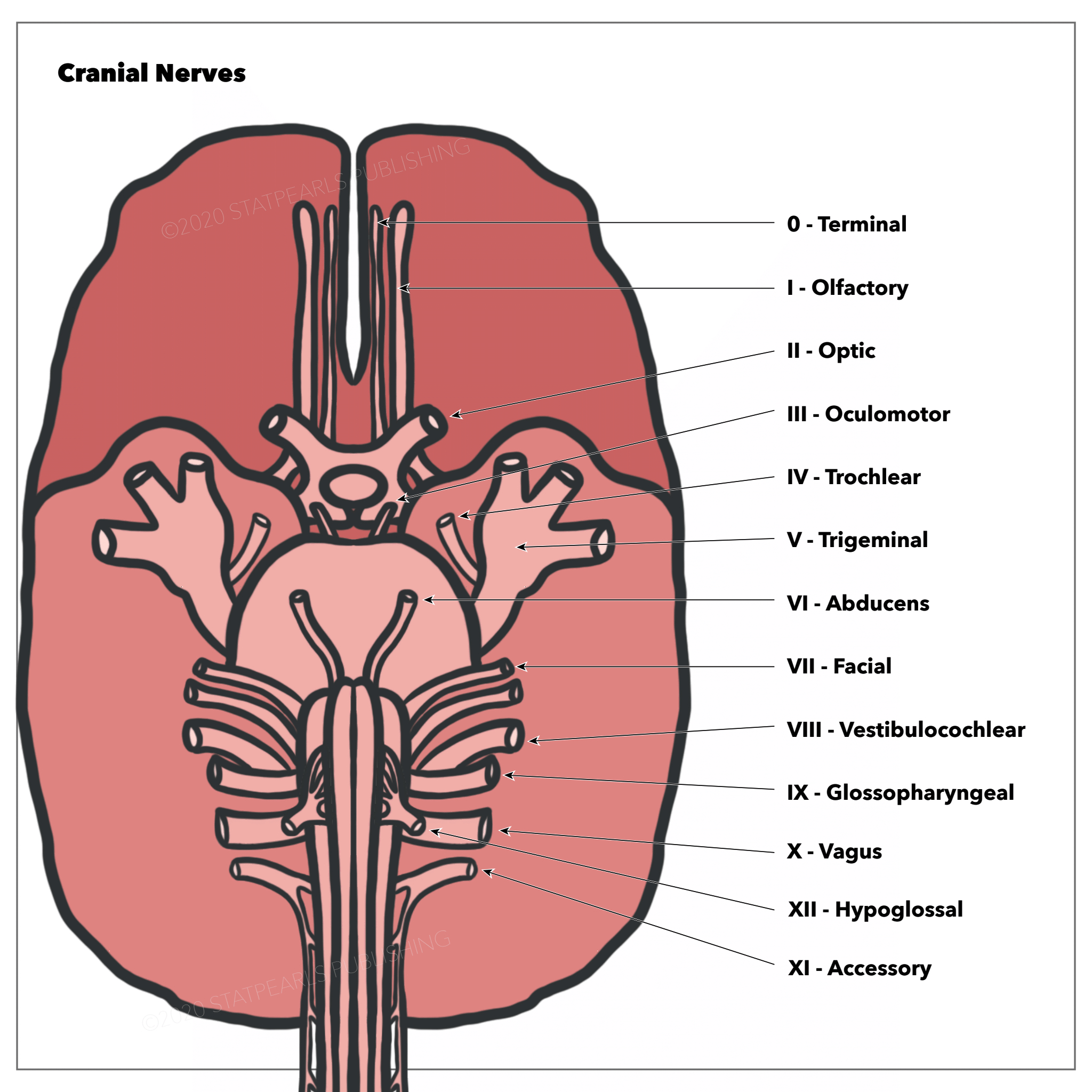
The optic nerve is the second cranial nerve (CN II) responsible for transmitting visual information. The optic nerve contains only afferent (sensory) fibers, and like all cranial nerves is paired. During embryogenesis, the optic nerve is formed in the retina, exits the orbit via the optic canal, and is relayed throughout the central nervous system (CNS). See Image. Optic Nerves Arising From Optic Canals. Synaptic targets of the optic nerve include, but are not limited to, the suprachiasmatic nucleus (SCN), lateral geniculate nucleus (LGN), pretectal nucleus, superior colliculus, and primary visual cortex. The stimulation of these different structures results in different functions. Furthermore, the optic nerve is the afferent limb for both the pupillary light reflex and the accommodation reflex.[1][2][3][3]
Structure and Function
