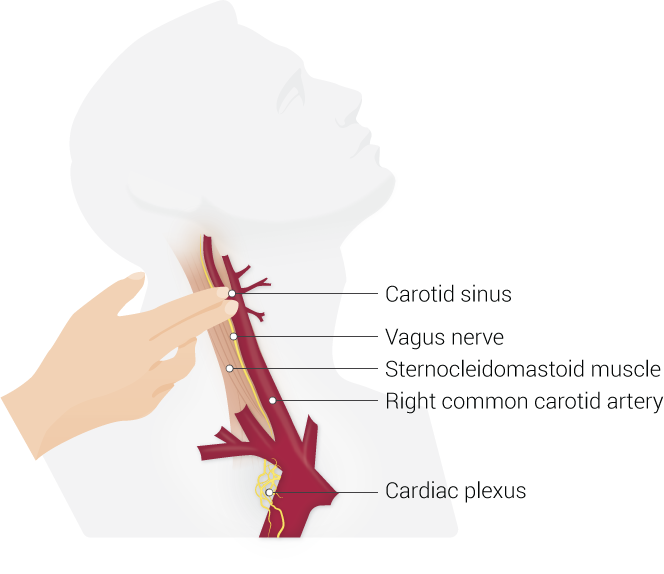
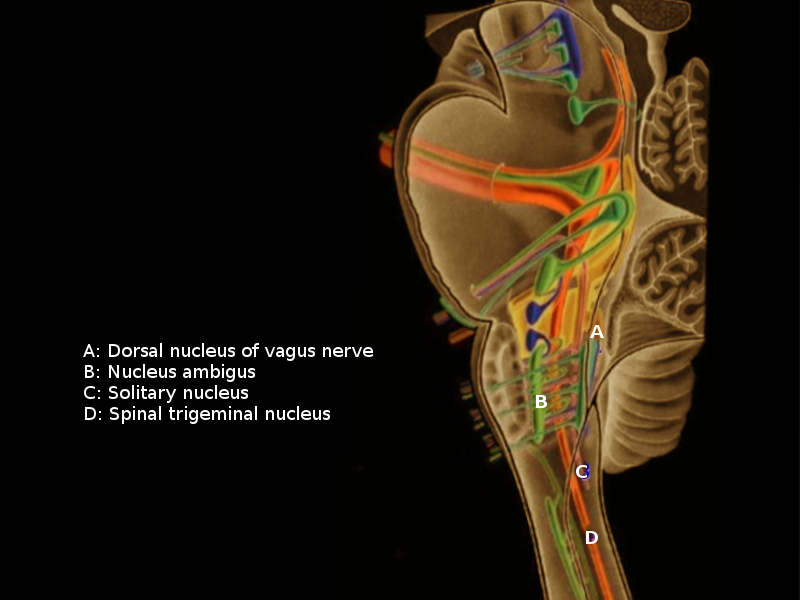
[1]
Lanska DJ. J.L. Corning and vagal nerve stimulation for seizures in the 1880s. Neurology. 2002 Feb 12:58(3):452-9
[PubMed PMID: 11839848]
[2]
Ohemeng KK, Parham K. Vagal Nerve Stimulation: Indications, Implantation, and Outcomes. Otolaryngologic clinics of North America. 2020 Feb:53(1):127-143. doi: 10.1016/j.otc.2019.09.008. Epub 2019 Nov 1
[PubMed PMID: 31685239]
[3]
Johnson RL, Wilson CG. A review of vagus nerve stimulation as a therapeutic intervention. Journal of inflammation research. 2018:11():203-213. doi: 10.2147/JIR.S163248. Epub 2018 May 16
[PubMed PMID: 29844694]
[4]
Ogbonnaya S, Kaliaperumal C. Vagal nerve stimulator: Evolving trends. Journal of natural science, biology, and medicine. 2013 Jan:4(1):8-13. doi: 10.4103/0976-9668.107254. Epub
[PubMed PMID: 23633829]
[5]
Wang J, Irnaten M, Neff RA, Venkatesan P, Evans C, Loewy AD, Mettenleiter TC, Mendelowitz D. Synaptic and neurotransmitter activation of cardiac vagal neurons in the nucleus ambiguus. Annals of the New York Academy of Sciences. 2001 Jun:940():237-46
[PubMed PMID: 11458681]
[7]
Ben-Menachem E. Modern management of epilepsy: Vagus nerve stimulation. Bailliere's clinical neurology. 1996 Dec:5(4):841-8
[PubMed PMID: 9068884]
[8]
Wilfong AA, Schultz RJ. Vagus nerve stimulation for treatment of epilepsy in Rett syndrome. Developmental medicine and child neurology. 2006 Aug:48(8):683-6
[PubMed PMID: 16836782]
[9]
Frost M, Gates J, Helmers SL, Wheless JW, Levisohn P, Tardo C, Conry JA. Vagus nerve stimulation in children with refractory seizures associated with Lennox-Gastaut syndrome. Epilepsia. 2001 Sep:42(9):1148-52
[PubMed PMID: 11580762]
[10]
Houser MV, Hennessy MD, Howard BC. Vagal nerve stimulator use during pregnancy for treatment of refractory seizure disorder. Obstetrics and gynecology. 2010 Feb:115(2 Pt 2):417-419. doi: 10.1097/AOG.0b013e3181bd1a8b. Epub
[PubMed PMID: 20093864]
[11]
Annegers JF, Coan SP, Hauser WA, Leestma J. Epilepsy, vagal nerve stimulation by the NCP system, all-cause mortality, and sudden, unexpected, unexplained death. Epilepsia. 2000 May:41(5):549-53
[PubMed PMID: 10802760]
[12]
Woodbury DM, Woodbury JW. Effects of vagal stimulation on experimentally induced seizures in rats. Epilepsia. 1990:31 Suppl 2():S7-19
[PubMed PMID: 2226368]
[13]
Lockard JS, Congdon WC, DuCharme LL. Feasibility and safety of vagal stimulation in monkey model. Epilepsia. 1990:31 Suppl 2():S20-6
[PubMed PMID: 2226362]
Level 2 (mid-level) evidence
[14]
Zabara J. Inhibition of experimental seizures in canines by repetitive vagal stimulation. Epilepsia. 1992 Nov-Dec:33(6):1005-12
[PubMed PMID: 1464256]
Level 3 (low-level) evidence
[15]
Pérez-Carbonell L, Faulkner H, Higgins S, Koutroumanidis M, Leschziner G. Vagus nerve stimulation for drug-resistant epilepsy. Practical neurology. 2020 May:20(3):189-198. doi: 10.1136/practneurol-2019-002210. Epub 2019 Dec 31
[PubMed PMID: 31892545]
[16]
Krahl SE, Clark KB, Smith DC, Browning RA. Locus coeruleus lesions suppress the seizure-attenuating effects of vagus nerve stimulation. Epilepsia. 1998 Jul:39(7):709-14
[PubMed PMID: 9670898]
[17]
Di Lazzaro V, Oliviero A, Pilato F, Saturno E, Dileone M, Meglio M, Colicchio G, Barba C, Papacci F, Tonali PA. Effects of vagus nerve stimulation on cortical excitability in epileptic patients. Neurology. 2004 Jun 22:62(12):2310-2
[PubMed PMID: 15210904]
[18]
O'Reardon JP, Cristancho P, Peshek AD. Vagus Nerve Stimulation (VNS) and Treatment of Depression: To the Brainstem and Beyond. Psychiatry (Edgmont (Pa. : Township)). 2006 May:3(5):54-63
[PubMed PMID: 21103178]
[19]
Harden CL, Pulver MC, Ravdin LD, Nikolov B, Halper JP, Labar DR. A Pilot Study of Mood in Epilepsy Patients Treated with Vagus Nerve Stimulation. Epilepsy & behavior : E&B. 2000 Apr:1(2):93-99
[PubMed PMID: 12609137]
Level 3 (low-level) evidence
[20]
Handforth A, DeGiorgio CM, Schachter SC, Uthman BM, Naritoku DK, Tecoma ES, Henry TR, Collins SD, Vaughn BV, Gilmartin RC, Labar DR, Morris GL 3rd, Salinsky MC, Osorio I, Ristanovic RK, Labiner DM, Jones JC, Murphy JV, Ney GC, Wheless JW. Vagus nerve stimulation therapy for partial-onset seizures: a randomized active-control trial. Neurology. 1998 Jul:51(1):48-55
[PubMed PMID: 9674777]
Level 1 (high-level) evidence
[21]
Martinez JM, Zboyan HA. Vagus nerve stimulation therapy in a patient with treatment-resistant depression: a case report of long-term follow-up and battery end-of-service. CNS spectrums. 2006 Feb:11(2):143-7
[PubMed PMID: 16520692]
Level 3 (low-level) evidence
[22]
Ashton AK. Depressive relapse after vagal nerve stimulator explantation. The American journal of psychiatry. 2010 Jun:167(6):719-20. doi: 10.1176/appi.ajp.2010.10010020. Epub
[PubMed PMID: 20516167]
[23]
Berry SM, Broglio K, Bunker M, Jayewardene A, Olin B, Rush AJ. A patient-level meta-analysis of studies evaluating vagus nerve stimulation therapy for treatment-resistant depression. Medical devices (Auckland, N.Z.). 2013:6():17-35. doi: 10.2147/MDER.S41017. Epub 2013 Mar 1
[PubMed PMID: 23482508]
[24]
Nemeroff CB, Mayberg HS, Krahl SE, McNamara J, Frazer A, Henry TR, George MS, Charney DS, Brannan SK. VNS therapy in treatment-resistant depression: clinical evidence and putative neurobiological mechanisms. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2006 Jul:31(7):1345-55
[PubMed PMID: 16641939]
[26]
Pavlov VA, Tracey KJ. The cholinergic anti-inflammatory pathway. Brain, behavior, and immunity. 2005 Nov:19(6):493-9
[PubMed PMID: 15922555]
[27]