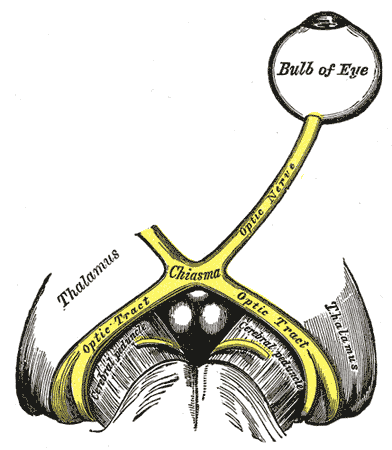
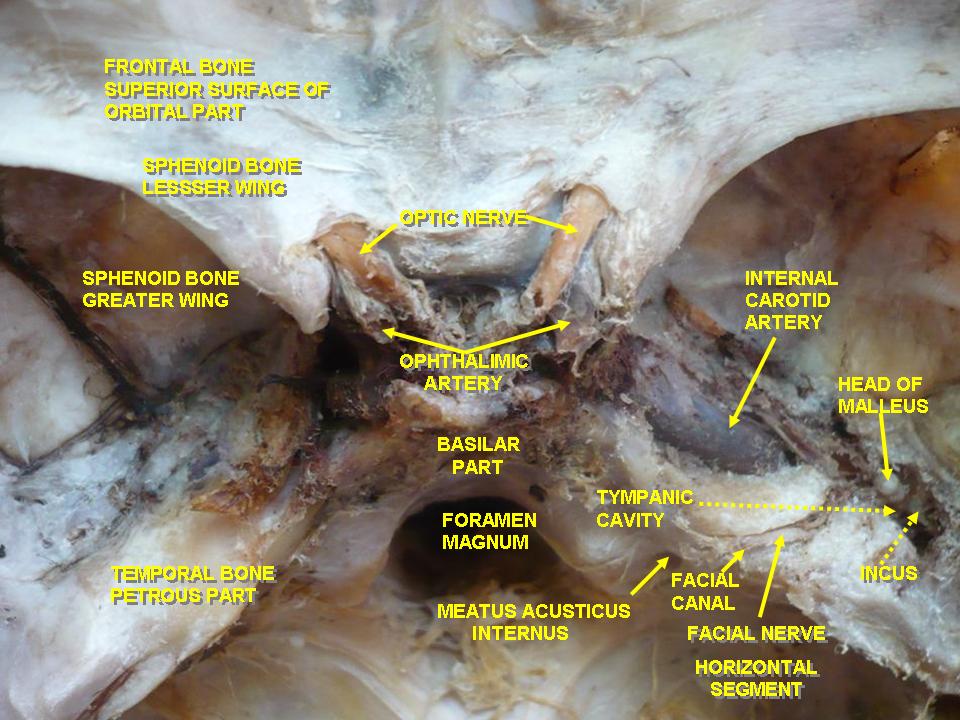
In the adult, the axons of about 1.2 million retinal ganglion cells converge at the optic disc to form the optic nerve. The optic disc is devoid of photoreceptors and therefore, forms a blind spot in each eye's visual field. At the optic disc, the optic nerve fibers exit the eye through fenestrations within the sclera, termed the lamina cribrosa. The retinal ganglion cell axons remain unmyelinated until traversing the lamina cribrosa. This is advantageous, as myelinated fibers within the retina would interfere with the path of incoming light. Accompanied by the ophthalmic artery, the optic nerve enters the bony skull through the optic foramen and travels within the optic canal to reach the middle cranial fossa. Located in the middle cranial fossa is the optic chiasm (see Image. The Cranial Nerves).