Bonaz B, Sinniger V, Pellissier S. Anti-inflammatory properties of the vagus nerve: potential therapeutic implications of vagus nerve stimulation. The Journal of physiology. 2016 Oct 15:594(20):5781-5790. doi: 10.1113/JP271539. Epub 2016 May 1
[PubMed PMID: 27059884]
[28]
Pavlov VA, Tracey KJ. The vagus nerve and the inflammatory reflex--linking immunity and metabolism. Nature reviews. Endocrinology. 2012 Dec:8(12):743-54. doi: 10.1038/nrendo.2012.189. Epub
[PubMed PMID: 23169440]
[29]
Tariq K, Das JM, Monaghan S, Miserocchi A, McEvoy A. A case report of Vagus nerve stimulation for intractable hiccups. International journal of surgery case reports. 2021 Jan:78():219-222. doi: 10.1016/j.ijscr.2020.12.023. Epub 2020 Dec 16
[PubMed PMID: 33360634]
Level 3 (low-level) evidence
[30]
Garcia RG, Lin RL, Lee J, Kim J, Barbieri R, Sclocco R, Wasan AD, Edwards RR, Rosen BR, Hadjikhani N, Napadow V. Modulation of brainstem activity and connectivity by respiratory-gated auricular vagal afferent nerve stimulation in migraine patients. Pain. 2017 Aug:158(8):1461-1472. doi: 10.1097/j.pain.0000000000000930. Epub
[PubMed PMID: 28541256]
[31]
Cook DN, Thompson S, Stomberg-Firestein S, Bikson M, George MS, Jenkins DD, Badran BW. Design and validation of a closed-loop, motor-activated auricular vagus nerve stimulation (MAAVNS) system for neurorehabilitation. Brain stimulation. 2020 May-Jun:13(3):800-803. doi: 10.1016/j.brs.2020.02.028. Epub 2020 Feb 27
[PubMed PMID: 32289710]
Level 1 (high-level) evidence
[32]
Saper CB, Kibbe MR, Hurley KM, Spencer S, Holmes HR, Leahy KM, Needleman P. Brain natriuretic peptide-like immunoreactive innervation of the cardiovascular and cerebrovascular systems in the rat. Circulation research. 1990 Dec:67(6):1345-54
[PubMed PMID: 1978807]
[34]
McGregor A, Wheless J, Baumgartner J, Bettis D. Right-sided vagus nerve stimulation as a treatment for refractory epilepsy in humans. Epilepsia. 2005 Jan:46(1):91-6
[PubMed PMID: 15660773]
[35]
Couch JD, Gilman AM, Doyle WK. Long-term Expectations of Vagus Nerve Stimulation: A Look at Battery Replacement and Revision Surgery. Neurosurgery. 2016 Jan:78(1):42-6. doi: 10.1227/NEU.0000000000000985. Epub
[PubMed PMID: 26678088]
[36]
Giordano F, Zicca A, Barba C, Guerrini R, Genitori L. Vagus nerve stimulation: Surgical technique of implantation and revision and related morbidity. Epilepsia. 2017 Apr:58 Suppl 1():85-90. doi: 10.1111/epi.13678. Epub
[PubMed PMID: 28386925]
[37]
Tronnier VM. Vagus Nerve Stimulation: Surgical Technique and Complications. Progress in neurological surgery. 2015:29():29-38. doi: 10.1159/000434653. Epub 2015 Sep 4
[PubMed PMID: 26393499]
[38]
Denski KM, Labiner DM. Should I offer vagus nerve stimulation as part of my neurology practice? Neurology. Clinical practice. 2014 Aug:4(4):313-318. doi: 10.1212/CPJ.0000000000000048. Epub