The optic nerves of both eyes meet at the optic chiasm and form the optic tracts (see Image. The Left Optic Nerve and the Optic Tracts). At the optic chiasm, the nasal retinal fibers from each optic nerve decussate (crossover) into the contralateral optic tract, while the temporal retinal fibers remain in the ipsilateral optic tract. The nasal retinal fibers relay sensory information from the temporal visual field of the corresponding eye. The temporal retinal fibers relay sensory information from the nasal visual field of the corresponding eye. Therefore, the optic tracts relay sensory information from the contralateral visual fields. For example, the right optic tract relays sensory information from the left visual field. Also, before forming the optic tracts, some nerve fibers from each eye will travel superiorly to synapse in the SCN located within the hypothalamus. This synaptic input influences the circadian rhythm controlled by the SCN.
The majority of the optic tract fibers synapse in the LGN of the thalamus. A small group of fibers synapses in the pretectal nucleus of the midbrain to initiate the pupillary light reflex. Another small group of fibers synapses in the superior colliculus to influence eye movements in response to environmental stimuli, such as noises and movements. Axons of the LGN form the optic radiations, also known as the geniculocalcarine tracts. The optic radiations are present bilaterally and are divided into superior and inferior divisions. The superior division (Baum’s loop) is composed of fibers from the superior retina, relaying sensory information from the inferior visual field. The inferior division (Meyer’s loop) is composed of fibers from the inferior retina, relaying sensory information from the superior visual field. Both divisions terminate in the primary visual cortex located around the calcarine sulcus of the occipital lobe. Visual information is processed in the primary visual cortex and is further analyzed through visual association areas within the CNS.[4][5][6][7][8][6]
Pupillary Light Reflex
The optic nerve is the afferent limb for the pupillary light reflex. When light is introduced to the eye, the photoreceptors become stimulated and propagate the signal to the retinal ganglion cells. The nasal retinal fibers relay the stimulus to the contralateral pretectal nucleus, and the temporal retinal fibers relay the stimulus to the ipsilateral pretectal nucleus. The fibers of both pretectal nuclei activate neurons in the Edinger-Westphal nucleus bilaterally, initiating the efferent limb of the reflex. The axons of these preganglionic parasympathetic neurons, travel along the periphery of the oculomotor nerve (CN III) and innervate the ciliary ganglion. The postganglionic parasympathetic axons of the ciliary ganglion form the short ciliary nerves. The short ciliary nerves stimulate the sphincter pupillae to constrict causing miosis. Due to the bilateral activation of both the pretectal nuclei and Edinger-Westphal nuclei, stimulation of one eye with light will cause pupillary constriction in the ipsilateral eye (direct response), as well as pupillary constriction in the contralateral eye (consensual response).[9]
Accommodation Reflex
The accommodation reflex is initiated by a transition of focus to a near object. The optic nerve is the afferent limb of this reflex. Unlike in the pupillary light reflex, an afferent stimulus is required to be relayed through the visual pathway reaching the primary visual cortex and visual association areas. The neurons in the visual association areas initiate the efferent limb of the reflex by stimulating the Edinger-Westphal nucleus and oculomotor nucleus located in the midbrain. Similar to the pupillary light reflex, pupillary constriction results from the activation of the two neuron system previously described. In addition to the stimulation of the sphincter pupillae, the short ciliary nerves also innervate the ciliary muscle. The contraction of the ciliary muscle causes the zonular fibers attached to the lens to relax. This relaxation results in the axial thickness to increase resulting in higher refractive power. Finally, the somatic fibers of the oculomotor nerves are responsible for the stimulation of the medial rectus muscle. Contraction of the medial rectus muscles will cause the adduction of the eyes, and when this coincides, convergence is achieved. Thus, the accommodation reflex is a combination of miosis, an increase in lens refractive power, and convergence.[10]
Embryology
The optic stalk is apparent during the fourth gestational week and ultimately becomes the optic nerve. The lumen of the optic stalk is continuous with the diencephalon of the forebrain proximally, and the optic vesicle distally. The lateral walls of the optic vesicle invaginate forming the optic cup, which develops into the retina. Invagination also occurs on the inferior surface of the optic stalk and vesicle, forming the choroidal fissure. Located within this fissure are the hyaloid artery and vein, which ultimately become the central retinal artery and vein after fusion of the fissure.