[PubMed PMID: 29473565]
[39]
Veldsema-Currie RD, Labruyère WT, Langemeijer MW. Depletion of total acetylcholine by hemicholinium-3 in isolated rat diaphragm is less in the presence of dexamethasone. Brain research. 1984 Dec 24:324(2):305-12
[PubMed PMID: 6529621]
[40]
Kim W, Clancy RR, Liu GT. Horner syndrome associated with implantation of a vagus nerve stimulator. American journal of ophthalmology. 2001 Mar:131(3):383-4
[PubMed PMID: 11239877]
[41]
. A randomized controlled trial of chronic vagus nerve stimulation for treatment of medically intractable seizures. The Vagus Nerve Stimulation Study Group. Neurology. 1995 Feb:45(2):224-30
[PubMed PMID: 7854516]
Level 1 (high-level) evidence
[42]
Fetzer S, Dibué M, Nagel AM, Trollmann R. A systematic review of magnetic resonance imaging in patients with an implanted vagus nerve stimulation system. Neuroradiology. 2021 Sep:63(9):1407-1417. doi: 10.1007/s00234-021-02705-y. Epub 2021 Apr 12
[PubMed PMID: 33846830]
Level 1 (high-level) evidence
[43]
Jain P, Arya R. Vagus Nerve Stimulation and Seizure Outcomes in Pediatric Refractory Epilepsy: Systematic Review and Meta-Analysis. Neurology. 2021 Apr 13:():. pii: 10.1212/WNL.0000000000012030. doi: 10.1212/WNL.0000000000012030. Epub 2021 Apr 13
[PubMed PMID: 33849993]
Level 1 (high-level) evidence
[44]
Mu Q, Bohning DE, Nahas Z, Walker J, Anderson B, Johnson KA, Denslow S, Lomarev M, Moghadam P, Chae JH, George MS. Acute vagus nerve stimulation using different pulse widths produces varying brain effects. Biological psychiatry. 2004 Apr 15:55(8):816-25
[PubMed PMID: 15050863]
[45]
Borland MS, Engineer CT, Vrana WA, Moreno NA, Engineer ND, Vanneste S, Sharma P, Pantalia MC, Lane MC, Rennaker RL, Kilgard MP. The Interval Between VNS-Tone Pairings Determines the Extent of Cortical Map Plasticity. Neuroscience. 2018 Jan 15:369():76-86. doi: 10.1016/j.neuroscience.2017.11.004. Epub 2017 Nov 10
[PubMed PMID: 29129793]
[46]
Nesbitt AD, Marin JC, Tompkins E, Ruttledge MH, Goadsby PJ. Initial use of a novel noninvasive vagus nerve stimulator for cluster headache treatment. Neurology. 2015 Mar 24:84(12):1249-53. doi: 10.1212/WNL.0000000000001394. Epub 2015 Feb 20
[PubMed PMID: 25713002]
[47]
Shi C, Flanagan SR, Samadani U. Vagus nerve stimulation to augment recovery from severe traumatic brain injury impeding consciousness: a prospective pilot clinical trial. Neurological research. 2013 Apr:35(3):263-76. doi: 10.1179/1743132813Y.0000000167. Epub
[PubMed PMID: 23485054]
Level 3 (low-level) evidence
[48]
Yakunina N, Nam EC. Direct and Transcutaneous Vagus Nerve Stimulation for Treatment of Tinnitus: A Scoping Review. Frontiers in neuroscience. 2021:15():680590. doi: 10.3389/fnins.2021.680590. Epub 2021 May 28
[PubMed PMID: 34122002]
Level 2 (mid-level) evidence
[49]
Slater C, Wang Q. Alzheimer's disease: An evolving understanding of noradrenergic involvement and the promising future of electroceutical therapies. Clinical and translational medicine. 2021 Apr:11(4):e397. doi: 10.1002/ctm2.397. Epub
[PubMed PMID: 33931975]
Level 3 (low-level) evidence
[50]
Mondal B, Choudhury S, Banerjee R, Roy A, Chatterjee K, Basu P, Singh R, Halder S, Shubham S, Baker SN, Baker MR, Kumar H. Non-invasive vagus nerve stimulation improves clinical and molecular biomarkers of Parkinson's disease in patients with freezing of gait. NPJ Parkinson's disease. 2021 May 27:7(1):46. doi: 10.1038/s41531-021-00190-x. Epub 2021 May 27
[PubMed PMID: 34045464]
[51]
van Hoorn A, Carpenter T, Oak K, Laugharne R, Ring H, Shankar R. Neuromodulation of autism spectrum disorders using vagal nerve stimulation. Journal of clinical neuroscience : official journal of the Neurosurgical Society of Australasia. 2019 May:63():8-12. doi: 10.1016/j.jocn.2019.01.042. Epub 2019 Feb 4
[PubMed PMID: 30732986]
[52]
Derakhshan N, Yaghmaei S, Keshavarz P. Vagal nerve stimulation for the treatment of male factor infertility. Andrologia. 2021 Jul:53(6):e14043. doi: 10.1111/and.14043. Epub 2021 Apr 30
[PubMed PMID: 33929756]
[53]
van Beekum CJ, Willis MA, von Websky MW, Sommer NP, Kalff JC, Wehner S, Vilz TO. Electrical vagus nerve stimulation as a prophylaxis for SIRS and postoperative ileus. Autonomic neuroscience : basic & clinical. 2021 Nov:235():102857. doi: 10.1016/j.autneu.2021.102857. Epub 2021 Jul 29
[PubMed PMID: 34343825]
[54]
Bonaz B, Sinniger V, Pellissier S. Therapeutic Potential of Vagus Nerve Stimulation for Inflammatory Bowel Diseases. Frontiers in neuroscience. 2021:15():650971. doi: 10.3389/fnins.2021.650971. Epub 2021 Mar 22
[PubMed PMID: 33828455]
[55]
Brock C, Rasmussen SE, Drewes AM, Møller HJ, Brock B, Deleuran B, Farmer AD, Pfeiffer-Jensen M. Vagal Nerve Stimulation-Modulation of the Anti-Inflammatory Response and Clinical Outcome in Psoriatic Arthritis or Ankylosing Spondylitis. Mediators of inflammation. 2021:2021():9933532. doi: 10.1155/2021/9933532. Epub 2021 May 27
[PubMed PMID: 34135691]
Level 2 (mid-level) evidence
[56]
Dasheiff RM, Sandberg T, Thompson J, Arrambide S, E03 and E05 Cooperative Study Group. Vagal nerve stimulation does not unkindle seizures. Journal of clinical neurophysiology : official publication of the American Electroencephalographic Society. 2001 Jan:18(1):68-74
[PubMed PMID: 11290941]
[57]
Thompson SL, O'Leary GH, Austelle CW, Gruber E, Kahn AT, Manett AJ, Short B, Badran BW. A Review of Parameter Settings for Invasive and Non-invasive Vagus Nerve Stimulation (VNS) Applied in Neurological and Psychiatric Disorders. Frontiers in neuroscience. 2021:15():709436. doi: 10.3389/fnins.2021.709436. Epub 2021 Jul 13
[PubMed PMID: 34326720]