The optic nerve is a CNS structure arising from the diencephalon. As a CNS structure, the optic nerve is myelinated by oligodendrocytes and is ensheathed in the three meningeal layers. The myelination process begins centrally, and as discussed earlier, terminates at the lamina cribrosa. During or shortly following birth, nearly all optic nerve fibers will have been myelinated. The three meningeal layers surrounding the optic nerve are continuous with the three meningeal layers of the brain. As a result, increased intracranial pressure may propagate from the intracranial subarachnoid space to the perineural subarachnoid space, producing papilledema seen on fundoscopy.
Blood Supply and Lymphatics
The ophthalmic artery (OA) is the major artery perfusing the optic nerve and is the first branch of the internal carotid artery (ICA). The OA branches from the ICA just distal to the cavernous sinus and passes through the optic canal. Within the optic canal, the OA courses inferior and lateral to the optic nerve. Numerous branches arise from the OA, but the 2 primary branches are the central retinal artery (CRA) and posterior ciliary arteries (PCAs). The CRA is the first branch of the OA, running within the dura mater of the optic nerve. The CRA then pierces the optic nerve at around 12 mm posterior to the globe and courses anteriorly to perfuse the inner layers of the retina. Several PCAs arise from the OA. The PCAs course anteriorly and penetrate the sclera to perfuse the optic nerve and posterior uveal tract.
Surgical Considerations
The optic nerve sheath fenestration is one of the accepted treatment modalities in benign intracranial hypertension. Optic nerve sheath meningioma and optic nerve glioma may need surgical intervention, at times.
Clinical Significance
Compromise of the optic nerve results in visual field defects and/or visual loss. The type of visual field defect depends on which region of the optic pathway is disrupted. Damage to the optic nerve anterior to the optic chiasm results in ipsilateral monocular vision loss. Damage at the optic chiasm disrupts the nasal retinal fibers of both eyes resulting in a loss of both temporal visual fields. This is termed bitemporal hemianopia and may result from a pituitary adenoma compressing the optic chiasm. Damage to the optic tract results in contralateral homonymous hemianopia. For example, a lesion in the right optic tract results in a loss of the left visual fields of both eyes.
Damage to the optic nerve may also attenuate the pupillary light reflex resulting in an afferent pupillary defect (APD) or a relative afferent pupillary defect (RAPD), also known as a Marcus Gunn pupil. When light is directed at an eye with a healthy optic nerve, constriction of both pupils is achieved. When light is directed at an eye with an APD or RAPD, a reduced amount of constriction occurs. The difference in pupil constrictions can be appreciated by performing the swinging flashlight test. For example, when performing the swinging flashlight test on a patient with a right APD or RAPD, both pupils constrict appropriately when a light is directed into the left eye. When light is directed into the abnormal right eye, both pupils mildly constrict and appear to be dilated. The light is then directed back to the left eye and both pupils again constrict appropriately. The difference in pupil constrictions is a result of damaged afferent retinal fibers originating from the right eye. Pathologies affecting the optic nerve and severe retinal diseases may result in an RAPD. Optic neuropathies, such as glaucoma; optic neuritis, as seen in multiple sclerosis, and severe macular degeneration may all cause an RAPD.